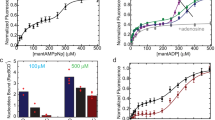
Abstract
E. coli RecBCD is a DNA helicase with two ATPase motors (RecB, a 3′→5′ translocase, and RecD, a 5′→3′ translocase) that function in repair of double-stranded DNA breaks. The RecBC heterodimer, with only the RecB motor, remains a processive helicase. Here we examined RecBC translocation along single-stranded DNA (ssDNA). Notably, we found RecBC to have two translocase activities: the primary translocase moves 3′→5′, whereas the secondary translocase moves RecBC along the opposite strand of a forked DNA at a similar rate. The secondary translocase is insensitive to the ssDNA backbone polarity, and we propose that it may fuel RecBCD translocation along double-stranded DNA ahead of the unwinding fork and ensure that the unwound single strands move through RecBCD at the same rate after interaction with a crossover hot-spot indicator (Chi) sequence.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Dillingham, M.S. & Kowalczykowski, S.C. RecBCD enzyme and the repair of double-stranded DNA breaks. Microbiol. Mol. Biol. Rev. 72, 642–671 (2008).
Singleton, M.R., Dillingham, M.S. & Wigley, D.B. Structure and mechanism of helicases and nucleic acid translocases. Annu. Rev. Biochem. 76, 23–50 (2007).
Dillingham, M.S., Spies, M. & Kowalczykowski, S.C. RecBCD enzyme is a bipolar DNA helicase. Nature 423, 893–897 (2003).
Taylor, A.F. & Smith, G.R. RecBCD enzyme is a DNA helicase with fast and slow motors of opposite polarity. Nature 423, 889–893 (2003).
Rigden, D.J. An inactivated nuclease-like domain in RecC with novel function: implications for evolution. BMC Struct. Biol. 5, 9 (2005).
Singleton, M.R., Dillingham, M.S., Gaudier, M., Kowalczykowski, S.C. & Wigley, D.B. Crystal structure of RecBCD enzyme reveals a machine for processing DNA breaks. Nature 432, 187–193 (2004).
Spies, M., Amitani, I., Baskin, R.J. & Kowalczykowski, S.C. RecBCD enzyme switches lead motor subunits in response to chi recognition. Cell 131, 694–705 (2007).
Bianco, P.R. & Kowalczykowski, S.C. Translocation step size and mechanism of the RecBC DNA helicase. Nature 405, 368–372 (2000).
Korangy, F. & Julin, D.A. Efficiency of ATP hydrolysis and DNA unwinding by the RecBC enzyme from Escherichia coli. Biochemistry 33, 9552–9560 (1994).
Wu, C.G. & Lohman, T.M. Influence of DNA end structure on the mechanism of initiation of DNA unwinding by the Escherichia coli RecBCD and RecBC helicases. J. Mol. Biol. 382, 312–326 (2008).
Ganesan, S. & Smith, G.R. Strand-specific binding to duplex DNA ends by the subunits of the Escherichia coli RecBCD enzyme. J. Mol. Biol. 229, 67–78 (1993).
Farah, J.A. & Smith, G.R. The RecBCD enzyme initiation complex for DNA unwinding: enzyme positioning and DNA opening. J. Mol. Biol. 272, 699–715 (1997).
Wong, C.J., Lucius, A.L. & Lohman, T.M. Energetics of DNA end binding by E. coli RecBC and RecBCD helicases indicate loop formation in the 3′-single-stranded DNA tail. J. Mol. Biol. 352, 765–782 (2005).
Wong, C.J., Rice, R.L., Baker, N.A., Ju, T. & Lohman, T.M. Probing 3′-ssDNA loop formation in E. coli RecBCD/RecBC-DNA complexes using non-natural DNA: a model for “Chi” recognition complexes. J. Mol. Biol. 362, 26–43 (2006).
Taylor, A. & Smith, G.R. Unwinding and rewinding of DNA by the RecBC enzyme. Cell 22, 447–457 (1980).
Bianco, P.R. et al. Processive translocation and DNA unwinding by individual RecBCD enzyme molecules. Nature 409, 374–378 (2001).
Spies, M. et al. A molecular throttle: the recombination hotspot chi controls DNA translocation by the RecBCD helicase. Cell 114, 647–654 (2003).
Lucius, A.L. et al. DNA unwinding step-size of E. coli RecBCD helicase determined from single turnover chemical quenched-flow kinetic studies. J. Mol. Biol. 324, 409–428 (2002).
Lucius, A.L. & Lohman, T.M. Effects of temperature and ATP on the kinetic mechanism and kinetic step-size for E. coli RecBCD helicase-catalyzed DNA unwinding. J. Mol. Biol. 339, 751–771 (2004).
Dillingham, M.S., Webb, M.R. & Kowalczykowski, S.C. Bipolar DNA translocation contributes to highly processive DNA unwinding by RecBCD enzyme. J. Biol. Chem. 280, 37069–37077 (2005).
Saikrishnan, K., Griffiths, S.P., Cook, N., Court, R. & Wigley, D.B. DNA binding to RecD: role of the 1B domain in SF1B helicase activity. EMBO J. 27, 2222–2229 (2008).
Wong, C.J. & Lohman, T.M. Kinetic control of Mg2+-dependent melting of duplex DNA ends by Escherichia coli RecBC. J. Mol. Biol. 378, 761–777 (2008).
Boehmer, P.E. & Emmerson, P.T. The RecB subunit of the Escherichia coli RecBCD enzyme couples ATP hydrolysis to DNA unwinding. J. Biol. Chem. 267, 4981–4987 (1992).
Phillips, R.J., Hickleton, D.C., Boehmer, P.E. & Emmerson, P.T. The RecB protein of Escherichia coli translocates along single-stranded DNA in the 3′ to 5′ direction: a proposed ratchet mechanism. Mol. Gen. Genet. 254, 319–329 (1997).
Fischer, C.J., Maluf, N.K. & Lohman, T.M. Mechanism of ATP-dependent translocation of E. coli UvrD monomers along single-stranded DNA. J. Mol. Biol. 344, 1287–1309 (2004).
Fischer, C.J. & Lohman, T.M. ATP-dependent translocation of proteins along single-stranded DNA: models and methods of analysis of pre-steady state kinetics. J. Mol. Biol. 344, 1265–1286 (2004).
Lucius, A.L., Maluf, N.K., Fischer, C.J. & Lohman, T.M. General methods for analysis of sequential “n-step” kinetic mechanisms: application to single turnover kinetics of helicase-catalyzed DNA unwinding. Biophys. J. 85, 2224–2239 (2003).
Hickson, I.D., Robson, C.N., Atkinson, K.E., Hutton, L. & Emmerson, P.T. Reconstitution of RecBC DNase activity from purified Escherichia coli RecB and RecC proteins. J. Biol. Chem. 260, 1224–1229 (1985).
Brendza, K.M. et al. Autoinhibition of Escherichia coli Rep monomer helicase activity by its 2B subdomain. Proc. Natl. Acad. Sci. USA 102, 10076–10081 (2005).
Dillingham, M.S., Wigley, D.B. & Webb, M.R. Direct measurement of single-stranded DNA translocation by PcrA helicase using the fluorescent base analogue 2-aminopurine. Biochemistry 41, 643–651 (2002).
Niedziela-Majka, A., Chesnik, M.A., Tomko, E.J. & Lohman, T.M. Bacillus stearothermophilus PcrA monomer is a single-stranded DNA translocase but not a processive helicase in vitro. J. Biol. Chem. 282, 27076–27085 (2007).
Tomko, E.J., Fischer, C.J., Niedziela-Majka, A. & Lohman, T.M. A nonuniform stepping mechanism for E. coli UvrD monomer translocation along single-stranded DNA. Mol. Cell 26, 335–347 (2007).
Antony, E. et al. Srs2 disassembles Rad51 filaments by a protein-protein interaction triggering ATP turnover and dissociation of Rad51 from DNA. Mol. Cell 35, 105–115 (2009).
Yeeles, J.T. & Dillingham, M.S.A. Dual-nuclease mechanism for DNA break processing by AddAB-type helicase-nucleases. J. Mol. Biol. 371, 66–78 (2007).
Unciuleac, M.C. & Shuman, S. Characterization of the mycobacterial AdnAB DNA motor provides insights into the evolution of bacterial motor-nuclease machines. J. Biol. Chem. 285, 2632–2641 (2010).
Lucius, A.L., Jason Wong, C. & Lohman, T.M. Fluorescence stopped-flow studies of single turnover kinetics of E. coli RecBCD helicase-catalyzed DNA unwinding. J. Mol. Biol. 339, 731–750 (2004).
Acknowledgements
We thank R. Galletto, C. Fischer, A. Lucius, K. Maluf, E. Galburt, N. Baker, P. Burgers, G. Smith, T. Ellenberger, S. Kowalczykowski and E. Antony for valuable discussions and comments on the manuscript, G. Smith (Fred Hutchinson Cancer Research Center), A. Taylor (Fred Hutchinson Cancer Research Center) and D. Julin (University of Maryland, College Park) for plasmids and cell lines and T. Ho (Washington University School of Medicine) for synthesis and purification of DNA. This work was supported in part by US National Institutes of Health grant GM045948 (to T.M.L.).
Author information
Authors and Affiliations
Contributions
C.G.W. and T.M.L. designed the experiments. C.G.W. purified the protein, carried out the stopped-flow and DNA-unwinding experiments and analyzed the data; C.B. and C.G.W. carried out the fluorescence resonance energy transfer and RecBC translocation experiments as a function of NaCl concentration. T.M.L. supervised the study, and C.G.W. and T.M.L. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Methods, Supplementary Figures 1–6, Supplementary Tables 1–3 and Supplementary Schemes 1–3 (including Supplementary Equations 1–3) (PDF 750 kb)
Rights and permissions
About this article
Cite this article
Wu, C., Bradford, C. & Lohman, T. Escherichia coli RecBC helicase has two translocase activities controlled by a single ATPase motor. Nat Struct Mol Biol 17, 1210–1217 (2010). https://doi.org/10.1038/nsmb.1901
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.1901
This article is cited by
-
Auxiliary ATP binding sites support DNA unwinding by RecBCD
Nature Communications (2022)
-
Structural basis for translocation by AddAB helicase–nuclease and its arrest at χ sites
Nature (2014)
-
Bacterial DNA repair: recent insights into the mechanism of RecBCD, AddAB and AdnAB
Nature Reviews Microbiology (2013)
-
One motor driving two translocases
Nature Structural & Molecular Biology (2010)