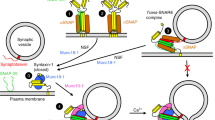
Abstract
The core of the neurotransmitter release machinery is formed by SNARE complexes, which bring the vesicle and plasma membranes together and are key for fusion, and by Munc18-1, which controls SNARE-complex formation and may also have a direct role in fusion. In addition, SNARE complex assembly is likely orchestrated by Munc13s and RIMs, active-zone proteins that function in vesicle priming and diverse forms of presynaptic plasticity. Synaptotagmin-1 mediates triggering of release by Ca2+, probably through interactions with SNAREs and both membranes, as well as through a tight interplay with complexins. Elucidation of the release mechanism will require a full understanding of the network of interactions among all these proteins and the membranes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Sudhof, T.C. The synaptic vesicle cycle. Annu. Rev. Neurosci. 27, 509–547 (2004).
Rosenmund, C., Rettig, J. & Brose, N. Molecular mechanisms of active zone function. Curr. Opin. Neurobiol. 13, 509–519 (2003).
Brunger, A.T. Structure and function of SNARE and SNARE-interacting proteins. Q. Rev. Biophys. 38, 1–47 (2005).
Rizo, J., Chen, X. & Arac, D. Unraveling the mechanisms of synaptotagmin and SNARE function in neurotransmitter release. Trends Cell Biol. 16, 339–350 (2006).
Jahn, R. & Scheller, R.H. SNAREs — engines for membrane fusion. Nat. Rev. Mol. Cell Biol. 7, 631–643 (2006).
Verhage, M. & Toonen, R.F. Regulated exocytosis: merging ideas on fusing membranes. Curr. Opin. Cell Biol. 19, 402–408 (2007).
Chapman, E.R. How does synaptotagmin trigger neurotransmitter release? Annu. Rev. Biochem. 77, 615–641 (2008).
Montecucco, C., Schiavo, G. & Pantano, S. SNARE complexes and neuroexocytosis: how many, how close? Trends Biochem. Sci. 30, 367–372 (2005).
Sollner, T., Bennett, M.K., Whiteheart, S.W., Scheller, R.H. & Rothman, J.E. A protein assembly-disassembly pathway in vitro that may correspond to sequential steps of synaptic vesicle docking, activation, and fusion. Cell 75, 409–418 (1993).
Sollner, T. et al. SNAP receptors implicated in vesicle targeting and fusion. Nature 362, 318–324 (1993). This paper introduces the SNARE hypothesis and postulates that a universal membrane fusion apparatus includes vesicle SNAREs and target-membrane SNAREs that bind to each other and mediate targeting specificity.
Sutton, R.B., Fasshauer, D., Jahn, R. & Brunger, A.T. Crystal structure of a SNARE complex involved in synaptic exocytosis at 2.4 Å resolution. Nature 395, 347–353 (1998). The crystal structure of the neuronal SNARE complex is described, providing a structural basis for understanding SNARE function at atomic resolution.
Poirier, M.A. et al. The synaptic SNARE complex is a parallel four-stranded helical bundle. Nat. Struct. Biol. 5, 765–769 (1998).
Hanson, P.I., Roth, R., Morisaki, H., Jahn, R. & Heuser, J.E. Structure and conformational changes in NSF and its membrane receptor complexes visualized by quick-freeze/deep-etch electron microscopy. Cell 90, 523–535 (1997). The SNARE motifs of syntaxin-1 and synaptobrevin are shown to bind in a parallel fashion, leading to the proposal that SNARE complex assembly may provide the energy for membrane fusion.
Weber, T. et al. SNAREpins: minimal machinery for membrane fusion. Cell 92, 759–772 (1998). This paper describes reconstitution experiments that provide a powerful tool to study the role of diverse factors in membrane fusion and suggested that SNAREs alone may be able to induce membrane fusion.
Hu, C. et al. Fusion of cells by flipped SNAREs. Science 300, 1745–1749 (2003).
Fernandez, I. et al. Three-dimensional structure of an evolutionarily conserved N-terminal domain of syntaxin 1A. Cell 94, 841–849 (1998).
Dulubova, I. et al. A conformational switch in syntaxin during exocytosis: role of munc18. EMBO J. 18, 4372–4382 (1999). Syntaxin-1 is shown to adopt a closed conformation that binds to Munc18-1 and is incompatible with SNARE complex formation, suggesting that syntaxin-1 undergoes a large conformational change during exocytosis.
Parlati, F. et al. Rapid and efficient fusion of phospholipid vesicles by the alpha-helical core of a SNARE complex in the absence of an N-terminal regulatory domain. Proc. Natl. Acad. Sci. USA 96, 12565–12570 (1999).
Melia, T.J. et al. Regulation of membrane fusion by the membrane-proximal coil of the t-SNARE during zippering of SNAREpins. J. Cell Biol. 158, 929–940 (2002).
Pobbati, A.V., Stein, A. & Fasshauer, D. N- to C-terminal SNARE complex assembly promotes rapid membrane fusion. Science 313, 673–676 (2006).
Zhang, F., Chen, Y., Kweon, D.H., Kim, C.S. & Shin, Y.K. The four-helix bundle of the neuronal target membrane SNARE complex is neither disordered in the middle nor uncoiled at the C-terminal region. J. Biol. Chem. 277, 24294–24298 (2002).
Guan, R., Dai, H. & Rizo, J. Binding of the Munc13–1 MUN domain to membrane-anchored SNARE complexes. Biochemistry 47, 1474–1481 (2008).
Weninger, K., Bowen, M.E., Choi, U.B., Chu, S. & Brunger, A.T. Accessory proteins stabilize the acceptor complex for synaptobrevin, the 1:1 syntaxin/SNAP-25 complex. Structure 16, 308–320 (2008).
Xu, T. et al. Inhibition of SNARE complex assembly differentially affects kinetic components of exocytosis. Cell 99, 713–722 (1999).
Sorensen, J.B. et al. Sequential N- to C-terminal SNARE complex assembly drives priming and fusion of secretory vesicles. EMBO J. 25, 955–966 (2006).
Chen, X. et al. Three-dimensional structure of the complexin/SNARE complex. Neuron 33, 397–409 (2002). A combination of NMR spectroscopy and X-ray crystallography reveals the interaction between complexin-I and the SNARE complex at atomic resolution and suggests that complexin binding stabilizes the C-terminus of the SNARE complex.
Li, F. et al. Energetics and dynamics of SNAREpin folding across lipid bilayers. Nat. Struct. Mol. Biol. 14, 890–896 (2007).
Cohen, F.S. & Melikyan, G.B. The energetics of membrane fusion from binding, through hemifusion, pore formation, and pore enlargement. J. Membr. Biol. 199, 1–14 (2004).
McNew, J.A. et al. Close is not enough: SNARE-dependent membrane fusion requires an active mechanism that transduces force to membrane anchors. J. Cell Biol. 150, 105–117 (2000).
Langosch, D. et al. Peptide mimics of SNARE transmembrane segments drive membrane fusion depending on their conformational plasticity. J. Mol. Biol. 311, 709–721 (2001).
Kweon, D.H., Kim, C.S. & Shin, Y.K. Regulation of neuronal SNARE assembly by the membrane. Nat. Struct. Biol. 10, 440–447 (2003).
Chen, X. et al. SNARE-mediated lipid mixing depends on the physical state of the vesicles. Biophys. J. 90, 2062–2074 (2006).
Dennison, S.M., Bowen, M.E., Brunger, A.T. & Lentz, B.R. Neuronal SNAREs do not trigger fusion between synthetic membranes but do promote PEG-mediated membrane fusion. Biophys. J. 90, 1661–1675 (2006).
Bowen, M.E., Weninger, K., Brunger, A.T. & Chu, S. Single molecule observation of liposome-bilayer fusion thermally induced by soluble N-ethyl maleimide sensitive-factor attachment protein receptors (SNAREs). Biophys. J. 87, 3569–3584 (2004). This paper shows that multiple SNARE complexes bridging a vesicle and a planar membrane can form without inducing membrane fusion, suggesting that SNARE complex formation is not sufficient for membrane fusion.
Kesavan, J., Borisovska, M. & Bruns, D. v-SNARE actions during Ca2+-triggered exocytosis. Cell 131, 351–363 (2007).
McNew, J.A., Weber, T., Engelman, D.M., Sollner, T.H. & Rothman, J.E. The length of the flexible SNAREpin juxtamembrane region is a critical determinant of SNARE-dependent fusion. Mol. Cell 4, 415–421 (1999).
Kim, C.S., Kweon, D.H. & Shin, Y.K. Membrane topologies of neuronal SNARE folding intermediates. Biochemistry 41, 10928–10933 (2002).
Deak, F., Shin, O.H., Kavalali, E.T. & Sudhof, T.C. Structural determinants of synaptobrevin 2 function in synaptic vesicle fusion. J. Neurosci. 26, 6668–6676 (2006).
Verhage, M. et al. Synaptic assembly of the brain in the absence of neurotransmitter secretion. Science 287, 864–869 (2000). This paper shows that neurotransmitter secretion from synaptic vesicles is completely abrogated in Munc18-1 knockout mice and that the brain can still assemble in the absence of neurotransmitter release.
Hata, Y., Slaughter, C.A. & Sudhof, T.C. Synaptic vesicle fusion complex contains unc-18 homologue bound to syntaxin. Nature 366, 347–351 (1993).
Yang, B., Steegmaier, M. & Gonzalez, L.C. Jr. & Scheller, R.H. nSec1 binds a closed conformation of syntaxin1A. J. Cell Biol. 148, 247–252 (2000).
Misura, K.M., Scheller, R.H. & Weis, W.I. Three-dimensional structure of the neuronal-Sec1-syntaxin 1a complex. Nature 404, 355–362 (2000). The crystal structure of syntaxin-1 bound to Munc18-1 is described, providing the first atomic resolution structure of a SM protein and revealing how Munc18-1 binds to the syntaxin-1 closed conformation.
Carr, C.M., Grote, E., Munson, M., Hughson, F.M. & Novick, P.J. Sec1p binds to SNARE complexes and concentrates at sites of secretion. J. Cell Biol. 146, 333–344 (1999). Sec1p is shown to bind to its cognate SNARE complex, providing the first evidence for formation of the SM protein–SNARE complex assemblies that may form the core of intracellular membrane fusion machineries.
Togneri, J., Cheng, Y.S., Munson, M., Hughson, F.M. & Carr, C.M. Specific SNARE complex binding mode of the Sec1/Munc-18 protein, Sec1p. Proc. Natl. Acad. Sci. USA 103, 17730–17735 (2006).
Nicholson, K.L. et al. Regulation of SNARE complex assembly by an N-terminal domain of the t-SNARE Sso1p. Nat. Struct. Biol. 5, 793–802 (1998).
Dulubova, I., Yamaguchi, T., Wang, Y., Sudhof, T.C. & Rizo, J. Vam3p structure reveals conserved and divergent properties of syntaxins. Nat. Struct. Biol. 8, 258–264 (2001).
Yamaguchi, T. et al. Sly1 binds to Golgi and ER syntaxins via a conserved N-terminal peptide motif. Dev. Cell 2, 295–305 (2002).
Dulubova, I. et al. How Tlg2p/Syntaxin 16 “snares” Vps45. EMBO J. 21, 3620–3631 (2002).
Bracher, A. & Weissenhorn, W. Structural basis for the Golgi membrane recruitment of Sly1p by Sed5p. EMBO J. 21, 6114–6124 (2002).
Dulubova, I. et al. Munc18–1 binds directly to the neuronal SNARE complex. Proc. Natl. Acad. Sci. USA 104, 2697–2702 (2007).
Shen, J., Tareste, D.C., Paumet, F., Rothman, J.E. & Melia, T.J. Selective activation of cognate SNAREpins by Sec1/Munc18 proteins. Cell 128, 183–195 (2007).
Khvotchev, M. et al. Dual modes of Munc18–1/SNARE interactions are coupled by functionally critical binding to syntaxin-1 N terminus. J. Neurosci. 27, 12147–12155 (2007).
Burkhardt, P., Hattendorf, D.A., Weis, W.I. & Fasshauer, D. Munc18a controls SNARE assembly through its interaction with the syntaxin N-peptide. EMBO J. 27, 923–933 (2008).
Medine, C.N., Rickman, C., Chamberlain, L.H. & Duncan, R.R. Munc18–1 prevents the formation of ectopic SNARE complexes in living cells. J. Cell Sci. 120, 4407–4415 (2007).
Zilly, F.E., Sorensen, J.B., Jahn, R. & Lang, T. Munc18-bound syntaxin readily forms snare complexes with synaptobrevin in native plasma membranes. PLoS Biol. 4, e330 (2006).
Gulyas-Kovacs, A. et al. Munc18–1: sequential interactions with the fusion machinery stimulate vesicle docking and priming. J. Neurosci. 27, 8676–8686 (2007).
Grote, E., Carr, C.M. & Novick, P.J. Ordering the final events in yeast exocytosis. J. Cell Biol. 151, 439–452 (2000).
Tareste, D., Shen, J., Melia, T.J. & Rothman, J.E. SNAREpin/Munc18 promotes adhesion and fusion of large vesicles to giant membranes. Proc. Natl. Acad. Sci. USA 105, 2380–2385 (2008).
Rizo, J. & Sudhof, T.C. C2-domains, structure and function of a universal Ca2+-binding domain. J. Biol. Chem. 273, 15879–15882 (1998).
Lu, J. et al. Structural basis for a Munc13–1 homodimer to Munc13–1/RIM heterodimer switch. PLoS Biol. 4, e192 (2006).
Dai, H. et al. Crystal structure of the RIM2 C2A-domain at 1.4 Å resolution. Biochemistry 44, 13533–13542 (2005).
Guan, R. et al. Crystal structure of the RIM1α C2B domain at 1.7 Å resolution. Biochemistry 46, 8988–8998 (2007).
Basu, J. et al. A minimal domain responsible for Munc13 activity. Nat. Struct. Mol. Biol. 12, 1017–1018 (2005).
Richmond, J.E., Weimer, R.M. & Jorgensen, E.M. An open form of syntaxin bypasses the requirement for UNC-13 in vesicle priming. Nature 412, 338–341 (2001). Rescue experiments suggest that the function of Unc13/Munc13s in vesicle priming is at least in part to facilitate opening of the syntaxin-1 conformation.
Rosenmund, C. et al. Differential control of vesicle priming and short-term plasticity by Munc13 isoforms. Neuron 33, 411–424 (2002). This paper clearly establishes a role for Munc13s in short-term presynaptic plasticity.
Rhee, J.S. et al. Beta phorbol ester- and diacylglycerol-induced augmentation of transmitter release is mediated by Munc13s and not by PKCs. Cell 108, 121–133 (2002).
Basu, J., Betz, A., Brose, N. & Rosenmund, C. Munc13–1 C1 domain activation lowers the energy barrier for synaptic vesicle fusion. J. Neurosci. 27, 1200–1210 (2007).
Junge, H.J. et al. Calmodulin and Munc13 form a Ca2+ sensor/effector complex that controls short-term synaptic plasticity. Cell 118, 389–401 (2004).
Kaeser, P.S. & Sudhof, T.C. RIM function in short- and long-term synaptic plasticity. Biochem. Soc. Trans. 33, 1345–1349 (2005).
Koushika, S.P. et al. A post-docking role for active zone protein Rim. Nat. Neurosci. 4, 997–1005 (2001).
Schoch, S. et al. RIM1α forms a protein scaffold for regulating neurotransmitter release at the active zone. Nature 415, 321–326 (2002). Biochemical and functional experiments suggest that RIM1α provides a scaffold to organize the active zone and plays a role in short-term presynaptic plasticity in addition to controlling vesicle priming.
Betz, A. et al. Functional interaction of the active zone proteins Munc13–1 and RIM1 in synaptic vesicle priming. Neuron 30, 183–196 (2001).
Dulubova, I. et al. A Munc13/RIM/Rab3 tripartite complex: from priming to plasticity? EMBO J. 24, 2839–2850 (2005).
Castillo, P.E., Schoch, S., Schmitz, F., Sudhof, T.C. & Malenka, R.C. RIM1α is required for presynaptic long-term potentiation. Nature 415, 327–330 (2002).
Castillo, P.E. et al. Rab3A is essential for mossy fibre long-term potentiation in the hippocampus. Nature 388, 590–593 (1997).
Calakos, N., Schoch, S., Südhof, T.C. & Malenka, R.C. Multiple roles for the active zone protein RIM1α in late stages of neurotransmitter release. Neuron 42, 889–896 (2004).
Powell, C.M. et al. The presynaptic active zone protein RIM1α is critical for normal learning and memory. Neuron 42, 143–153 (2004).
Ostermeier, C. & Brunger, A.T. Structural basis of Rab effector specificity: crystal structure of the small G protein Rab3A complexed with the effector domain of rabphilin-3A. Cell 96, 363–374 (1999).
Sutton, R.B., Davletov, B.A., Berghuis, A.M., Sudhof, T.C. & Sprang, S.R. Structure of the first C2 domain of synaptotagmin I: a novel Ca2+/phospholipid-binding fold. Cell 80, 929–938 (1995).
Ubach, J., Zhang, X., Shao, X., Sudhof, T.C. & Rizo, J. Ca2+ binding to synaptotagmin: how many Ca2+ ions bind to the tip of a C2-domain? EMBO J. 17, 3921–3930 (1998).
Shao, X., Fernandez, I., Sudhof, T.C. & Rizo, J. Solution structures of the Ca2+-free and Ca2+-bound C2A domain of synaptotagmin I: does Ca2+ induce a conformational change? Biochemistry 37, 16106–16115 (1998).
Fernandez, I. et al. Three-dimensional structure of the synaptotagmin 1 C2B-domain: synaptotagmin 1 as a phospholipid binding machine. Neuron 32, 1057–1069 (2001).
Chapman, E.R. & Davis, A.F. Direct interaction of a Ca2+-binding loop of synaptotagmin with lipid bilayers. J. Biol. Chem. 273, 13995–14001 (1998).
Zhang, X., Rizo, J. & Sudhof, T.C. Mechanism of phospholipid binding by the C2A-domain of synaptotagmin I. Biochemistry 37, 12395–12403 (1998).
Fernandez-Chacon, R. et al. Synaptotagmin I functions as a calcium regulator of release probability. Nature 410, 41–49 (2001). A point mutation in synaptotagmin-1 causes a parallel decrease in its apparent Ca2+ affinity and in the Ca2+ sensitivity of release, leaving little doubt for the proposed role of synaptotagmin-1 as a Ca2+ sensor in release.
Rhee, J.S. et al. Augmenting neurotransmitter release by enhancing the apparent Ca2+ affinity of synaptotagmin 1. Proc. Natl. Acad. Sci. USA 102, 18664–18669 (2005).
Robinson, I.M., Ranjan, R. & Schwarz, T.L. Synaptotagmins I and IV promote transmitter release independently of Ca2+ binding in the C2A domain. Nature 418, 336–340 (2002).
Mackler, J.M., Drummond, J.A., Loewen, C.A., Robinson, I.M. & Reist, N.E. The C2B Ca2+-binding motif of synaptotagmin is required for synaptic transmission in vivo . Nature 418, 340–344 (2002). A mutation in the Ca2+ binding sites of the synaptotagmin-1 C 2 B domain causes a dramatic impairment in neurotransmitter release, showing the crucial importance of Ca2+ binding to the C 2 B domain for release.
Arac, D. et al. Close membrane-membrane proximity induced by Ca(2+)-dependent multivalent binding of synaptotagmin-1 to phospholipids. Nat. Struct. Mol. Biol. 13, 209–217 (2006). The observation of simultaneous binding of synaptotagmin-1 to two membranes suggests that this Ca2+ sensor functions by bringing the vesicle and plasma membranes into close proximity, similar to the SNAREs but in a Ca2+-dependent manner.
Arac, D., Murphy, T. & Rizo, J. Facile detection of protein-protein interactions by one-dimensional NMR spectroscopy. Biochemistry 42, 2774–2780 (2003).
Bhalla, A., Chicka, M.C., Tucker, W.C. & Chapman, E.R. Ca2+-synaptotagmin directly regulates t-SNARE function during reconstituted membrane fusion. Nat. Struct. Mol. Biol. 13, 323–330 (2006).
Dai, H., Shen, N., Arac, D. & Rizo, J.A. Quaternary SNARE-synaptotagmin-Ca(2+)-phospholipid complex in neurotransmitter release. J. Mol. Biol. 367, 848–863 (2007).
Bowen, M.E., Weninger, K., Ernst, J., Chu, S. & Brunger, A.T. Single-molecule studies of synaptotagmin and complexin binding to the SNARE complex. Biophys. J. 89, 690–702 (2005).
Zimmerberg, J., Akimov, S.A. & Frolov, V. Synaptotagmin: fusogenic role for calcium sensor? Nat. Struct. Mol. Biol. 13, 301–303 (2006).
Lynch, K.L. et al. Synaptotagmin C2A loop 2 mediates Ca2+-dependent SNARE interactions essential for Ca2+-triggered vesicle exocytosis. Mol. Biol. Cell 18, 4957–4968 (2007).
Martens, S., Kozlov, M.M. & McMahon, H.T. How synaptotagmin promotes membrane fusion. Science 316, 1205–1208 (2007).
Tucker, W.C., Weber, T. & Chapman, E.R. Reconstitution of Ca2+-regulated membrane fusion by synaptotagmin and SNAREs. Science 304, 435–438 (2004).
Stein, A., Radhakrishnan, A., Riedel, D., Fasshauer, D. & Jahn, R. Synaptotagmin activates membrane fusion through a Ca2+-dependent trans interaction with phospholipids. Nat. Struct. Mol. Biol. 14, 904–911 (2007).
Pabst, S. et al. Selective interaction of complexin with the neuronal SNARE complex. Determination of the binding regions. J. Biol. Chem. 275, 19808–19818 (2000).
McMahon, H.T., Missler, M., Li, C. & Sudhof, T.C. Complexins: cytosolic proteins that regulate SNAP receptor function. Cell 83, 111–119 (1995).
Giraudo, C.G., Eng, W.S., Melia, T.J. & Rothman, J.E. A clamping mechanism involved in SNARE-dependent exocytosis. Science 313, 676–680 (2006).
Schaub, J.R., Lu, X., Doneske, B., Shin, Y.K. & McNew, J.A. Hemifusion arrest by complexin is relieved by Ca2+–synaptotagmin I. Nat. Struct. Mol. Biol. 13, 748–750 (2006).
Tang, J. et al. A complexin/synaptotagmin 1 switch controls fast synaptic vesicle exocytosis. Cell 126, 1175–1187 (2006).
Huntwork, S. & Littleton, J.T. A complexin fusion clamp regulates spontaneous neurotransmitter release and synaptic growth. Nat. Neurosci. 10, 1235–1237 (2007).
Reim, K. et al. Complexins regulate a late step in Ca2+-dependent neurotransmitter release. Cell 104, 71–81 (2001). Complexins I and II are shown to play a critical active role in the Ca2+-triggered step of neurotransmitter release.
Reim, K. et al. Structurally and functionally unique complexins at retinal ribbon synapses. J. Cell Biol. 169, 669–680 (2005).
Roggero, C.M. et al. Complexin/synaptotagmin interplay controls acrosomal exocytosis. J. Biol. Chem. 282, 26335–26343 (2007).
Xue, M. et al. Distinct domains of complexin I differentially regulate neurotransmitter release. Nat. Struct. Mol. Biol. 14, 949–958 (2007).
Fuson, K.L., Montes, M., Robert, J.J. & Sutton, R.B. Structure of human synaptotagmin 1 C2AB in the absence of Ca2+ reveals a novel domain association. Biochemistry 46, 13041–13048 (2007).
Weimer, R.M. et al. UNC-13 and UNC-10/rim localize synaptic vesicles to specific membrane domains. J. Neurosci. 26, 8040–8047 (2006).
Voets, T. et al. Munc18–1 promotes large dense-core vesicle docking. Neuron 31, 581–591 (2001).
de Wit, H., Cornelisse, L.N., Toonen, R.F. & Verhage, M. Docking of secretory vesicles is syntaxin dependent. PLoS ONE 1, e126 (2006).
Hammarlund, M., Palfreyman, M.T., Watanabe, S., Olsen, S. & Jorgensen, E.M. Open syntaxin docks synaptic vesicles. PLoS Biol. 5, e198 (2007).
Acknowledgements
Work in the authors' laboratories is supported by US National Institutes of Health grants NS40944 and NS37200 (to J.R.) and NS050655 and NS051262 (to C.R.).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Rizo, J., Rosenmund, C. Synaptic vesicle fusion. Nat Struct Mol Biol 15, 665–674 (2008). https://doi.org/10.1038/nsmb.1450
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.1450
This article is cited by
-
DYRK1-mediated phosphorylation of endocytic components is required for extracellular lumen expansion in ascidian notochord
Biological Research (2023)
-
Conformational change of Syntaxin-3b in regulating SNARE complex assembly in the ribbon synapses
Scientific Reports (2022)
-
Transcriptomic profiling on localized gastric cancer identified CPLX1 as a gene promoting malignant phenotype of gastric cancer and a predictor of recurrence after surgery and subsequent chemotherapy
Journal of Gastroenterology (2022)
-
Early embryogenesis and organogenesis in the annelid Owenia fusiformis
EvoDevo (2021)
-
CAPS1 is involved in hippocampal synaptic plasticity and hippocampus-associated learning
Scientific Reports (2021)