Abstract
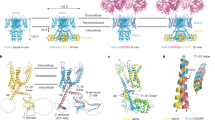
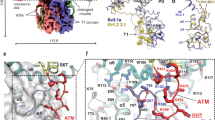
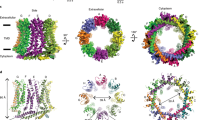
Brain IA and cardiac Ito currents arise from complexes containing Kv4 voltage-gated potassium channels and cytoplasmic calcium-sensor proteins (KChIPs). Here, we present X-ray crystallographic and small-angle X-ray scattering data that show that the KChIP1–Kv4.3 N-terminal cytoplasmic domain complex is a cross-shaped octamer bearing two principal interaction sites. Site 1 comprises interactions between a unique Kv4 channel N-terminal hydrophobic segment and a hydrophobic pocket formed by displacement of the KChIP H10 helix. Site 2 comprises interactions between a T1 assembly domain loop and the KChIP H2 helix. Functional and biochemical studies indicate that site 1 influences channel trafficking, whereas site 2 affects channel gating, and that calcium binding is intimately linked to KChIP folding and complex formation. Together, the data resolve how Kv4 channels and KChIPs interact and provide a framework for understanding how KChIPs modulate Kv4 function.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Hille, B. Ion Channels of Excitable Membranes (Sinauer Associates, Inc., Sunderland, Massachusetts, USA, 2001).
Roux, B. Ion conduction and selectivity in K(+) channels. Annu. Rev. Biophys. Biomol. Struct. 34, 153–171 (2005).
Tombola, F., Pathak, M.M. & Isacoff, E.Y. How far will you go to sense voltage? Neuron 48, 719–725 (2005).
Levitan, I.B. Signaling protein complexes associated with neuronal ion channels. Nat. Neurosci. 9, 305–310 (2006).
Rosati, B. & McKinnon, D. Regulation of ion channel expression. Circ. Res. 94, 874–883 (2004).
Birnbaum, S.G. et al. Structure and function of Kv4-family transient potassium channels. Physiol. Rev. 84, 803–833 (2004).
Patel, S.P. & Campbell, D.L. Transient outward potassium current, 'Ito', phenotypes in the mammalian left ventricle: underlying molecular, cellular and biophysical mechanisms. J. Physiol. (Lond.) 569, 7–39 (2005).
An, W.F. et al. Modulation of A-type potassium channels by a family of calcium sensors. Nature 403, 553–556 (2000).
Rhodes, K.J. et al. KChIPs and Kv4 alpha subunits as integral components of A-type potassium channels in mammalian brain. J. Neurosci. 24, 7903–7915 (2004).
Guo, W., Malin, S.A., Johns, D.C., Jeromin, A. & Nerbonne, J.M. Modulation of Kv4-encoded K(+) currents in the mammalian myocardium by neuronal calcium sensor-1. J. Biol. Chem. 277, 26436–26443 (2002).
Nakamura, T.Y. et al. A role for frequenin, a Ca2+-binding protein, as a regulator of Kv4 K+-currents. Proc. Natl. Acad. Sci. USA 98, 12808–12813 (2001).
Burgoyne, R.D., O'Callaghan, D.W., Hasdemir, B., Haynes, L.P. & Tepikin, A.V. Neuronal Ca2+-sensor proteins: multitalented regulators of neuronal function. Trends Neurosci. 27, 203–209 (2004).
Bahring, R. et al. Conserved Kv4 N-terminal domain critical for effects of Kv channel-interacting protein 2.2 on channel expression and gating. J. Biol. Chem. 276, 23888–23894 (2001).
Holmqvist, M.H. et al. Elimination of fast inactivation in Kv4 A-type potassium channels by an auxiliary subunit domain. Proc. Natl. Acad. Sci. USA 99, 1035–1040 (2002).
Shibata, R. et al. A fundamental role for KChIPs in determining the molecular properties and trafficking of Kv4.2 potassium channels. J. Biol. Chem. 278, 36445–36454 (2003).
Patel, S.P., Parai, R., Parai, R. & Campbell, D.L. Regulation of Kv4.3 voltage-dependent gating kinetics by KChIP2 isoforms. J. Physiol. (Lond.) 557, 19–41 (2004).
Scannevin, R.H. et al. Two N-terminal domains of Kv4 K(+) channels regulate binding to and modulation by KChIP1. Neuron 41, 587–598 (2004).
Salvador-Recatala, V., Gallin, W.J., Abbruzzese, J., Ruben, P.C. & Spencer, A.N. A potassium channel (Kv4) cloned from the heart of the tunicate Ciona intestinalis and its modulation by a KChIP subunit. J. Exp. Biol. 209, 731–747 (2006).
Hatano, N., Ohya, S. & Imaizumi, Y. Functional interaction between KChIP1 and GFP-fused Kv4.3L co-expressed in HEK293 cells. Pflugers Arch. 444, 80–88 (2002).
Kuo, H.C. et al. A defect in the Kv channel-interacting protein 2 (KChIP2) gene leads to a complete loss of I(to) and confers susceptibility to ventricular tachycardia. Cell 107, 801–813 (2001).
Long, S.B., Campbell, E.B. & Mackinnon, R. Crystal structure of a mammalian voltage-dependent Shaker family K+ channel. Science 309, 897–903 (2005).
Kim, L.A. et al. Three-dimensional structure of I(to); Kv4.2-KChIP2 ion channels by electron microscopy at 21 Angstrom resolution. Neuron 41, 513–519 (2004).
Zhou, W., Qian, Y., Kunjilwar, K., Pfaffinger, P.J. & Choe, S. Structural insights into the functional interaction of KChIP1 with Shal-type K(+) channels. Neuron 41, 573–586 (2004).
Ren, X., Shand, S.H. & Takimoto, K. Effective association of Kv channel-interacting proteins with Kv4 channel is mediated with their unique core peptide. J. Biol. Chem. 278, 43564–43570 (2003).
Derewenda, Z.S. & Vekilov, P.G. Entropy and surface engineering in protein crystallization. Acta Crystallogr. D Biol. Crystallogr. 62, 116–124 (2006).
Kunjilwar, K., Strang, C., DeRubeis, D. & Pfaffinger, P.J. KChIP3 rescues the functional expression of Shal channel tetramerization mutants. J. Biol. Chem. 279, 54542–54551 (2004).
Bourne, Y., Dannenberg, J., Pollmann, V., Marchot, P. & Pongs, O. Immunocytochemical localization and crystal structure of human frequenin (neuronal calcium sensor 1). J. Biol. Chem. 276, 11949–11955 (2001).
Callsen, B. et al. Contribution of N- and C-terminal channel domains to Kv4.2 domains to KChIP interaction. J. Physiol. (Lond.) [corrected] 568, 397–412 (2005).
Svergun, D.I. & Koch, M.H.J. Small-angle scattering studies of biological macromolecules in solution. Rep. Prog. Phys. 66, 1735–1782 (2003).
Svergun, D.I. Determination of the regularization parameter in indirect-transform methods using perceptual criteria. J. Appl. Cryst. 25, 495–503 (1992).
Svergun, D.I., Barberato, C. & Koch, M.H.J. CRYSOL—a program to evaluate X-ray solution scattering of biological macromolecules from atomic coordinates. J. Appl. Cryst. 28, 768–773 (1995).
Svergun, D.I., Petoukhov, M.V. & Koch, M.H. Determination of domain structure of proteins from X-ray solution scattering. Biophys. J. 80, 2946–2953 (2001).
Volkov, V.V. & Svergun, D.I. Uniqueness of ab initio shape determination in small-angle scattering. J. Appl. Cryst. 36, 860–864 (2003).
O'Callaghan, D.W., Hasdemir, B., Leighton, M. & Burgoyne, R.D. Residues within the myristoylation motif determine intracellular targeting of the neuronal Ca2+ sensor protein KChIP1 to post-ER transport vesicles and traffic of Kv4 K+ channels. J. Cell Sci. 116, 4833–4845 (2003).
Minor, D.L., Jr. et al. The polar T1 interface is linked to conformational changes that open the voltage-gated potassium channel. Cell 102, 657–670 (2000).
Jerng, H.H. & Covarrubias, M.K. + channel inactivation mediated by the concerted action of the cytoplasmic N- and C-terminal domains. Biophys. J. 72, 163–174 (1997).
Bahring, R., Boland, L.M., Varghese, A., Gebauer, M. & Pongs, O. Kinetic analysis of open- and closed-state inactivation transitions in human Kv4.2 A-type potassium channels. J. Physiol. (Lond.) 535, 65–81 (2001).
Gebauer, M. et al. N-type inactivation features of Kv4.2 channel gating. Biophys. J. 86, 210–223 (2004).
Wang, G. & Covarrubias, M. Voltage-dependent gating rearrangements in the intracellular T1–T1 interface of a K+ channel. J. Gen. Physiol. 127, 391–400 (2006).
Wang, G. et al. Functionally active t1-t1 interfaces revealed by the accessibility of intracellular thiolate groups in kv4 channels. J. Gen. Physiol. 126, 55–69 (2005).
Decher, N., Barth, A.S., Gonzalez, T., Steinmeyer, K. & Sanguinetti, M.C. Novel KChIP2 isoforms increase functional diversity of transient outward potassium currents. J. Physiol. (Lond.) 557, 761–772 (2004).
Deschenes, I. et al. Regulation of Kv4.3 current by KChIP2 splice variants: a component of native cardiac I(to)? Circulation 106, 423–429 (2002).
Patel, S.P., Campbell, D.L., Morales, M.J. & Strauss, H.C. Heterogeneous expression of KChIP2 isoforms in the ferret heart. J. Physiol. (Lond.) 539, 649–656 (2002).
Patel, S.P., Campbell, D.L. & Strauss, H.C. Elucidating KChIP effects on Kv4.3 inactivation and recovery kinetics with a minimal KChIP2 isoform. J. Physiol. (Lond.) 545, 5–11 (2002).
Bahring, R. et al. Coupling of voltage-dependent potassium channel inactivation and oxidoreductase active site of Kvbeta subunits. J. Biol. Chem. 276, 22923–22929 (2001).
Rettig, J. et al. Inactivation properties of voltage-gated K+ channels altered by presence of beta-subunit. Nature 369, 289–294 (1994).
Gu, C., Jan, Y.N. & Jan, L.Y. A conserved domain in axonal targeting of Kv1 (Shaker) voltage-gated potassium channels. Science 301, 646–649 (2003).
Nakahira, K., Shi, G., Rhodes, K.J. & Trimmer, J.S. Selective interaction of voltage-gated K+ channel beta-subunits with alpha-subunits. J. Biol. Chem. 271, 7084–7089 (1996).
Yang, E.K., Alvira, M.R., Levitan, E.S. & Takimoto, K. Kvbeta subunits increase expression of Kv4.3 channels by interacting with their C termini. J. Biol. Chem. 276, 4839–4844 (2001).
Deschenes, I. & Tomaselli, G.F. Modulation of Kv4.3 current by accessory subunits. FEBS Lett. 528, 183–188 (2002).
Hasdemir, B., Fitzgerald, D.J., Prior, I.A., Tepikin, A.V. & Burgoyne, R.D. Traffic of Kv4 K+ channels mediated by KChIP1 is via a novel post-ER vesicular pathway. J. Cell Biol. 171, 459–469 (2005).
Rosati, B. et al. Concordant expression of KChIP2 mRNA, protein and transient outward current throughout the canine ventricle. J. Physiol. (Lond.) 548, 815–822 (2003).
Rosati, B. et al. Regulation of KChIP2 potassium channel beta subunit gene expression underlies the gradient of transient outward current in canine and human ventricle. J. Physiol. (Lond.) 533, 119–125 (2001).
Kaab, S. et al. Molecular basis of transient outward potassium current downregulation in human heart failure: a decrease in Kv4.3 mRNA correlates with a reduction in current density. Circulation 98, 1383–1393 (1998).
Bowlby, M.R. et al. Identification and characterization of small molecule modulators of KChIP/Kv4 function. Bioorg. Med. Chem. 13, 6112–6119 (2005).
Van Petegem, F., Clark, K.A., Chatelain, F.C. & Minor, D.L., Jr. Structure of a complex between a voltage-gated calcium channel beta-subunit and an alpha-subunit domain. Nature 429, 671–675 (2004).
Kholod, N. & Mustelin, T. Novel vectors for co-expression of two proteins in E. coli. Biotechniques 31, 322–3, 326–8 (2001).
Minor, D.L., Jr., Masseling, S.J., Jan, Y.N. & Jan, L.Y. Transmembrane structure of an inwardly rectifying potassium channel. Cell 96, 879–891 (1999).
Nakamura, T.Y. et al. Different effects of the Ca(2+)-binding protein, KChIP1, on two Kv4 subfamily members, Kv4.1 and Kv4.2. FEBS Lett. 499, 205–209 (2001).
Acknowledgements
We thank D. Julius and F. Van Petegem for comments on the manuscript, J. Holton at ALS beamline 8.3.1 for data collection help, L.Y. Jan (University of California San Francisco) for the Kv4.3 clone and Minor laboratory members for support. This work was supported by awards to D.L.M. from the McKnight Foundation for Neuroscience, Rita Allen Foundation, Searle Scholars Award, Arnold and Mabel Beckman Foundation and US National Institutes of Health. G.L.H. was supported by US National Cancer Institute grant CA92584 and US Department of Energy contract DE-AC03-76SF00098. D.L.M. is a McKnight Scholar, Rita Allen Scholar, Searle Scholar and Beckman Young Investigator.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Monomer of the KChIP1-T1N fusion, superposition of frequenin EF2 and KChIP1 EF2 and frequenin structure. (PDF 91 kb)
Supplementary Fig. 2
T1N helix and linker structure. (PDF 562 kb)
Supplementary Fig. 3
Solution analysis of the KChIP1–Kv4.3 T1 complex. (PDF 113 kb)
Supplementary Fig. 4
Gel-filtration elution profiles for purified KChIP37–216 and EF mutants. (PDF 186 kb)
Rights and permissions
About this article
Cite this article
Pioletti, M., Findeisen, F., Hura, G. et al. Three-dimensional structure of the KChIP1–Kv4.3 T1 complex reveals a cross-shaped octamer. Nat Struct Mol Biol 13, 987–995 (2006). https://doi.org/10.1038/nsmb1164
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb1164
This article is cited by
-
Structural basis for the gating modulation of Kv4.3 by auxiliary subunits
Cell Research (2022)
-
Structural basis of gating modulation of Kv4 channel complexes
Nature (2021)
-
Crystal structure of a Ca2+-dependent regulator of flagellar motility reveals the open-closed structural transition
Scientific Reports (2018)
-
Modulation of human Kv4.3/KChIP2 channel inactivation kinetics by cytoplasmic Ca2+
Pflügers Archiv - European Journal of Physiology (2017)
-
T-type channels buddy up
Pflügers Archiv - European Journal of Physiology (2014)