Abstract
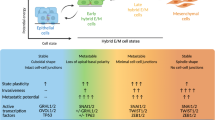
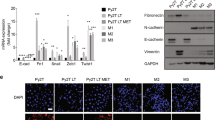
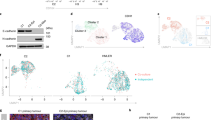
Epithelial-to-mesenchymal transition (EMT) is an extreme example of cell plasticity that is important for normal development, injury repair and malignant progression. Widespread epigenetic reprogramming occurs during stem cell differentiation and malignant transformation, but EMT-related epigenetic reprogramming is poorly understood. Here we investigated epigenetic modifications during EMT mediated by transforming growth factor beta. Although DNA methylation was unchanged during EMT, we found a global reduction in the heterochromatin mark H3 Lys9 dimethylation (H3K9Me2), an increase in the euchromatin mark H3 Lys4 trimethylation (H3K4Me3) and an increase in the transcriptional mark H3 Lys36 trimethylation (H3K36Me3). These changes depended largely on lysine-specific demethylase-1 (Lsd1), and loss of Lsd1 function had marked effects on EMT-driven cell migration and chemoresistance. Genome-scale mapping showed that chromatin changes were mainly specific to large organized heterochromatin K9 modifications (LOCKs), which suggests that EMT is characterized by reprogramming of specific chromatin domains across the genome.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Kalluri, R. & Weinberg, R.A. The basics of epithelial-mesenchymal transition. J. Clin. Invest. 119, 1420–1428 (2009).
Thiery, J.P., Acloque, H., Huang, R.Y.J. & Nieto, M.A. Epithelial-mesenchymal transitions in development and disease. Cell 139, 871–890 (2009).
Mani, S.A. et al. The epithelial-mesenchymal transition generates cells with properties of stem cells. Cell 133, 704–715 (2008).
Gupta, P.B. et al. Identification of selective inhibitors of cancer stem cells by high-throughput screening. Cell 138, 645–659 (2009).
Feinberg, A.P. Phenotypic plasticity and the epigenetics of human disease. Nature 447, 433–440 (2007).
Jenuwein, T. & Allis, C.D. Translating the histone code. Science 293, 1074–1080 (2001).
Hawkins, R.D. et al. Distinct epigenomic landscapes of pluripotent and lineage-committed human cells. Cell Stem Cell 6, 479–491 (2010).
Jones, P.A. & Baylin, S.B. The epigenomics of cancer. Cell 128, 683–692 (2007).
Roussos, E.T. et al. AACR Special Conference on Epithelial-Mesenchymal Transition and Cancer Progression and Treatment. Cancer Res. 70, 7360–7364 (2010).
Rahimi, R.A. & Leof, E.B. TGF-β signaling: a tale of two responses. J. Cell. Biochem. 102, 593–608 (2007).
Xu, J., Lamouille, S. & Derynck, R. TGF-β-induced epithelial to mesenchymal transition. Cell Res. 19, 156–172 (2009).
Dooley, S. et al. Hepatocyte-specific Smad7 expression attenuates TGF-β-mediated fibrogenesis and protects against liver damage. Gastroenterology 135, 642–659 (2008).
Yang, Y., Pan, X., Lei, W., Wang, J. & Song, J. Transforming growth factor-beta1 induces epithelial-to-mesenchymal transition and apoptosis via a cell cycle-dependent mechanism. Oncogene 25, 7235–7244 (2006).
Kaimori, A. et al. Transforming growth factor-β induces an epithelial-to-mesenchymal transition state in mouse hepatocytes in vitro. J. Biol. Chem. 282, 22089–22101 (2007).
Pan, X. et al. Nitric oxide suppresses transforming growth factor-β1–induced epithelial-to-mesenchymal transition and apoptosis in mouse hepatocytes. Hepatology 50, 1577–1587 (2009).
Wu, J.C., Merlino, G. & Fausto, N. Establishment and characterization of differentiated, nontransformed hepatocyte cell lines derived from mice transgenic for transforming growth factor alpha. Proc. Natl. Acad. Sci. USA 91, 674–678 (1994).
Mills, S.E. et al. Sternbergs Diagnostic Surgical Pathology (Lippincott Williams & Wilkins, Philadelphia, 2004).
Dumont, N. et al. Sustained induction of epithelial to mesenchymal transition activates DNA methylation of genes silenced in basal-like breast cancers. Proc. Natl. Acad. Sci. USA 105, 14867–14872 (2008).
Irizarry, R.A. et al. Comprehensive high-throughput arrays for relative methylation (CHARM). Genome Res. 18, 780–790 (2008).
Irizarry, R.A. et al. The human colon cancer methylome shows similar hypo- and hypermethylation at conserved tissue-specific CpG island shores. Nat. Genet. 41, 178–186 (2009).
Tachibana, M. et al. G9a histone methyltransferase plays a dominant role in euchromatic histone H3 lysine 9 methylation and is essential for early embryogenesis. Genes Dev. 16, 1779–1791 (2002).
Rice, J.C. et al. Histone methyltransferases direct different degrees of methylation to define distinct chromatin domains. Mol. Cell 12, 1591–1598 (2003).
Peters, A.H.F.M. et al. Partitioning and plasticity of repressive histone methylation states in mammalian chromatin. Mol. Cell 12, 1577–1589 (2003).
Wen, B., Wu, H., Shinkai, Y., Irizarry, R.A. & Feinberg, A.P. Large histone H3 lysine 9 dimethylated chromatin blocks distinguish differentiated from embryonic stem cells. Nat. Genet. 41, 246–250 (2009).
Chen, E.S. et al. Cell cycle control of centromeric repeat transcription and heterochromatin assembly. Nature 451, 734–737 (2008).
Kizer, K.O. et al. A novel domain in Set2 mediates RNA polymerase II interaction and couples histone H3 K36 methylation with transcript elongation. Mol. Cell. Biol. 25, 3305–3316 (2005).
Mikkelsen, T.S. et al. Genome-wide maps of chromatin state in pluripotent and lineage-committed cells. Nature 448, 553–560 (2007).
Kouzarides, T. Chromatin modifications and their function. Cell 128, 693–705 (2007).
Onder, T.T. et al. Loss of E-cadherin promotes metastasis via multiple downstream transcriptional pathways. Cancer Res. 68, 3645–3654 (2008).
Zheng, G. et al. Disruption of E-cadherin by matrix metalloproteinase directly mediates epithelial-mesenchymal transition downstream of transforming growth factor-β in renal tubular epithelial cells. Am. J. Pathol. 175, 580–591 (2009).
Shi, Y. et al. Histone demethylation mediated by the nuclear amine oxidase homolog Lsd1. Cell 119, 941–953 (2004).
Metzger, E. et al. Lsd1 demethylates repressive histone marks to promote androgen-receptor-dependent transcription. Nature 437, 436–439 (2005).
Wang, Y. et al. Lsd1 is a subunit of the NuRD complex and targets the metastasis programs in breast cancer. Cell 138, 660–672 (2009).
Shi, Y.-J. Regulation of Lsd1 histone demethylase activity by its associated factors. Mol. Cell 19, 857–864 (2005).
Wang, J. et al. Opposing Lsd1 complexes function in developmental gene activation and repression programmes. Nature 446, 882–887 (2007).
Liu, Y., Subrahmanyam, R., Chakraborty, T., Sen, R. & Desiderio, S. A plant homeodomain in Rag-2 that binds hypermethylated lysine 4 of histone H3 is necessary for efficient antigen-receptor-gene rearrangement. Immunity 27, 561–571 (2007).
Matthews, A.G.W. et al. RAG2 PHD finger couples histone H3 lysine 4 trimethylation with V(D)J recombination. Nature 450, 1106–1110 (2007).
Borde, V. et al. Histone H3 lysine 4 trimethylation marks meiotic recombination initiation sites. EMBO J. 28, 99–111 (2009).
Faucher, D. & Wellinger, R.J. Methylated H3K4, a transcription-associated histone modification, is involved in the DNA damage response pathway. PLoS Genet. 6, e1001082 (2010).
Guelen, L. et al. Domain organization of human chromosomes revealed by mapping of nuclear lamina interactions. Nature 453, 948–951 (2008).
Huang, D.W., Sherman, B.T. & Lempicki, R.A. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat. Protoc. 4, 44–57 (2008).
Bernstein, B.E. et al. A bivalent chromatin structure marks key developmental genes in embryonic stem cells. Cell 125, 315–326 (2006).
Misteli, T. & Soutoglou, E. The emerging role of nuclear architecture in DNA repair and genome maintenance. Nat. Rev. Mol. Cell Biol. 10, 243–254 (2009).
Chosed, R. & Dent, S.Y.R.A. Two-way street: Lsd1 regulates chromatin boundary formation in S. pombe and Drosophila. Mol. Cell 26, 160–162 (2007).
Sierra, J., Yoshida, T., Joazeiro, C.A. & Jones, K.A. The APC tumor suppressor counteracts β-catenin activation and H3K4 methylation at Wnt target genes. Genes Dev. 20, 586–600 (2006).
Lin, T., Ponn, A., Hu, X., Law, B.K. & Lu, J. Requirement of the histone demethylase Lsd1 in Snai1-mediated transcriptional repression during epithelial-mesenchymal transition. Oncogene 29, 4896–4904 (2010).
Efroni, S. et al. Global transcription in pluripotent embryonic stem cells. Cell Stem Cell 2, 437–447 (2008).
Samavarchi-Tehrani, P. et al. Functional genomics reveals a BMP-driven mesenchymal-to-epithelial transition in the initiation of somatic cell reprogramming. Cell Stem Cell 7, 64–77 (2010).
Li, R. et al. A mesenchymal-to-epithelial transition initiates and is required for the nuclear reprogramming of mouse fibroblasts. Cell Stem Cell 7, 51–63 (2010).
Lin, C. et al. Nuclear receptor-induced chromosomal proximity and DNA breaks underlie specific translocations in cancer. Cell 139, 1069–1083 (2009).
Haffner, M.C. et al. Androgen-induced TOP2B-mediated double-strand breaks and prostate cancer gene rearrangements. Nat. Genet. 42, 668–675 (2010).
Ju, B.-G. et al. A topoisomerase IIβ-mediated dsDNA break required for regulated transcription. Science 312, 1798–1802 (2006).
Feng, S., Jacobsen, S.E. & Reik, W. Epigenetic reprogramming in plant and animal development. Science 330, 622–627 (2010).
Shechter, D., Dormann, H.L., Allis, C.D. & Hake, S.B. Extraction, purification and analysis of histones. Nat. Protoc. 2, 1445–1457 (2007).
Litt, M.D., Simpson, M., Recillas-Targa, F., Prioleau, M.-N. & Felsenfeld, G. Transitions in histone acetylation reveal boundaries of three separately regulated neighboring loci. EMBO J. 20, 2224–2235 (2001).
Lee, M.G., Wynder, C., Norman, J. & Shiekhattar, R. Isolation and characterization of histone H3 lysine 4 demethylase-containing complexes. Methods 40, 327–330 (2006).
Acknowledgements
This work was supported by US National Institutes of Health grant 5R37CA054358 to A.P.F. We thank A. Tackett for assistance with mass spectrometry experiments and data analysis; C. McCall for advice on large-scale immunoprecipitation experiments; the microscopy facility at Johns Hopkins University for electron microscopy; and K. Reddy and S. Taverna for advice.
Author information
Authors and Affiliations
Contributions
O.G.M. and A.P.F. conceived this work, designed the experiments and wrote the manuscript. H.W. performed the statistical analyses. W.T. performed the immunofluorescence. A.D. performed the CHARM.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–9 and Supplementary Tables 1–5 (PDF 2802 kb)
Supplementary Data 1
Locations of K9Me2 LOCKs identified in AML12 cells (XLSX 183 kb)
Rights and permissions
About this article
Cite this article
McDonald, O., Wu, H., Timp, W. et al. Genome-scale epigenetic reprogramming during epithelial-to-mesenchymal transition. Nat Struct Mol Biol 18, 867–874 (2011). https://doi.org/10.1038/nsmb.2084
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.2084
This article is cited by
-
PRC2 mediated KLF2 down regulation: a therapeutic and diagnostic axis during tumor progression
Cancer Cell International (2023)
-
The lncRNA HOTAIR: a pleiotropic regulator of epithelial cell plasticity
Journal of Experimental & Clinical Cancer Research (2023)
-
Nuclear lamina strain states revealed by intermolecular force biosensor
Nature Communications (2023)
-
Epigenetic markers and therapeutic targets for metastasis
Cancer and Metastasis Reviews (2023)
-
Pancreatic cancer epigenetics: adaptive metabolism reprograms starving primary tumors for widespread metastatic outgrowth
Cancer and Metastasis Reviews (2023)