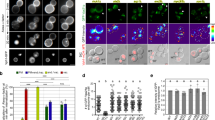
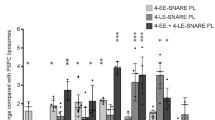
Abstract
Rab small G proteins control membrane trafficking events required for many processes including secretion, lipid metabolism, antigen presentation and growth factor signaling. Rabs recruit effectors that mediate diverse functions including vesicle tethering and fusion. However, many mechanistic questions about Rab-regulated vesicle tethering are unresolved. Using chemically defined reaction systems, we discovered that Vps21, a Saccharomyces cerevisiae ortholog of mammalian endosomal Rab5, functions in trans with itself and with at least two other endosomal Rabs to directly mediate GTP-dependent tethering. Vps21-mediated tethering was stringently and reversibly regulated by an upstream activator, Vps9, and an inhibitor, Gyp1, which were sufficient to drive dynamic cycles of tethering and detethering. These experiments reveal a previously undescribed mode of tethering by endocytic Rabs. In our working model, the intrinsic tethering capacity Vps21 operates in concert with conventional effectors and SNAREs to drive efficient docking and fusion.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Kornmann, B. et al. An ER-mitochondria tethering complex revealed by a synthetic biology screen. Science 325, 477–481 (2009).
Grosshans, B.L., Ortiz, D. & Novick, P. Rabs and their effectors: achieving specificity in membrane traffic. Proc. Natl. Acad. Sci. USA 103, 11821–11827 (2006).
Barr, F.A. Rab GTPase function in Golgi trafficking. Semin. Cell Dev. Biol. 20, 780–783 (2009).
Singer-Kruger, B. et al. Role of three Rab5-like GTPases, Ypt51p, Ypt52p, and Ypt53p, in the endocytic and vacuolar protein sorting pathways of yeast. J. Cell Biol. 125, 283–298 (1994).
Horazdovsky, B.F., Busch, G.R. & Emr, S.D. VPS21 encodes a Rab5-like GTP binding protein that is required for the sorting of yeast vacuolar proteins. EMBO J. 13, 1297–1309 (1994).
Gerrard, S.R., Bryant, N.J. & Stevens, T.H. VPS21 controls entry of endocytosed and biosynthetic proteins into the yeast prevacuolar compartment. Mol. Biol. Cell 11, 613–626 (2000).
Gorvel, J.P., Chavrier, P., Zerial, M. & Gruenberg, J. Rab5 controls early endosome fusion in vitro. Cell 64, 915–925 (1991).
Bucci, C. et al. Co-operative regulation of endocytosis by three Rab5 isoforms. FEBS Lett. 366, 65–71 (1995).
Barbieri, M.A. et al. Evidence for a symmetrical requirement for Rab5-GTP in in vitro endosome-endosome fusion. J. Biol. Chem. 273, 25850–25855 (1998).
Simonsen, A. et al. EEA1 links PI(3)K function to Rab5 regulation of endosome fusion. Nature 394, 494–498 (1998).
Christoforidis, S., McBride, H.M., Burgoyne, R.D. & Zerial, M. The Rab5 effector EEA1 is a core component of endosome docking. Nature 397, 621–625 (1999).
Christoforidis, S. et al. Phosphatidylinositol-3-OH kinases are Rab5 effectors. Nat. Cell Biol. 1, 249–252 (1999).
Lawe, D.C. et al. Sequential roles for phosphatidylinositol 3-phosphate and Rab5 in tethering and fusion of early endosomes via their interaction with EEA1. J. Biol. Chem. 277, 8611–8617 (2002).
Davie, E.W. A brief historical review of the waterfall/cascade of blood coagulation. J. Biol. Chem. 278, 50819–50832 (2003).
Weber, T. et al. SNAREpins: minimal machinery for membrane fusion. Cell 92, 759–772 (1998).
Antonny, B., Madden, D., Hamamoto, S., Orci, L. & Schekman, R. Dynamics of the COPII coat with GTP and stable analogues. Nat. Cell Biol. 3, 531–537 (2001).
Wollert, T. & Hurley, J.H. Molecular mechanism of multivesicular body biogenesis by ESCRT complexes. Nature 464, 864–869 (2010).
Drin, G., Morello, V., Casella, J.F., Gounon, P. & Antonny, B. Asymmetric tethering of flat and curved lipid membranes by a golgin. Science 320, 670–673 (2008).
Hickey, C.M. & Wickner, W. HOPS initiates vacuole docking by tethering membranes prior to trans-SNARE complex assembly. Mol. Biol. Cell 21, 2297–2305 (2010).
Hickey, C.M., Stroupe, C. & Wickner, W. The major role of the Rab Ypt7p in vacuole fusion is supporting HOPS membrane association. J. Biol. Chem. 284, 16118–16125 (2009).
Stroupe, C., Hickey, C.M., Mima, J., Burfeind, A.S. & Wickner, W. Minimal membrane docking requirements revealed by reconstitution of Rab GTPase-dependent membrane fusion from purified components. Proc. Natl. Acad. Sci. USA 106, 17626–17633 (2009).
Peterson, M.R., Burd, C.G. & Emr, S.D. Vac1p coordinates Rab and phosphatidylinositol 3-kinase signaling in Vps45p-dependent vesicle docking/fusion at the endosome. Curr. Biol. 9, 159–162 (1999).
Tall, G.G., Hama, H., DeWald, D.B. & Horazdovsky, B.F. The phosphatidylinositol 3-phosphate binding protein Vac1p interacts with a Rab GTPase and a Sec1p homologue to facilitate vesicle-mediated vacuolar protein sorting. Mol. Biol. Cell 10, 1873–1889 (1999).
Horazdovsky, B.F., Cowles, C.R., Mustol, P., Holmes, M. & Emr, S.D. A novel RING finger protein, Vps8p, functionally interacts with the small GTPase, Vps21p, to facilitate soluble vacuolar protein localization. J. Biol. Chem. 271, 33607–33615 (1996).
Peplowska, K., Markgraf, D.F., Ostrowicz, C.W., Bange, G. & Ungermann, C. The CORVET tethering complex interacts with the yeast Rab5 homolog Vps21 and is involved in endo-lysosomal biogenesis. Dev. Cell 12, 739–750 (2007).
Markgraf, D.F. et al. The CORVET subunit Vps8 cooperates with the Rab5 homolog Vps21 to induce clustering of late endosomal compartments. Mol. Biol. Cell 20, 5276–5289 (2009).
Plemel, R.L. et al. Subunit organization and Rab interactions of Vps-C protein complexes that control endolysosomal membrane traffic. Mol. Biol. Cell 22, 1353–1363 (2011).
Gureasko, J. et al. Membrane-dependent signal integration by the Ras activator Son of sevenless. Nat. Struct. Mol. Biol. 15, 452–461 (2008).
Hochuli, E., Dobeli, H. & Schacher, A. New metal chelate adsorbent selective for proteins and peptides containing neighbouring histidine residues. J. Chromatogr. A 411, 177–184 (1987).
Wang, L., Seeley, E.S., Wickner, W. & Merz, A.J. Vacuole fusion at a ring of vertex docking sites leaves membrane fragments within the organelle. Cell 108, 357–369 (2002).
Ghaemmaghami, S. et al. Global analysis of protein expression in yeast. Nature 425, 737–741 (2003).
Takamori, S. et al. Molecular anatomy of a trafficking organelle. Cell 127, 831–846 (2006).
Patel, S.S. & Rexach, M.F. Discovering novel interactions at the nuclear pore complex using bead halo: a rapid method for detecting molecular interactions of high and low affinity at equilibrium. Mol. Cell. Proteomics 7, 121–131 (2008).
Braun, P. et al. An experimentally derived confidence score for binary protein-protein interactions. Nat. Methods 6, 91–97 (2009).
Chen, Y.C., Rajagopala, S.V., Stellberger, T. & Uetz, P. Exhaustive benchmarking of the yeast two-hybrid system. Nat. Methods 7, 667–668 author reply 668 (2010).
Brett, C.L. et al. Efficient termination of vacuolar Rab GTPase signaling requires coordinated action by a GAP and a protein kinase. J. Cell Biol. 182, 1141–1151 (2008).
Ho, Y. et al. Systematic identification of protein complexes in Saccharomyces cerevisiae by mass spectrometry. Nature 415, 180–183 (2002).
Daitoku, H., Isida, J., Fujiwara, K., Nakajima, T. & Fukamizu, A. Dimerization of small GTPase Rab5. Int. J. Mol. Med. 8, 397–404 (2001).
Pawelec, A., Arsic, J. & Kolling, R. Mapping of Vps21 and HOPS binding sites in Vps8 and effect of binding site mutants on endocytic trafficking. Eukaryot. Cell 9, 602–610 (2010).
Hama, H., Tall, G.G. & Horazdovsky, B.F. Vps9p is a guanine nucleotide exchange factor involved in vesicle-mediated vacuolar protein transport. J. Biol. Chem. 274, 15284–15291 (1999).
Buvelot Frei, S. et al. Bioinformatic and comparative localization of Rab proteins reveals functional insights into the uncharacterized GTPases Ypt10p and Ypt11p. Mol. Cell. Biol. 26, 7299–7317 (2006).
Du, L.L., Collins, R.N. & Novick, P.J. Identification of a Sec4p GTPase-activating protein (GAP) as a novel member of a Rab GAP family. J. Biol. Chem. 273, 3253–3256 (1998).
Pan, X., Eathiraj, S., Munson, M. & Lambright, D.G. TBC-domain GAPs for Rab GTPases accelerate GTP hydrolysis by a dual-finger mechanism. Nature 442, 303–306 (2006).
Lee, M.C. et al. Sar1p N-terminal helix initiates membrane curvature and completes the fission of a COPII vesicle. Cell 122, 605–617 (2005).
Pucadyil, T.J. & Schmid, S.L. Conserved functions of membrane active GTPases in coated vesicle formation. Science 325, 1217–1220 (2009).
Wittmann, J.G. & Rudolph, M.G. Crystal structure of Rab9 complexed to GDP reveals a dimer with an active conformation of switch II. FEBS Lett. 568, 23–29 (2004).
Pasqualato, S. et al. The structural GDP/GTP cycle of Rab11 reveals a novel interface involved in the dynamics of recycling endosomes. J. Biol. Chem. 279, 11480–11488 (2004).
Scapin, S.M. et al. The crystal structure of the small GTPase Rab11b reveals critical differences relative to the Rab11a isoform. J. Struct. Biol. 154, 260–268 (2006).
Beck, R. et al. Membrane curvature induced by Arf1-GTP is essential for vesicle formation. Proc. Natl. Acad. Sci. USA 105, 11731–11736 (2008).
Chapman, E.R. How does synaptotagmin trigger neurotransmitter release? Annu. Rev. Biochem. 77, 615–641 (2008).
Li, F. et al. Energetics and dynamics of SNAREpin folding across lipid bilayers. Nat. Struct. Mol. Biol. 14, 890–896 (2007).
Schwartz, M.L. & Merz, A.J. Capture and release of partially zipped trans-SNARE complexes on intact organelles. J. Cell Biol. 185, 535–549 (2009).
Nielsen, E. et al. Rabenosyn-5, a novel Rab5 effector, is complexed with hVPS45 and recruited to endosomes through a FYVE finger domain. J. Cell Biol. 151, 601–612 (2000).
Furgason, M.L. et al. The N-terminal peptide of the syntaxin Tlg2p modulates binding of its closed conformation to Vps45p. Proc. Natl. Acad. Sci. USA 106, 14303–14308 (2009).
Sinka, R., Gillingham, A.K., Kondylis, V. & Munro, S. Golgi coiled-coil proteins contain multiple binding sites for Rab family G proteins. J. Cell Biol. 183, 607–615 (2008).
Hayes, G.L. et al. Multiple Rab GTPase binding sites in GCC185 suggest a model for vesicle tethering at the trans-Golgi. Mol. Biol. Cell 20, 209–217 (2009).
Angers, C.G. & Merz, A.J. New links between vesicle coats and Rab-mediated vesicle targeting. Semin. Cell Dev. Biol. 22, 18–26 (2011).
Ohya, T. et al. Reconstitution of Rab- and SNARE-dependent membrane fusion by synthetic endosomes. Nature 459, 1091–1097 (2009).
Acknowledgements
We thank P. Brennwald and J. Taraska for helpful discussion and D. Baker and R. Koga for assistance with multiangle light scattering experiments. High-throughput screening and support of M.V. was through the Yeast Resource Center (US National Institutes of Health (NIH) P41 RR11823). S.L. was supported in part by University of Washington Nanotechnology Integrative Graduate Education and Research Traineeship award (US National Science Foundation DGE-0504573). We thank the Murdock Charitable Trust and the Washington Research Foundation for generous support of our electron cryomicroscopy laboratory. T.G. was a Howard Hughes Medical Institute early career scientist and S.F. is a Howard Hughes Medical Institute investigator. This work was supported by NIH grant GM077349 and research scholar grant 10-026-01-CSM from the American Cancer Society.
Author information
Authors and Affiliations
Contributions
S.Y.L. and A.J.M. conceived the project. S.Y.L. developed and validated the QLS-based tethering system; expressed, purified and characterized proteins; prepared liposomes and carried out and interpreted all QLS tethering experiments. C.L.B. and A.J.M. conceived and C.L.B. and S.Y.L. implemented the fluorescence microscopy-based tethering assays. T.G. did the E M. S.F. and M.V. developed the high-throughput yeast two-hybrid technology, and R.L.P. and M.V. executed and interpreted yeast two-hybrid screens and assays. S.Y.L. and A.J.M. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–4, Supplementary Tables 1–4 and Supplementary Methods (PDF 2106 kb)
Rights and permissions
About this article
Cite this article
Lo, SY., Brett, C., Plemel, R. et al. Intrinsic tethering activity of endosomal Rab proteins. Nat Struct Mol Biol 19, 40–47 (2012). https://doi.org/10.1038/nsmb.2162
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.2162
This article is cited by
-
Self-assemblies of Rab- and Arf-family small GTPases on lipid bilayers in membrane tethering
Biophysical Reviews (2021)
-
Reconstitution of membrane tethering mediated by Rab-family small GTPases
Biophysical Reviews (2018)
-
An endosomal tether undergoes an entropic collapse to bring vesicles together
Nature (2016)
-
Neuropeptide substance P and the immune response
Cellular and Molecular Life Sciences (2016)
-
Multiple roles for the actin cytoskeleton during regulated exocytosis
Cellular and Molecular Life Sciences (2013)