Abstract
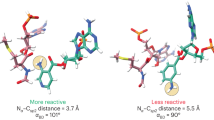
α-sarcin ribotoxins comprise a unique family of ribonucleases that cripple the ribosome by catalyzing endoribonucleolytic cleavage of ribosomal RNA at a specific location in the sarcin/ricin loop (SRL). The SRL structure alone is cleaved site-specifically by the ribotoxin, but the ribosomal context enhances the reaction rate by several orders of magnitude. We show that, for the α-sarcin–like ribotoxin restrictocin, this catalytic advantage arises from favorable electrostatic interactions with the ribosome. Restrictocin binds at many sites on the ribosomal surface and under certain conditions cleaves the SRL with a second-order rate constant of 1.7 × 1010 M−1 s−1, a value that matches the predicted frequency of random restrictocin-ribosome encounters. The results suggest a mechanism of target location whereby restrictocin encounters ribosomes randomly and diffuses within the ribosomal electrostatic field to the SRL. These studies show a role for electrostatics in protein-ribosome recognition.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Jennings, J.C., Olson, B.H., Roga, V., Junek, A.J. & Schuurmans, D.M. Alpha sarcin, a new antitumor agent. II. Fermentation and antitumor spectrum. Appl. Microbiol. 13, 322–326 (1965).
Turnay, J., Olmo, N., Jimenez, A., Lizarbe, M.A. & Gavilanes, J.G. Kinetic study of the cytotoxic effect of alpha-sarcin, a ribosome inactivating protein from Aspergillus giganteus, on tumour cell lines: protein biosynthesis inhibition and cell binding. Mol. Cell. Biochem. 122, 39–47 (1993).
Wool, I.G. Structure and mechanism of action of the cytotoxic ribonuclease α-sarcin. in Ribonucleases: Structures and Functions (eds. D'Alesio, G. & Riordan, J.F.) 131–162 (Academic Press, New York, 1997).
Olmo, N. et al. Cytotoxic mechanism of the ribotoxin alpha-sarcin: induction of cell death via apoptosis. Eur. J. Biochem. 268, 2113–2123 (2001).
Martinez-Ruiz, A., Kao, R., Davies, J. & Martinez del Pozo, A. Ribotoxins are a more widespread group of proteins within the filamentous fungi than previously believed. Toxicon 37, 1549–1563 (1999).
Yang, X. & Moffat, K. Insights into specificity of cleavage and mechanism of cell entry from the crystal structure of the highly specific Aspergillus ribotoxin, restrictocin. Structure 4, 837–852 (1996).
Lacadena, J. et al. Role of histidine-50, glutamic acid-96, and histidine-137 in the ribonucleolytic mechanism of the ribotoxin alpha-sarcin. Proteins 37, 474–484 (1999).
Gluck, A. & Wool, I.G. Determination of the 28 S ribosomal RNA identity element (G4319) for alpha-sarcin and the relationship of recognition to the selection of the catalytic site. J. Mol. Biol. 256, 838–848 (1996).
Yang, X., Gerczei, T., Glover, L. & Correll, C.C. Crystal structures of restrictocin-inhibitor complexes with implications for RNA recognition and base flipping. Nat. Struct. Biol. 8, 968–973 (2001).
Northrup, S.H. & Erickson, H.P. Kinetics of protein-protein association explained by Brownian dynamics computer simulation. Proc. Natl. Acad. Sci. USA 89, 3338–3342 (1992).
Stroppolo, M.E. et al. Single mutation at the intersubunit interface confers extra efficiency to Cu, Zn superoxide dismutase. FEBS Lett. 483, 17–20 (2000).
Stroppolo, M.E., Falconi, M., Caccuri, A.M. & Desideri, A. Superefficient enzymes. Cell. Mol. Life Sci. 58, 1451–1460 (2001).
Gluck, A., Endo, Y. & Wool, I.G. The ribosomal RNA identity elements for ricin and for alpha-sarcin: mutations in the putative CG pair that closes a GAGA tetraloop. Nucleic Acids Res. 22, 321–324 (1994).
Macbeth, M.R. & Wool, I.G. Characterization of in vitro and in vivo mutations in non-conserved nucleotides in the ribosomal RNA recognition domain for the ribotoxins ricin and sarcin and the translation elongation factors. J. Mol. Biol. 285, 567–580 (1999).
Park, C. & Raines, R.T. Quantitative analysis of the effect of salt concentration on enzymatic catalysis. J. Am. Chem. Soc. 123, 11472–11479 (2001).
Day-Storms, J.J., Niranjanakumari, S. & Fierke, C.A. Ionic interactions between PRNA and P protein in Bacillus subtilis RNase P characterized using a magnetocapture-based assay. RNA 10, 1595–1608 (2004).
Hall, K.B. & Stump, W.T. Interaction of N-terminal domain of U1A protein with an RNA stem/loop. Nucleic Acids Res. 20, 4283–4290 (1992).
Saecker, R.M. & Record, M.T. Jr. Protein surface salt bridges and paths for DNA wrapping. Curr. Opin. Struct. Biol. 12, 311–319 (2002).
Record, M.T., Jr., Zhang, W. & Anderson, C.F. Analysis of effects of salts and uncharged solutes on protein and nucleic acid equilibria and processes: a practical guide to recognizing and interpreting polyelectrolyte effects, Hofmeister effects, and osmotic effects of salts. Adv. Protein Chem. 51, 281–353 (1998).
Record, M.T., Jr., deHaseth, P.L. & Lohman, T.M. Interpretation of monovalent and divalent cation effects on the lac repressor-operator interaction. Biochemistry 16, 4791–4796 (1977).
Chen, S.-w.W. & Honig, B. Monovalent and divalent salt effects on electrostatic free energies defined by the nonlinear poisson-Boltzmann equation: application to DNA binding reactions. J. Phys. Chem. B 101, 9113–9118 (1997).
Dong, H., Nilsson, L. & Kurland, C.G. Co-variation of tRNA abundance and codon usage in Escherichia coli at different growth rates. J. Mol. Biol. 260, 649–663 (1996).
Porse, B.T., Leviev, I., Mankin, A.S. & Garrett, R.A. The antibiotic thiostrepton inhibits a functional transition within protein L11 at the ribosomal GTPase centre. J. Mol. Biol. 276, 391–404 (1998).
Trylska, J., Konecny, R., Tama, F., Brooks, C.L., III & McCammon, J.A. Ribosome motions modulate electrostatic properties. Biopolymers 74, 423–431 (2004).
Baker, N.A., Sept, D., Joseph, S., Holst, M.J. & McCammon, J.A. Electrostatics of nanosystems: application to microtubules and the ribosome. Proc. Natl. Acad. Sci. USA 98, 10037–10041 (2001).
Moore, P.B. & Steitz, T.A. The structural basis of large ribosomal subunit function. Annu. Rev. Biochem. 72, 813–850 (2003).
Selzer, T. & Schreiber, G. Predicting the rate enhancement of protein complex formation from the electrostatic energy of interaction. J. Mol. Biol. 287, 409–419 (1999).
Selzer, T., Albeck, S. & Schreiber, G. Rational design of faster associating and tighter binding protein complexes. Nat. Struct. Biol. 7, 537–541 (2000).
Hemsath, L., Dvorsky, R., Fiegen, D., Carlier, M.F. & Ahmadian, M.R. An electrostatic steering mechanism of Cdc42 recognition by Wiskott-Aldrich syndrome proteins. Mol. Cell 20, 313–324 (2005).
von Hippel, P.H. & Berg, O.G. Facilitated target location in biological systems. J. Biol. Chem. 264, 675–678 (1989).
Futami, J. et al. Optimum modification for the highest cytotoxicity of cationized ribonuclease. J. Biochem. 132, 223–228 (2002).
Vives, E., Richard, J.P., Rispal, C. & Lebleu, B. TAT peptide internalization: seeking the mechanism of entry. Curr. Protein Pept. Sci. 4, 125–132 (2003).
Mathews, D.H., Sabina, J., Zuker, M. & Turner, D.H. Expanded sequence dependence of thermodynamic parameters improves prediction of RNA secondary structure. J. Mol. Biol. 288, 911–940 (1999).
Spedding, G. Isolation and analysis of ribosomes from prokaryotes, eukaryotes, and organelles. in Ribosomes and Protein Synthesis—A Practical Approach (ed. Speddings, G.) 1–29 (Oxford University Press, New York, 1990).
Endo, Y., Huber, P.W. & Wool, I.G. The ribonuclease activity of the cytotoxin alpha-sarcin. The characteristics of the enzymatic activity of alpha-sarcin with ribosomes and ribonucleic acids as substrates. J. Biol. Chem. 258, 2662–2667 (1983).
Demeler, B. UltraScan: a comprehensive data analysis software package for analytical ultracentrifugation experiments. in Modern Analytical Ultracentrifugation: Techniques and Methods (eds. Scott, D., Harding, S.E. & Rowe, A.J.) 210–229 (Royal Society of Chemistry, Cambridge, 2005).
Schuck, P. Size-distribution analysis of macromolecules by sedimentation velocity ultracentrifugation and lamm equation modeling. Biophys. J. 78, 1606–1619 (2000).
Mendes, P. & Kell, D. Non-linear optimization of biochemical pathways: applications to metabolic engineering and parameter estimation. Bioinformatics 14, 869–883 (1998).
Carson, M. Ribbons. Methods Enzymol. 277, 493–505 (1991).
Guex, N. & Peitsch, M.C. SWISS-MODEL and the Swiss-PdbViewer: an environment for comparative protein modeling. Electrophoresis 18, 2714–2723 (1997).
Ban, N., Nissen, P., Hansen, J., Moore, P.B. & Steitz, T.A. The complete atomic structure of the large ribosomal subunit at 2.4 Å resolution. Science 289, 905–920 (2000).
Klein, D.J., Schmeing, T.M., Moore, P.B. & Steitz, T.A. The kink-turn: a new RNA secondary structure motif. EMBO J. 20, 4214–4221 (2001).
Patel, V.B., Cunningham, C.C. & Hantgan, R.R. Physiochemical properties of rat liver mitochondrial ribosomes. J. Biol. Chem. 276, 6739–6746 (2001).
Dube, P. et al. The 80S rat liver ribosome at 25 Å resolution by electron cryomicroscopy and angular reconstitution. Structure 6, 389–399 (1998).
Acknowledgements
We are grateful to Y.-L. Chan for valuable advice and discussion, J. Olvera for excellent technical assistance and M. Yousef for the help at the Biophysical Core Facility at the University of Chicago. We thank I.G. Wool, P.B. Moore and members of the Piccirilli laboratory for helpful comments on the manuscript. This work was supported by grants from the Burroughs Wellcome Fund to A.V.K., from the Howard Hughes Medical Institute to J.A.P. and from the US National Institutes of Health (GM59782) to C.C.C.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Cleavage of the rat SRL by restrictocin (PDF 140 kb)
Supplementary Fig. 2
Inhibition of ribotoxin-mediated RNA cleavage by salt (PDF 132 kb)
Supplementary Fig. 3
Km for SRL cleavage by restrictocin equals the binding constant Kd (PDF 82 kb)
Supplementary Fig. 4
Cleavage of ribosomes and total rRNA by restrictocin (PDF 615 kb)
Supplementary Fig. 5
Kinetic characterization of the 3/D triple mutant (PDF 205 kb)
Supplementary Table 1
Effect of buffer composition on the rate of SRL cleavage by restrictocin (PDF 51 kb)
Supplementary Table 2
Sequences of the RNA and DNA oligonucleotides used (PDF 67 kb)
Rights and permissions
About this article
Cite this article
Korennykh, A., Piccirilli, J. & Correll, C. The electrostatic character of the ribosomal surface enables extraordinarily rapid target location by ribotoxins. Nat Struct Mol Biol 13, 436–443 (2006). https://doi.org/10.1038/nsmb1082
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb1082