Abstract
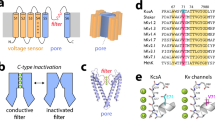
The prokaryotic K+ channel KcsA, although lacking a 'standard' voltage-sensing domain, shows voltage-dependent gating that leads to an increase in steady-state open probability of almost two orders of magnitude between +150 and −150 mV. Here we show that voltage-dependent gating in KcsA is associated with the movement of ∼0.7 equivalent electronic charges. This charge movement produces an increase in the rate of entry into a long-lived inactivated state and seems to be independent of the proton-activation mechanism. Charge neutralization at position 71 renders the channel essentially voltage-independent by preventing entry into the inactivated state. A mechanism for voltage-dependent gating at the selectivity filter is proposed that is based on the reorientation of the carboxylic moiety of Glu71 and its influence in the conformational dynamics of the selectivity filter.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Hille, B. Ion channels of excitable membranes (Sinauer, Sunderland, Massachusetts, 2001).
Armstrong, C.M. Voltage-gated K channels. Sci. STKE 2003, re10 (2003).
Bezanilla, F. The voltage sensor in voltage-dependent ion channels. Physiol. Rev. 80, 555–592 (2000).
Yellen, G. The voltage-gated potassium channels and their relatives. Nature 419, 35–42 (2002).
Murata, Y., Iwasaki, H., Sasaki, M., Inaba, K. & Okamura, Y. Phosphoinositide phosphatase activity coupled to an intrinsic voltage sensor. Nature 435, 1239–1243 (2005).
Hodgkin, A. & Huxley, A. A quantitative description of membrane current and its application to conduction and excitation in nerve. J. Physiol. (Lond.) 117, 500–544 (1952).
Aggarwal, S.K. & MacKinnon, R. Contribution of the S4 segment to gating charge in the Shaker K+ channel. Neuron 16, 1169–1177 (1996).
Seoh, S.A., Sigg, D., Papazian, D.M. & Bezanilla, F. Voltage-sensing residues in the S2 and S4 segments of the Shaker K+ channel. Neuron 16, 1159–1167 (1996).
Jiang, Y. et al. X-ray structure of a voltage-dependent K+ channel. Nature 423, 33–41 (2003).
Cuello, L.G., Cortes, D.M. & Perozo, E. Molecular architecture of the KvAP voltage-dependent K+ channel in a lipid bilayer. Science 306, 491–495 (2004).
Long, S.B., Campbell, E.B. & Mackinnon, R. Crystal structure of a mammalian voltage-dependent Shaker family K+ channel. Science 309, 897–903 (2005).
Long, S.B., Campbell, E.B. & Mackinnon, R. Voltage sensor of Kv1.2: structural basis of electromechanical coupling. Science 309, 903–908 (2005).
Bezanilla, F. The voltage-sensor structure in a voltage-gated channel. Trends Biochem. Sci. 30, 166–168 (2005).
Ahern, C.A. & Horn, R. Stirring up controversy with a voltage sensor paddle. Trends Neurosci. 27, 303–307 (2004).
Cohen, B.E., Grabe, M. & Jan, L.Y. Answers and questions from the KvAP structures. Neuron 39, 395–400 (2003).
Pusch, M., Ludewig, U., Rehfeldt, A. & Jentsch, T.J. Gating of the voltage-dependent chloride channel CIC-0 by the permeant anion. Nature 373, 527–531 (1995).
Doyle, D.A. et al. The structure of the potassium channel: molecular basis of K+ conduction and selectivity. Science 280, 69–77 (1998).
Cuello, L.G., Romero, J.G., Cortes, D.M. & Perozo, E. pH-dependent gating in the Streptomyces lividans K+ channel. Biochemistry 37, 3229–3236 (1998).
Heginbotham, L., LeMasurier, M., Kolmakova-Partensky, L. & Miller, C. Single Streptomyces lividans K+ channels: functional asymmetries and sidedness of proton activation. J. Gen. Physiol. 114, 551–560 (1999).
Schoppa, N.E., McCormack, K., Tanouye, M.A. & Sigworth, F.J. The size of gating charge in wild-type and mutant Shaker potassium channels. Science 255, 1712–1715 (1992).
Cordero-Morales, J.F. et al. Molecular determinants of gating at the potassium channel selectivity filter. Nat. Struct. Mol. Biol., advance online publication 12 March 2006 (doi:10.1038/nsmb1069).
McLaughlin, S. The electrostatic properties of membranes. Annu. Rev. Biophys. Biophys. Chem. 18, 113–136 (1989).
Cortes, D.M., Cuello, L.G. & Perozo, E. Molecular architecture of full-length KcsA: role of cytoplasmic domains in ion permeation and activation gating. J. Gen. Physiol. 117, 165–180 (2001).
Zhou, Y., Morais-Cabral, J.H., Kaufman, A. & MacKinnon, R. Chemistry of ion coordination and hydration revealed by a K+ channel-Fab complex at 2.0 A resolution. Nature 414, 43–48 (2001).
Berneche, S. & Roux, B. Molecular dynamics of the KcsA K(+) channel in a bilayer membrane. Biophys. J. 78, 2900–2917 (2000).
Berneche, S. & Roux, B. The ionization state and the conformation of Glu-71 in the KcsA K+ channel. Biophys. J. 82, 772–780 (2002).
Guidoni, L., Torre, V. & Carloni, P. Potassium and sodium binding to the outer mouth of the K+ channel. Biochemistry 38, 8599–8604 (1999).
Ranatunga, K.M., Shrivastava, I.H., Smith, G.R. & Sansom, M.S. Side-chain ionization states in a potassium channel. Biophys. J. 80, 1210–1219 (2001).
Choi, H. & Heginbotham, L. Functional influence of the pore helix glutamate in the KcsA K+ channel. Biophys. J. 86, 2137–2144 (2004).
Cortes, D.M. & Perozo, E. Structural dynamics of the Streptomyces lividans K+ channel (SKC1): oligomeric stoichiometry and stability. Biochemistry 36, 10343–10352 (1997).
Perozo, E., Cortes, D.M. & Cuello, L.G. Three-dimensional architecture and gating mechanism of a K+ channel studied by EPR spectroscopy. Nat. Struct. Biol. 5, 459–469 (1998).
Delcour, A.H., Martinac, B., Adler, J. & Kung, C. Modified reconstitution method used in patch-clamp studies of Escherichia coli ion channels. Biophys. J. 56, 631–636 (1989).
Acknowledgements
We thank B. Roux for illuminating discussions and for critically reading the manuscript and S. Chakrapani, C. Ptak, D.M. Cortes and V. Vasquez for useful discussions and advice. This work was supported by grants from the US National Institutes of Health to E.P.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Cordero-Morales, J., Cuello, L. & Perozo, E. Voltage-dependent gating at the KcsA selectivity filter. Nat Struct Mol Biol 13, 319–322 (2006). https://doi.org/10.1038/nsmb1070
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb1070