Abstract
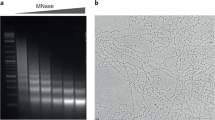
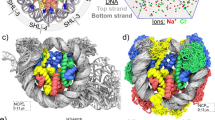
Local nucleosome-nucleosome interactions in cis drive chromatin folding, whereas interactions in trans lead to fiber-fiber oligomerization. Here we show that peptides derived from the histone H4 tail and Kaposi's sarcoma herpesvirus LANA protein can replace the endogenous H4 tail, resulting in array folding and oligomerization. Neutralization of a LANA binding site on the histone surface enhanced rather than abolished nucleosome-nucleosome interactions. We maintain that the contoured nucleosome surface is centrally involved in regulating chromatin condensation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Barbera, A.J. et al. Science 311, 856–861 (2006).
Dorigo, B. et al. Science 306, 1571–1573 (2004).
Gordon, F., Luger, K. & Hansen, J.C. J. Biol. Chem. 280, 33701–33706 (2005).
Hansen, J.C. Annu. Rev. Biophys. Biomol. Struct. 31, 361–392 (2002).
Schwarz, P.M., Felthauser, A., Fletcher, T.M. & Hansen, J.C. Biochemistry 35, 4009–4015 (1996).
Dorigo, B., Schalch, T., Bystricky, K. & Richmond, T.J. J. Mol. Biol. 327, 85–96 (2003).
Cary, P.D., Crane-Robinson, C., Bradbury, E.M. & Dixon, G.H. Eur. J. Biochem. 127, 137–143 (1982).
Luger, K., Maeder, A.W., Richmond, R.K., Sargent, D.F. & Richmond, T.J. Nature 389, 251–259 (1997).
Narita, M. et al. Cell 113, 703–716 (2003).
Shogren-Knaak, M. et al. Science 311, 844–847 (2006).
Freitas, M.A., Sklenar, A.R. & Parthun, M.R. J. Cell. Biochem. 92, 691–700 (2004).
Park, J.H., Cosgrove, M.S., Youngman, E., Wolberger, C. & Boeke, J.D. Nat. Genet. 32, 273–279 (2002).
Matsubara, K., Sano, N., Umehara, T. & Horikoshi, M. Genes Cells 12, 13–33 (2007).
Suto, R.K., Clarkson, M.J., Tremethick, D.J. & Luger, K. Nat. Struct. Biol. 7, 1121–1124 (2000).
Nicholls, A., Sharp, K.A. & Honig, B. Proteins 11, 281–296 (1991).
DeLano, W.L. The PyMOL User's Manual (DeLano Scientific, San Carlos, California, USA, 2002).
Acknowledgements
We thank P.N. Dyer for help with the preparation of histones and DNA. Supported by US National Institutes of Health grants GM067777 to K.L., CA82036 to K.M.K. and GM45916 to J.C.H. K.L. is also supported by the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–3, Supplementary Table 1, Supplementary Methods (PDF 154 kb)
Rights and permissions
About this article
Cite this article
Chodaparambil, J., Barbera, A., Lu, X. et al. A charged and contoured surface on the nucleosome regulates chromatin compaction. Nat Struct Mol Biol 14, 1105–1107 (2007). https://doi.org/10.1038/nsmb1334
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb1334
This article is cited by
-
The solid and liquid states of chromatin
Epigenetics & Chromatin (2021)
-
Nucleosome plasticity is a critical element of chromatin liquid–liquid phase separation and multivalent nucleosome interactions
Nature Communications (2021)
-
Mapping the electrostatic potential of the nucleosome acidic patch
Scientific Reports (2021)
-
Nucleosome structure and dynamics are coming of age
Nature Structural & Molecular Biology (2019)
-
Histone variants: critical determinants in tumour heterogeneity
Frontiers of Medicine (2019)