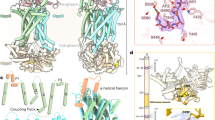
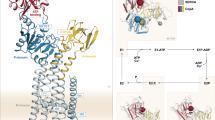
Abstract
The reaction mechanism of BtuCD–F–catalyzed vitamin B12 transport into Escherichia coli is currently unclear. Here we present the structure of the last missing state in the form of AMP-PNP–bound BtuCD, trapped by a disulfide cross-link. Our structural and biochemical data allow a consistent mechanism to be formulated, thus rationalizing the roles of substrate, ATP and substrate-binding protein.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Rees, D.C., Johnson, E. & Lewinson, O. Nat. Rev. Mol. Cell Biol. 10, 218–227 (2009).
Klein, J.S. & Lewinson, O. Metallomics 3, 1098–1108 (2011).
Locher, K.P., Lee, A.T. & Rees, D.C. Science 296, 1091–1098 (2002).
Korkhov, V.M., Mireku, S.A. & Locher, K.P. Nature 490, 367–372 (2012).
Joseph, B., Korkhov, V.M., Yulikov, M., Jeschke, G. & Bordignon, E. J. Biol. Chem. 289, 3176–3185 (2014).
Joseph, B., Jeschke, G., Goetz, B.A., Locher, K.P. & Bordignon, E. J. Biol. Chem. 286, 41008–41017 (2011).
Pinkett, H.W., Lee, A.T., Lum, P., Locher, K.P. & Rees, D.C. Science 315, 373–377 (2007).
Hvorup, R.N. et al. Science 317, 1387–1390 (2007).
Korkhov, V.M., Mireku, S.A., Hvorup, R.N. & Locher, K.P. FEBS Lett. 586, 972–976 (2012).
Woo, J.S., Zeltina, A., Goetz, B.A. & Locher, K.P. Nat. Struct. Mol. Biol. 19, 1310–1315 (2012).
Borths, E.L., Poolman, B., Hvorup, R.N., Locher, K.P. & Rees, D.C. Biochemistry 44, 16301–16309 (2005).
Patzlaff, J.S., van der Heide, T. & Poolman, B. J. Biol. Chem. 278, 29546–29551 (2003).
Chen, J. Curr. Opin. Struct. Biol. 23, 492–498 (2013).
Gould, A.D., Telmer, P.G. & Shilton, B.H. Biochemistry 48, 8051–8061 (2009).
Ritchie, T.K. et al. Methods Enzymol. 464, 211–231 (2009).
Brandt, T., Petrovich, M., Joerger, A.C. & Veprintsev, D.B. BMC Genomics 10, 628 (2009).
Schuck, P., Perugini, M.A., Gonzales, N.R., Howlett, G.J. & Schubert, D. Biophys. J. 82, 1096–1111 (2002).
Otwinowski, Z. & Minor, W. Methods Enzymol. 276, 307–326 (1997).
Strong, M. et al. Proc. Natl. Acad. Sci. USA 103, 8060–8065 (2006).
Emsley, P., Lohkamp, B., Scott, W.G. & Cowtan, K. Acta Crystallogr. D Biol. Crystallogr. 66, 486–501 (2010).
Adams, P.D. et al. Acta Crystallogr. D Biol. Crystallogr. 66, 213–221 (2010).
Acknowledgements
This work was supported by the Swiss National Science Foundation grants SNF 31003A-116191 and SNF 31003A-146191 to K.P.L. We gratefully acknowledge the assistance of staff scientists at the PX beamline of the Swiss Light Source, Villigen, Switzerland.
Author information
Authors and Affiliations
Contributions
V.M.K. and K.P.L. designed experiments, analyzed data and wrote the manuscript. V.M.K. and S.A.M. performed experiments, D.B.V. contributed to performing experiments, and S.A.M. contributed to data analysis and manuscript writing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Features of the BtuCDEQNC electron density map.
The density map (2Fo-Fc) of BtuCDEQNC, contoured at 1.5 σ, allows to discern key features of the nucleotide-bound complex structure, including: (a) Loops between helices 5 and 5a of adjacent BtuC monomers, forming the periplasmic gate (labeled as “peri”), shown as a stereo image, (b) residues N83-L85 of BtuC, forming the closed cytoplasmic gate II, (c) loops connecting the TM4 and TM5, forming the open cytoplasmic gate I, (d) well-resolved molecules of AMPPNP (orange) and Mg2+ (pink) bound at the interface of two BtuD subunits, (e) disulphide bridge between two C162 residues (yellow) stabilizing the nucleotide-bound closed sandwich dimer of BtuCDEQNC (stereo image). Protein chains are colored red and shown as cartoon (side chains shown as lines, with the exception of b and e, where side chains of residues of interest are shown as sticks).
Supplementary Figure 2 Labeling of BtuF Q145C and formation of BtuCD–Ffluo complex.
(a) Position of BtuF residue (Q145) chosen for labeling with fluorescein-maleimide. (b) BtuFQ145C was labeled with fluorescein-maleimide overnight at 4°C prior to preparative size-exclusion chromatography step using a Superdex 200 10/300 GL column, yielding the final BtuFfluo preparation. Stocks of BtuFfluo were split into aliquots, flash-frozen in liquid nitrogen and stored at -80 °C until the day of experiment. Coomassie-blue stained and SDS PAGE gel confirms purity of BtuFfluo preparation; the same gel prior to staining was subjected to imaging using a Typhoon fluorescence scanner, confirming association of fluorescein with BtuF. (c-e) BtuFfluo is able to form a stable complex with BtuCD in detergent (LDAO); complex formation was performed as described in “Online Methods”. The fluorescence peak of BtuFfluo (shown in d) shifts entirely to the left in the presence of 2-fold molar excess of BtuCD (shown in e), corresponding to the elution volume of the full BtuCD-F complex (for comparison, SEC profile of BtuCD alone is shown in c).
Supplementary Figure 3 Biophysical characterization of BtuFfluo binding to BtuCD or BtuCDEQNC in detergent.
(a-b) Schematic illustrating BtuCD (in b - BtuCDEQNC) in detergent micelles (orange cones) with BtuFfluo in the presence or absence of AMPPNP, as used in the experiments in c-f. The yellow line connecting the two BtuD subunits in BtuCDEQNC represents the disulfide cross-link. (c-d) Microscale thermophoresis data of BtuFfluo binding to BtuCD (c) or BtuCDEQNC (d). Protein samples of interest (a range of concentrations of BtuCD in LDAO and BtuFfluo at 25 nM concentration) were mixed in the presence or absence of 1 mM AMPPNP, incubated at 10 °C for 20 min. Capillaries filled with the sample mixtures were introduced into the microscale thermophoresis instrument and the measurements were performed using the following settings: LED power 20%, MST power 80%. Microscale thermophoresis measurements were performed as described in “Online Methods”. The observed affinities are indistinguishable in all four cases. (d; here and in f BtuCDEQNC is labeled as “EQNC”). Normalized fluorescence of BtuFfluo (Fnorm) is plotted against transporter concentration. The calculated Kd ± SEM values are as follows: “BtuCD”, 4.3 ± 5 nM (n = 5); “BtuCD + AMPPNP”, 0.85 ± 2.5 nM (n = 4); “EQNC”, 6.8 ± 6.6 nM (n = 7); “EQNC + AMPPNP”, 1.3 ± 3.8 nM (n = 7). The values were not significantly different, judged by one-way ANOVA (P > 0.05). (e) Sedimentation velocity analytical ultracentrifugation, performed as described in “Online Methods” using mixtures of BtuFfluo (10 nM) and BtuCD (10-1000 nM) in the absence or presence of AMPPNP (apparent Kd of 4.9 ± 1.2 nM and 58.0 ± 15.1 nM, respectively; n = 4). (f). Experiments as in e, with mixtures of BtuFfluo and BtuCDEQNC in the absence or presence of AMPPNP (apparent Kd of 8.9 ± 1.1 nM and 109.1 ± 12.8 nM, respectively; n = 2). In the presence of AMPPNP, the interaction appears weakened because separation during a sedimentation velocity experiment is irreversible. Results shown represent the integrated observed fraction of BtuCD-F complex with sedimentation coefficient of ~3.5S (absent in sample with BtuFfluo alone which displays apparent sedimentation coefficient of ~0.8S), plotted against transporter concentration; each data point represents mean ± SD.
Supplementary Figure 4 BtuFfluo binding to proteoliposome-reconstituted BtuCD.
(a) Schematic representation of experiment shown in b, wherein the effect of luminal AMPPNP or ATP (resupplied by ATP regeneration system “ARS” in the presence of Mg2+) on the binding of BtuFfluo was tested. BtuFfluo, but not unlabeled BtuF, is indicated by a green hexagon. (b) Quantitation of bound BtuFfluo is shown under various conditions. The content of the proteoliposome lumen is indicated below the horizontal axis, the concentration of competing, unlabeled BtuF is indicated. Data shown are mean ± SEM, n = 4. (c-d) Experiments were performed as in b, but in the absence of unlabeled BtuF; instead, all reactions were performed in the absence or presence of 100-fold molar excess of vitamin B12 over BtuF (2.5 μM), as illustrated in c. Data shown are mean ± SEM, n = 3; data for “25 nM BtuFfluo” are independent from those shown in b.
Supplementary Figure 5 Binding of BtuFfluo to nanodisc-reconstituted BtuCD.
(a-d) Size exclusion chromatography of BtuFfluo alone (a), mixed with empty nanodiscs (MSP1-E3D1, purified as described in “Online Methods”, omitting the Ni-NTA step prior to gel filtration) (b), BtuCD-containing nanodiscs, purified as described in “Online Methods” (c), or BtuCD-containing nanodiscs and ATP (d). Nanodiscs formed using MSP1-E3D1 scaffold protein, with or without incorporated BtuCD (2-4 μM final) were mixed with BtuFfluo (0.5-1 μM final) and/or ATP (1 mM) in a buffer of following composition: 20 mM Tris (pH 7.5), 200 mM NaCl, 0.5 mM EDTA, 5 mM MgCl2. The mixtures were incubated for 20 min at 4°C and injected into Superdex 200 10/300 GL column; 240 ml fractions were collected. Elution was monitored online using absorbance at 280 nm; fractions were analyzed using fluorescence plate reader. Schematics to the right of each panel illustrate the experimental setup. Each experiment was reproduced 2-4 times in total, using either Superdex 200 10/300 GL or Superdex 200 5/150 GL columns. Peak integration revealed that the ratio of BtuFfluo bound to BtuCD compared to unbound is increased by a factor of 2.3 in the presence of nucleotide (BtuFfluo bound/unbound is 1.71 in the presence of ATP and 0.73 in the absence of ATP). The elution of BtuCD-BtuFfluo complex is indicated by blue arrows, free BtuFfluo is indicated by an asterisk). (e and f) MST experiments with nanodisc-reconstituted BtuCD and BtuFfluo performed at similar conditions as SEC experiments. The concentration of BtuFfluo was 25 nM, BtuCD concentrations ranged from 0.8 nM to 13 μM. Mixtures were incubated for 20 min at 10°C prior to measurements. A Dissociation constant could not be determined accurately in the absence of nucleotide but was estimated as ~24 μM (e). In the presence of ATP and Mg2+, a Kd of 3.6 ± 0.2 μM was determined (f).
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5 and Supplementary Table 1 (PDF 1061 kb)
Rights and permissions
About this article
Cite this article
Korkhov, V., Mireku, S., Veprintsev, D. et al. Structure of AMP-PNP–bound BtuCD and mechanism of ATP-powered vitamin B12 transport by BtuCD–F. Nat Struct Mol Biol 21, 1097–1099 (2014). https://doi.org/10.1038/nsmb.2918
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.2918
This article is cited by
-
Cryo-EM reveals unique structural features of the FhuCDB Escherichia coli ferrichrome importer
Communications Biology (2021)
-
Single-molecule probing of the conformational homogeneity of the ABC transporter BtuCD
Nature Chemical Biology (2018)
-
Vitamin B12 import is all about timing
Nature Chemical Biology (2018)
-
Conformational Change of a Tryptophan Residue in BtuF Facilitates Binding and Transport of Cobinamide by the Vitamin B12 Transporter BtuCD-F
Scientific Reports (2017)
-
Structural basis of nanobody-mediated blocking of BtuF, the cognate substrate-binding protein of the Escherichia coli vitamin B12 transporter BtuCD
Scientific Reports (2017)