Abstract
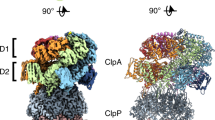
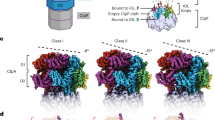
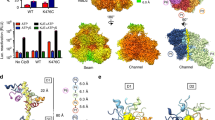
Proteolytic AAA+ unfoldases use ATP hydrolysis to power conformational changes that mechanically denature protein substrates and then translocate the polypeptide through a narrow pore into a degradation chamber. We show that a tyrosine residue in a pore loop of the hexameric ClpX unfoldase links ATP hydrolysis to mechanical work by gripping substrates during unfolding and translocation. Removal of the aromatic ring in even a few ClpX subunits results in slippage, frequent failure to denature the substrate and an enormous increase in the energetic cost of substrate unfolding. The tyrosine residue is part of a conserved aromatic-hydrophobic motif, and the effects of mutations in both residues vary with the nucleotide state of the resident subunit. These results support a model in which nucleotide-dependent conformational changes in these pore loops drive substrate translocation and unfolding, with the aromatic ring transmitting force to the polypeptide substrate.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Ogura, T. & Wilkinson, A.J. AAA+ superfamily ATPases: common structure—diverse function. Genes Cells 6, 575–597 (2001).
Hanson, P.I. & Whiteheart, S.W. AAA+ proteins: have engine, will work. Nat. Rev. Mol. Cell Biol. 6, 519–529 (2005).
Gottesman, S., Maurizi, M.R. & Wickner, S. Regulatory subunits of energy-dependent proteases. Cell 91, 435–438 (1997).
Neuwald, A.F., Aravind, L., Spouge, J.L. & Koonin, E.V. AAA+: a class of chaperone-like ATPases associated with the assembly, operation, and disassembly of protein complexes. Genome Res. 9, 27–43 (1999).
Sauer, R.T. et al. Sculpting the proteome with AAA+ proteases and disassembly machines. Cell 119, 9–18 (2004).
Wang, J. et al. Crystal structures of the HslVU peptidase-ATPase complex reveal an ATP-dependent proteolysis mechanism. Structure 9, 177–184 (2001).
Yamada-Inagawa, T., Okuno, T., Karata, K., Yamanaka, K. & Ogura, T. Conserved pore residues in the AAA protease FtsH are important for proteolysis and its coupling to ATP hydrolysis. J. Biol. Chem. 278, 50182–50187 (2003).
Wang, J. et al. Nucleotide-dependent conformational changes in a protease-associated ATPase HsIU. Structure 9, 1107–1116 (2001).
Song, H.K. et al. Mutational studies on HslU and its docking mode with HslV. Proc. Natl. Acad. Sci. USA 97, 14103–14108 (2000).
Lum, R., Tkach, J.M., Vierling, E. & Glover, J.R. Evidence for an unfolding/threading mechanism for protein disaggregation by Saccharomyces cerevisiae Hsp104. J. Biol. Chem. 279, 29139–29146 (2004).
Schlieker, C. et al. Substrate recognition by the AAA+ chaperone ClpB. Nat. Struct. Mol. Biol. 11, 607–615 (2004).
Siddiqui, S.M., Sauer, R.T. & Baker, T.A. Role of the protein-processing pore of ClpX, a AAA+ ATPase, in recognition and engagement of specific protein substrates. Genes Dev. 18, 369–374 (2004).
Weibezahn, J. et al. Thermotolerance requires refolding of aggregated proteins by substrate translocation through the central pore of ClpB. Cell 119, 653–665 (2004).
Park, E. et al. Role of the GYVG pore motif of HslU ATPase in protein unfolding and translocation for degradation by HslV peptidase. J. Biol. Chem. 280, 22892–22898 (2005).
Hinnerwisch, J., Fenton, W.A., Furtak, K.J., Farr, G.W. & Horwich, A.L. Loops in the central channel of ClpA chaperone mediate protein binding, unfolding, and translocation. Cell 121, 1029–1041 (2005).
Okuno, T., Yamanaka, K. & Ogura, T. Characterization of mutants of the Escherichia coli AAA protease, FtsH, carrying a mutation in the central pore region. J. Struct. Biol. 156, 109–114 (2006).
Graef, M. & Langer, T. Substrate specific consequences of central pore mutations in the i-AAA protease Yme1 on substrate engagement. J. Struct. Biol. 156, 101–108 (2006).
Kenniston, J.A., Baker, T.A., Fernandez, J.M. & Sauer, R.T. Linkage between ATP consumption and mechanical unfolding during the protein processing reactions of a AAA+ degradation machine. Cell 114, 511–520 (2003).
Singleton, M.R., Sawaya, M.R., Ellenberger, T. & Wigley, D.B. Crystal structure of T7 gene 4 ring helicase indicates a mechanism for sequential hydrolysis of nucleotides. Cell 101, 589–600 (2000).
Hishida, T., Han, Y.W., Fujimoto, S., Iwasaki, H. & Shinagawa, H. Direct evidence that a conserved arginine in RuvB AAA+ ATPase acts as an allosteric effector for the ATPase activity of the adjacent subunit in a hexamer. Proc. Natl. Acad. Sci. USA 101, 9573–9577 (2004).
Hersch, G.L., Burton, R.E., Bolon, D.N., Baker, T.A. & Sauer, R.T. Asymmetric interactions of ATP with the AAA+ ClpX6 unfoldase: allosteric control of a protein machine. Cell 121, 1017–1027 (2005).
Martin, A., Baker, T.A. & Sauer, R.T. Rebuilt AAA+ motors reveal operating principles for ATP-fueled machines. Nature 437, 1115–1120 (2005).
Enemark, E.J. & Joshua-Tor, L. Mechanism of DNA translocation in a replicative hexameric helicase. Nature 442, 270–275 (2006).
Joshi, S.A., Hersch, G.L., Baker, T.A. & Sauer, R.T. Communication between ClpX and ClpP during substrate processing and degradation. Nat. Struct. Mol. Biol. 11, 404–411 (2004).
Martin, A., Baker, T.A. & Sauer, R.T. Diverse pore loops of the AAA+ ClpX machine mediate unassisted and adaptor-dependent recognition of ssrA-tagged substrates. Mol. Cell 29, 441–450 (2008).
Kenniston, J.A., Baker, T.A. & Sauer, R.T. Partitioning between unfolding and release of native domains during ClpXP degradation determines substrate selectivity and partial processing. Proc. Natl. Acad. Sci. USA 102, 1390–1395 (2005).
Martin, A., Baker, T.A. & Sauer, R.T. Protein unfolding by a AAA+ protease is dependent on ATP-hydrolysis rates and substrate energy landscapes. Nat. Struct. Mol. Biol. 15, 139–145 (2008).
Thompson, M.W. & Maurizi, M.R. Activity and specificity of Escherichia coli ClpAP protease in cleaving model peptide substrates. J. Biol. Chem. 269, 18201–18208 (1994).
Bochtler, M. et al. The structures of HsIU and the ATP-dependent protease HsIU-HsIV. Nature 403, 800–805 (2000).
Bieniossek, C. et al. The molecular architecture of the metalloprotease FtsH. Proc. Natl. Acad. Sci. USA 103, 3066–3071 (2006).
Martin, A., Baker, T.A. & Sauer, R.T. Distinct static and dynamic interactions control ATPase-peptidase communication in a AAA+ protease. Mol. Cell 27, 41–52 (2007).
Burton, R.E., Baker, T.A. & Sauer, R.T. Energy-dependent degradation: linkage between ClpX-catalyzed nucleotide hydrolysis and protein-substrate processing. Protein Sci. 12, 893–902 (2003).
Acknowledgements
This research was supported by the US National Institutes of Health grant AI-15706. We thank J. Davis, E. Gur, A. Keating, M. Laub and S. Sundar for helpful discussions. A.M. was supported by a Merck/Massachusetts Institute of Technology CSBi postdoctoral fellowship. T.A.B. is an employee of the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Contributions
A.M. designed and performed experiments; A.M., T.A.B. and R.T.S. analyzed data and wrote the manuscript.
Corresponding author
Rights and permissions
About this article
Cite this article
Martin, A., Baker, T. & Sauer, R. Pore loops of the AAA+ ClpX machine grip substrates to drive translocation and unfolding. Nat Struct Mol Biol 15, 1147–1151 (2008). https://doi.org/10.1038/nsmb.1503
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.1503
This article is cited by
-
A novel and atypical NF-KB pro-inflammatory program regulated by a CamKII-proteasome axis is involved in the early activation of Muller glia by high glucose
Cell & Bioscience (2022)
-
Atomic structures of anthrax toxin protective antigen channels bound to partially unfolded lethal and edema factors
Nature Communications (2020)
-
The molecular principles governing the activity and functional diversity of AAA+ proteins
Nature Reviews Molecular Cell Biology (2020)
-
Molecular and structural insights into an asymmetric proteolytic complex (ClpP1P2) from Mycobacterium smegmatis
Scientific Reports (2019)
-
The peroxisomal AAA-ATPase Pex1/Pex6 unfolds substrates by processive threading
Nature Communications (2018)