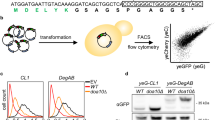
Abstract
The proteasome controls the concentrations of most proteins in eukaryotic cells. It recognizes its protein substrates through ubiquitin tags and initiates degradation at disordered regions within the substrate. Here we show that the proteasome has pronounced preferences for the amino acid sequence of the regions at which it initiates degradation. Specifically, proteins in which the initiation regions have biased amino acid compositions show longer half-lives in yeast than proteins with unbiased sequences in the regions. The relationship is also observed on a genomic scale in mouse cells. These preferences affect the degradation rates of proteins in vitro, can explain the unexpected stability of natural proteins in yeast and may affect the accumulation of toxic proteins in disease. We propose that the proteasome's sequence preferences provide a second component to the degradation code and may fine-tune protein half-life in cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Schrader, E.K., Harstad, K.G. & Matouschek, A. Targeting proteins for degradation. Nat. Chem. Biol. 5, 815–822 (2009).
Komander, D. & Rape, M. The ubiquitin code. Annu. Rev. Biochem. 81, 203–229 (2012).
Prakash, S., Tian, L., Ratliff, K.S., Lehotzky, R.E. & Matouschek, A. An unstructured initiation site is required for efficient proteasome-mediated degradation. Nat. Struct. Mol. Biol. 11, 830–837 (2004).
Lee, C., Schwartz, M.P., Prakash, S., Iwakura, M. & Matouschek, A. ATP-dependent proteases degrade their substrates by processively unraveling them from the degradation signal. Mol. Cell 7, 627–637 (2001).
Nathan, J.A., Kim, H.T., Ting, L., Gygi, S.P. & Goldberg, A.L. Why do cellular proteins linked to K63-polyubiquitin chains not associate with proteasomes? EMBO J. 32, 552–565 (2013).
Fishbain, S., Prakash, S., Herrig, A., Elsasser, S. & Matouschek, A. Rad23 escapes degradation because it lacks a proteasome initiation region. Nat. Commun. 2, 192 (2011).
Inobe, T., Fishbain, S., Prakash, S. & Matouschek, A. Defining the geometry of the two-component proteasome degron. Nat. Chem. Biol. 7, 161–167 (2011).
Schauber, C. et al. Rad23 links DNA repair to the ubiquitin/proteasome pathway. Nature 391, 715–718 (1998).
Watkins, J.F., Sung, P., Prakash, L. & Prakash, S. The Saccharomyces cerevisiae DNA repair gene RAD23 encodes a nuclear protein containing a ubiquitin-like domain required for biological function. Mol. Cell. Biol. 13, 7757–7765 (1993).
Heessen, S., Masucci, M.G. & Dantuma, N.P. The UBA2 domain functions as an intrinsic stabilization signal that protects Rad23 from proteasomal degradation. Mol. Cell 18, 225–235 (2005).
Heinen, C., Acs, K., Hoogstraten, D. & Dantuma, N.P. C-terminal UBA domains protect ubiquitin receptors by preventing initiation of protein degradation. Nat. Commun. 2, 191 (2011).
Punta, M. et al. The Pfam protein families database. Nucleic Acids Res. 40, D290–D301 (2012).
Stack, J.H., Whitney, M., Rodems, S.M. & Pollok, B.A. A ubiquitin-based tagging system for controlled modulation of protein stability. Nat. Biotechnol. 18, 1298–1302 (2000).
Goebl, M.G., Goetsch, L. & Byers, B. The Ubc3 (Cdc34) ubiquitin-conjugating enzyme is ubiquitinated and phosphorylated in vivo. Mol. Cell. Biol. 14, 3022–3029 (1994).
Banerjee, A., Gregori, L., Xu, Y. & Chau, V. The bacterially expressed yeast CDC34 gene product can undergo autoubiquitination to form a multiubiquitin chain-linked protein. J. Biol. Chem. 268, 5668–5675 (1993).
Chau, V. et al. A multiubiquitin chain is confined to specific lysine in a targeted short-lived protein. Science 243, 1576–1583 (1989).
Bachmair, A. & Varshavsky, A. The degradation signal in a short-lived protein. Cell 56, 1019–1032 (1989).
Wootton, J.C. Non-globular domains in protein sequences: automated segmentation using complexity measures. Comput. Chem. 18, 269–285 (1994).
Wootton, J.C. & Federhen, S. Analysis of compositionally biased regions in sequence databases. Methods Enzymol. 266, 554–571 (1996).
Daskalogianni, C. et al. Gly-Ala repeats induce position- and substrate-specific regulation of 26 S proteasome-dependent partial processing. J. Biol. Chem. 283, 30090–30100 (2008).
Hoyt, M.A. et al. Glycine-alanine repeats impair proper substrate unfolding by the proteasome. EMBO J. 25, 1720–1729 (2006).
Sharipo, A., Imreh, M., Leonchiks, A., Imreh, S. & Masucci, M.G. A minimal glycine-alanine repeat prevents the interaction of ubiquitinated IκBα with the proteasome: a new mechanism for selective inhibition of proteolysis. Nat. Med. 4, 939–944 (1998).
Zhang, M. & Coffino, P. Repeat sequence of Epstein-Barr virus-encoded nuclear antigen 1 protein interrupts proteasome substrate processing. J. Biol. Chem. 279, 8635–8641 (2004).
Tian, L., Holmgren, R.A. & Matouschek, A. A conserved processing mechanism regulates the activity of transcription factors Cubitus interruptus and NF-κB. Nat. Struct. Mol. Biol. 12, 1045–1053 (2005).
Pratt, G. & Rechsteiner, M. Proteasomes cleave at multiple sites within polyglutamine tracts: activation by PA28γ(K188E). J. Biol. Chem. 283, 12919–12925 (2008).
Juenemann, K. et al. Expanded polyglutamine-containing N-terminal huntingtin fragments are entirely degraded by mammalian proteasomes. J. Biol. Chem. 288, 27068–27084 (2013).
Venkatraman, P., Wetzel, R., Tanaka, M., Nukina, N. & Goldberg, A.L. Eukaryotic proteasomes cannot digest polyglutamine sequences and release them during degradation of polyglutamine-containing proteins. Mol. Cell 14, 95–104 (2004).
Akopian, T.N., Kisselev, A.F. & Goldberg, A.L. Processive degradation of proteins and other catalytic properties of the proteasome from Thermoplasma acidophilum. J. Biol. Chem. 272, 1791–1798 (1997).
Udenfriend, S. et al. Fluorescamine: a reagent for assay of amino acids, peptides, proteins, and primary amines in the picomole range. Science 178, 871–872 (1972).
Romero, P. et al. Sequence complexity of disordered protein. Proteins 42, 38–48 (2001).
Tompa, P. & Fersht, A.R. Structure and Function of Intrinsically Disordered Proteins (CRC Press, 2010).
van der Lee, R. et al. Intrinsically disordered segments affect protein half-life in the cell and during evolution. Cell Rep. 8, 1832–1844 (2014).
Schwanhäusser, B. et al. Global quantification of mammalian gene expression control. Nature 473, 337–342 (2011).
Zuccato, C., Valenza, M. & Cattaneo, E. Molecular mechanisms and potential therapeutical targets in Huntington's disease. Physiol. Rev. 90, 905–981 (2010).
DiFiglia, M., Sapp, E., Chase, K.O., Davies, S.W. & Bates, G.P. Aggregation of huntingtin in neuronal intranuclear inclusions and dystrophic neurites in brain. Science 277, 1990–1993 (1997).
Kalchman, M.A. et al. Huntingtin is ubiquitinated and interacts with a specific ubiquitin-conjugating enzyme. J. Biol. Chem. 271, 19385–19394 (1996).
Douglas, P.M. & Dillin, A. Protein homeostasis and aging in neurodegeneration. J. Cell Biol. 190, 719–729 (2010).
Jana, N.R., Zemskov, E.A., Gh, W. & Nukina, N. Altered proteasomal function due to the expression of polyglutamine-expanded truncated N-terminal huntingtin induces apoptosis by caspase activation through mitochondrial cytochrome c release. Hum. Mol. Genet. 10, 1049–1059 (2001).
Waelter, S. et al. Accumulation of mutant huntingtin fragments in aggresome-like inclusion bodies as a result of insufficient protein degradation. Mol. Biol. Cell 12, 1393–1407 (2001).
Wyttenbach, A. et al. Effects of heat shock, heat shock protein 40 (HDJ-2), and proteasome inhibition on protein aggregation in cellular models of Huntington's disease. Proc. Natl. Acad. Sci. USA 97, 2898–2903 (2000).
Fiumara, F., Fioriti, L., Kandel, E.R. & Hendrickson, W.A. Essential role of coiled coils for aggregation and activity of Q/N-rich prions and polyQ proteins. Cell 143, 1121–1135 (2010).
Young, L.S. & Rickinson, A.B. Epstein-Barr virus: 40 years on. Nat. Rev. Cancer 4, 757–768 (2004).
Levitskaya, J. et al. Inhibition of antigen processing by the internal repeat region of the Epstein-Barr virus nuclear antigen-1. Nature 375, 685–688 (1995).
Kraut, D.A. & Matouschek, A. Proteasomal degradation from internal sites favors partial proteolysis via remote domain stabilization. ACS Chem. Biol. 6, 1087–1095 (2011).
Kraut, D.A. et al. Sequence- and species-dependence of proteasomal processivity. ACS Chem. Biol. 7, 1444–1453 (2012).
Babu, M.M., Kriwacki, R.W. & Pappu, R.V. Structural biology. Versatility from protein disorder. Science 337, 1460–1461 (2012).
Mao, A.H., Crick, S.L., Vitalis, A., Chicoine, C.L. & Pappu, R.V. Net charge per residue modulates conformational ensembles of intrinsically disordered proteins. Proc. Natl. Acad. Sci. USA 107, 8183–8188 (2010).
Das, R.K. & Pappu, R.V. Conformations of intrinsically disordered proteins are influenced by linear sequence distributions of oppositely charged residues. Proc. Natl. Acad. Sci. USA 110, 13392–13397 (2013).
Müller-Späth, S. et al. Charge interactions can dominate the dimensions of intrinsically disordered proteins. Proc. Natl. Acad. Sci. USA 107, 14609–14614 (2010).
Moesa, H.A., Wakabayashi, S., Nakai, K. & Patil, A. Chemical composition is maintained in poorly conserved intrinsically disordered regions and suggests a means for their classification. Mol. Biosyst. 8, 3262–3273 (2012).
Brown, C.J., Johnson, A.K., Dunker, A.K. & Daughdrill, G.W. Evolution and disorder. Curr. Opin. Struct. Biol. 21, 441–446 (2011).
Hagai, T. & Levy, Y. Ubiquitin not only serves as a tag but also assists degradation by inducing protein unfolding. Proc. Natl. Acad. Sci. USA 107, 2001–2006 (2010).
Beskow, A. et al. A conserved unfoldase activity for the p97 AAA-ATPase in proteasomal degradation. J. Mol. Biol. 394, 732–746 (2009).
Raman, M., Haven, C.G., Walter, J.C. & Harper, J.W. A genome-wide screen identifies p97 as an essential regulator of DNA damage-dependent CDT1 destruction. Mol. Cell 44, 72–82 (2011).
Zoghbi, H.Y. & Orr, H.T. Glutamine repeats and neurodegeneration. Annu. Rev. Neurosci. 23, 217–247 (2000).
Bancher, C. et al. An antigenic profile of Lewy bodies: immunocytochemical indication for protein phosphorylation and ubiquitination. J. Neuropathol. Exp. Neurol. 48, 81–93 (1989).
Iqbal, K. & Grundke-Iqbal, I. Ubiquitination and abnormal phosphorylation of paired helical filaments in Alzheimer's disease. Mol. Neurobiol. 5, 399–410 (1991).
Bence, N.F., Sampat, R.M. & Kopito, R.R. Impairment of the ubiquitin-proteasome system by protein aggregation. Science 292, 1552–1555 (2001).
Hipp, M.S. et al. Indirect inhibition of 26S proteasome activity in a cellular model of Huntington's disease. J. Cell Biol. 196, 573–587 (2012).
Holmberg, C.I., Staniszewski, K.E., Mensah, K.N., Matouschek, A. & Morimoto, R.I. Inefficient degradation of truncated polyglutamine proteins by the proteasome. EMBO J. 23, 4307–4318 (2004).
Bennett, E.J., Bence, N.F., Jayakumar, R. & Kopito, R.R. Global impairment of the ubiquitin-proteasome system by nuclear or cytoplasmic protein aggregates precedes inclusion body formation. Mol. Cell 17, 351–365 (2005).
Kruegel, U. et al. Elevated proteasome capacity extends replicative lifespan in Saccharomyces cerevisiae. PLoS Genet. 7, e1002253 (2011).
Lee, B.H. et al. Enhancement of proteasome activity by a small-molecule inhibitor of USP14. Nature 467, 179–184 (2010).
Ward, J.J., Sodhi, J.S., McGuffin, L.J., Buxton, B.F. & Jones, D.T. Prediction and functional analysis of native disorder in proteins from the three kingdoms of life. J. Mol. Biol. 337, 635–645 (2004).
Muñoz, V. & Serrano, L. Development of the multiple sequence approximation within the AGADIR model of α-helix formation: comparison with Zimm-Bragg and Lifson-Roig formalisms. Biopolymers 41, 495–509 (1997).
Saeki, Y., Isono, E. & Toh-e, A. Preparation of ubiquitinated substrates by the PY motif-insertion method for monitoring 26S proteasome activity. Methods Enzymol. 399, 215–227 (2005).
Lander, G.C. et al. Complete subunit architecture of the proteasome regulatory particle. Nature 482, 186–191 (2012).
Evans, C.H. & Ridella, J.D. An evaluation of fluorometric proteinase assays which employ fluorescamine. Anal. Biochem. 142, 411–420 (1984).
Dosztányi, Z., Csizmók, V., Tompa, P. & Simon, I. The pairwise energy content estimated from amino acid composition discriminates between folded and intrinsically unstructured proteins. J. Mol. Biol. 347, 827–839 (2005).
Obradovic, Z., Peng, K., Vucetic, S., Radivojac, P. & Dunker, A.K. Exploiting heterogeneous sequence properties improves prediction of protein disorder. Proteins 61 (suppl. 7), 176–182 (2005).
Harbi, D., Kumar, M. & Harrison, P.M. LPS-annotate: complete annotation of compositionally biased regions in the protein knowledgebase. Database (Oxford) 2011, baq031 (2011).
Harrison, P.M. & Gerstein, M. A method to assess compositional bias in biological sequences and its application to prion-like glutamine/asparagine-rich domains in eukaryotic proteomes. Genome Biol. 4, R40 (2003).
Acknowledgements
We thank J. Brickner and R.A. Lamb (Northwestern University), M. Glickman (Technion), R.V. Pappu (Washington University) and Y. Saeki (Tokyo Metropolitan Institute of Medical Science) for advice and reagents, as well as A. Zokarkar for early experiments on this project. We are also grateful to K. Brown, A. Gnanam and E.J. Cho for help with the CD experiments. This work was supported by the US National Institutes of Health (R01GM063004, U54GM105816 and U54CA143869 (A.M.)); the Welch Foundation (F-1817 (A.M.)); the Program to Disseminate Tenure Track System from the Ministry of Education, Culture, Sports, Science and Technology, Japan (T.I.); the UK Medical Research Council (MC_U105185859 (S.C. and M.M.B.); and the European Molecular Biology Organization (Long-Term Fellowship (S.C.) and Young Investigator Program (M.M.B.)).
Author information
Authors and Affiliations
Contributions
S.F., T.I., E.I., S.C., H.Y. and G.K. performed experiments, analyzed data and co-wrote the paper. M.M.B. and A.M. directed experiments, analyzed data and co-wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Proteasome degradation of Cdc34.
Cdc34 with a 95 amino acid initiation region at its C terminus was synthesized by coupled in vitro transcription and translation, ubiquitinated and then presented to purified yeast proteasome. Cdc34-95 was degraded by the proteasome in the presence of ATP (blue triangles; +ATP) but degradation was inhibited with the depletion of ATP by hexokinase treatment (red diamonds; -ATP) or the addition of 100 µM MG-132 (black diamonds; +MG-132). Data points represent mean values of three repeat experiments.
Supplementary Figure 2 Ubiquitination of Cdc34 in vivo.
Cdc34 becomes ubiquitinated when overexpressed in yeast yet remains stable (Goebl, M.G., Goetsch, L. & Byers, B. The Ubc3 (Cdc34) ubiquitin-conjugating enzyme is ubiquitinated and phosphorylated in vivo. Mol. Cell Biol. 14, 3022-3029 (1994)). Attaching a 95 amino acid long tail leads to degradation of Cdc34-95 but does not increase detectable ubiquitination of Cdc34. Ubiquitination tags are labile because of the large number of deubiquitinating enzymes present in cells. To preserve ubiquitination patterns as far as possible, we followed protocols developed to preserve ubiquitiations by the Walter lab (Cox, J. S., Chapman, R. E. & Walter, P. The unfolded protein response coordinates the production of endoplasmic reticulum protein and endoplasmic reticulum membrane. Mol. Biol. Cell 8, 1805–1814 (1997)) and modified by the Dohlman lab (http://www.med.unc.edu/~hdohlman/TCA.html). Yeast strains expressing N-terminally HA tagged Cdc34 or Cdc34-95 were grown to mid log phase, cultures were diluted to OD600nm 0.2, and then grown for four more hours in the presence or absence of the proteasome inhibitor MG132. At that stage, cells were rapidly frozen in liquid nitrogen and then lysed with glass beads in the presence of >5% trichloroacetic acid (TCA). Cell lysate was washed in TCA buffer and the TCA pellet dissolved in buffered SDS PAGE loading buffer. Cell lysates were separated by SDS PAGE, and analyzed by Western blotting. Cdc34 and Cdc-95 were then visualized with antibodies directed against their HA tag, and Scs2 was used as a loading control. Probing for the HA-tag on Cdc34 and Cdc34-95 reveals the characteristic pattern of polyubiquitin modification. The polyubiquitin pattern is more pronounced for the Cdc34 lacking the unstructured tail (Cdc34) than for the Cdc34 variant with the unstructured tail (Cdc34-95) as expected if the ubiquitinated Cdc34-95 is rapidly degraded. Inhibiting the proteasome with MG132 allows the accumulation of some of the ubiquitinated Cdc34, however because proteasome inhibition in vivo is incomplete, only small amounts of ubiquitinated Cdc34-95 can be detected.
Supplementary Figure 3 Proteasome degradation reactions for model proteins with different initiation regions.
Degradation kinetics of substrates with different initiation regions. The substrates contained an E. coli DHFR domain and were targeted to the proteasome by an N-terminal Ub4 tag. Unstructured tails were placed at the C-terminus of DHFR to serve as initiation regions. Proteins were synthesized by coupled in vitro transcription and translation, partially purified and presented to purified yeast proteasome in the presence of an ATP regenerating system at 30 ˚C. The graph shows a plot of the amount of protein remaining at the given reaction times. Degradation kinetics of substrates with the tails 35ΔK (red solid circles; n=6), NS2 (pink solid squares; n=4), NB (blue solid diamonds; n=4), SP2 (emerald solid triangles, n=3), SP1 (light blue solid inverted triangles; n=3), SRR (yellow open circles; n=3), ODC (red open diamonds; n=5), eRR (magenta open triangles; n=3), Subunit9 (purple open inverted triangles; n=3), SNS (purple crosses; n=4), SPmix (blue pluses; n=4), PEST (light blue close circles; n=3), RPB (green close squares; n=3), DRR (light green close diamonds; n=3), and GRR (yellow open squares; n=3). The amino acid sequences of the tails are shown in Supplementary Table 2. Data points represent mean values, and error bars show standard errors calculated from three to six repeat experiments as indicated above. The solid lines are fits of single exponential decay curve to data.
Supplementary Figure 4 Structural analysis of protein tails by protease sensitivity and CD.
(a) We constructed expression vectors to produce versions of 11 of the 15 proteins tested in Figure 5 without the tetra-ubiquitin tag. The proteins thus consisted of a central DHFR domain with an N-terminal deca-histidine tag for purification and C-terminal tails. We synthesized the proteins by coupled in vitro transcription and translation in E. coli lysate in the presence of 35S-methionine and analyzed them by SDS PAGE and electronic autoradiography (InstantImager, Packard) before and after treatment with the nonspecific protease proteinase K at 0.1 mg/ml (50 mM Tris, 5 mg MgCl2, 5% glycerol, pH 7.4) for 10 minutes on ice, largely as described previously (Prakash, S., Tian, L., Ratliff, K. S., Lehotzky, R. E., & Matouschek, A. (2004). An unstructured initiation site is required for efficient proteasome-mediated degradation. Nat. Struct. & Molec. Biol. 11, 830–837). Before protease treatment, the proteins run approximately according their calculated molecular weight keeping in mind that the amino acid composition of the tails affects SDS binding and thus mobility on SDS PAGE. Proteinase K treatment lead to collapse of all proteins to the same band representing DHFR without tail. The intensity of the collapsed band corresponded to approximately 90% of the protein present before protease treatment correcting for the loss of the tail. This result suggests that the tails were highly sensitive to proteinase K, presumably because they were disordered, whereas the DHFR domain resisted proteinase K treatment, presumably because it was folded into a compact structure. (b – g) We also investigated the structures of a subset of tails (12 out of 15) by circular dichroism spectroscopy. To do so, we expressed proteins consisting of a DHFR domain with an N-terminal deca-histidine tag and C-terminal tails in E. coli and purified them by affinity chromatography and gel filtration. Circular dichroism spectra were recorded at 0.2mg/ml at 25 ˚C on a Jasco J-815 circular dichroism spectrometer. The mean residue ellipticity (b) and molar ellipticity (c) were calculated based on CD signal, protein sample concentration and protein length (number of amino acids) for DHFR (black solid squares) and DHFR with C-terminal sequences: 35ΔK (red solid circles), NS2 (pink solid squares), NB (blue solid diamonds), SP2 (emerald solid triangles), SP1 (light blue solid inverted triangles), ODC (red open diamonds), eRR (magenta open triangles), SNS (purple crosses), SPmix (blue pluses), RPB (green solid squares), DRR (light green solid diamonds; n=3), and GRR (yellow open squares). Spectra plotting molar ellipticity (c) show the same overall shape with some small variations suggesting that different tails show small amounts of secondary structure. However, importantly, the variations in the spectra do not relate to the ability of the tails to support degradation of DHFR model substrates by the proteasome. Neither mean residue ellipticity at 222 nm (d) nor molar ellipticity at 222 nm (e) show any correlation with the initial degradation rate of Ub4-DHFR-tail substrates (f, g; initial degradation rates taken from Fig. 5). Thus, we did not detect any correlation between the secondary structure of the initiation regions and the initial degradation rates of substrates.
Supplementary Figure 5 Proteasome degradation rates are not dominated by proteolytic site specificity.
The unstructured tails analyzed as proteasome initiation sites were synthesized as peptides and presented to purified 26S proteasome. Peptide proteolysis was followed by the fluorescamine method. (a) Proteolytic kinetics of 35ΔK (red solid circles), NS2 (pink solid squares), NB (blue solid diamonds), SP2 (emerald solid triangles), SP1 (light blue solid inverted triangles), SRR (yellow open circles), DRR (light green close diamonds), SP231 (light blue open triangles), NRR (green open inverted triangles), and GRR (yellow open squares). Initial rate of proteolysis of different peptides was plotted according to the total charge (b), net charge (c), hydrophobicity (d), helix propensity (e), volume (f), and amino acid sequence complexity (g) of the peptide sequence. Data points represent mean values and error bars show standard errors calculated from three repeat experiments. The solid lines are linear fits to proteolysis data.
Supplementary Figure 6 Distribution of ubiquitination sites among the classes of proteins.
Ubiquitination sites for mouse proteins in five different tissues was obtained from Wagner, S.A., Beli, P., Weinert, B.T., Nielsen, M.L., Cox, J., Mann, M., and Choudhary, C. (2011). A proteome-wide, quantitative survey of in vivo ubiquitylation sites reveals widespread regulatory roles. Mol. Cell. Proteomics 10, M111 013284. Since the number of data points were limited, we classified the proteins into tertiles representing proteins that contained low, medium and high number of ubiquitination sites (a). For both proteins with N-terminal disorder (b) and proteins with C-terminal disorder (c), we tested if non-disordered proteins, proteins with N- or C-terminal disorder without amino acid bias and proteins with N- or C-terminal disorder with amino acid bias had differences in the number of proteins in each class of number of ubiquitination sites. Statistical significance was estimated using Fisher’s test.
Supplementary Figure 7 Size-exclusion chromatography of Htt exon 1 constructs.
Htt proteins elute at the expected monomer molecular weight. The experiment was repeated three times and one representative result is shown. Radioactive ubiquitin-tagged Htt proteins were resolved on Sephacryl S-100 high-resolution matrix (Amersham) in 10ml column. Approximately 0.15 ml fractions were collected and 35 µl of each were analyzed by 7.5% SDS PAGE and proteins were quantified by electronic autoradiography (Instant Imager, Packard). Elution profiles of Ub4-Htt exon1(Q52) -/+ 95tail (a) and Ub4-DHFR-Q52 -/+ 95tail (b) as detected by SDS PAGE. Fraction numbers and values of elution peak volumes are indicated. A Sephacryl-100 size exclusion chromatography column was equilibrated with buffer (0.05 M sodium phosphate, 0.15 M NaCl) and calibrated with the following protein standards: bovine serum albumin (66 kDa), ovalbumin (44 kDa), Myoglobin (17 kDa) and Vitamin B12 (1.35 kDa). The elution pattern of the protein size markers was linear on a semilog plot (c). Elution data are represented as log molecular weight to Kav. Kav was calculated as in the equation (Ve-V0)/(Vt-V0). Ve, Elution volume; Vo, Void volume (determined by the elution of Thyroglobulin, 670 kDa); Vt, total column volume. (d) Calculated and measured molecular weight values of Ub4-tagged Htt proteins. Radioactive ubiquitin-tagged Htt constructs were expressed in 50 µl reactions as described above. Following high-speed centrifugation the samples were diluted in 400 µl buffer SEC (0.05 M sodium phosphate, 0.15 M NaCl) and then loaded on a Sephacryl S-100 high-resolution matrix (Amersham) in a 10 ml column. Approximately 0.15 ml fractions were collected and 35 μl of each were analyzed by 7.5% SDS PAGE and quantification by electronic autoradiography (Instant Imager, Packard).
Supplementary Figure 8 Proteasome preparation.
Proteasome was purified from yeast by affinity chromatography as described in the methods section. Proteasome preparations were analyzed by SDS PAGE, stained with Coomassie (InstantBlue, Expedeon) and compared to published proteasome compositions (Saeki, Y., Isono, E. and Toh-E, A. Preparation of ubiquitinated substrates by the PY motif-insertion method for monitoring 26S proteasome activity. Meth. Enzymol. 399, 215–227 (2005)).
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8 and Supplementary Tables 1–3. (PDF 2314 kb)
Supplementary Data Set 1
Supplementary Dataset for Figures 1b, 2b, 3a, 4, 5, and 7 (PDF 3852 kb)
Rights and permissions
About this article
Cite this article
Fishbain, S., Inobe, T., Israeli, E. et al. Sequence composition of disordered regions fine-tunes protein half-life. Nat Struct Mol Biol 22, 214–221 (2015). https://doi.org/10.1038/nsmb.2958
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.2958
This article is cited by
-
Alleviating the unwanted effects of oxidative stress on Aβ clearance: a review of related concepts and strategies for the development of computational modelling
Translational Neurodegeneration (2023)
-
Orphan quality control by an SCF ubiquitin ligase directed to pervasive C-degrons
Nature Communications (2023)
-
Systematic prediction of degrons and E3 ubiquitin ligase binding via deep learning
BMC Biology (2022)
-
Intracellular arginine-dependent translation sensor reveals the dynamics of arginine starvation response and resistance in ASS1-negative cells
Cancer & Metabolism (2021)
-
Flexibility of intrinsically disordered degrons in AUX/IAA proteins reinforces auxin co-receptor assemblies
Nature Communications (2020)