Abstract
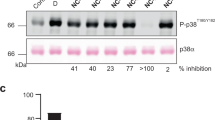
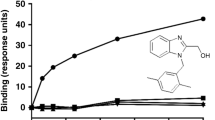
The p38 MAP kinase plays a crucial role in regulating the production of proinflammatory cytokines, such as tumor necrosis factor and interleukin-1. Blocking this kinase may offer an effective therapy for treating many inflammatory diseases. Here we report a new allosteric binding site for a diaryl urea class of highly potent and selective inhibitors against human p38 MAP kinase. The formation of this binding site requires a large conformational change not observed previously for any of the protein Ser/Thr kinases. This change is in the highly conserved Asp-Phe-Gly motif within the active site of the kinase. Solution studies demonstrate that this class of compounds has slow binding kinetics, consistent with the requirement for conformational change. Improving interactions in this allosteric pocket, as well as establishing binding interactions in the ATP pocket, enhanced the affinity of the inhibitors by 12,000-fold. One of the most potent compounds in this series, BIRB 796, has picomolar affinity for the kinase and low nanomolar inhibitory activity in cell culture.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Feldmann, M., Brennan, F.M., & Maini, R.N. Annu. Rev. Immunol. 14, 397–440 (1996).
Weinblatt, M.E. et al. N. Engl. J. Med. 340, 253–259 (1999).
Jarvis, B. & Faulds, D. Drugs 57, 945–966 (1999).
Prichett, W., Hand, A., Sheilds, J., & Dunnington, D. J. Inflamm. 45, 97–105 (1995).
Lee, J.C. et al. Nature 372, 739–746 (1994).
Lee, J.C. et al. Ann. NY Acad. Sci. 696, 149–170 (1993).
Dumas, J. et al. Bioorg. Med. Chem. Lett. 10, 2047–2050 (2000).
Tong, L. et al. Nature Struct. Biol. 4, 311–316 (1997).
Frantz, B. et al. Biochemistry 37, 13846–13853 (1998).
Young, P.R. et al. J. Biol. Chem. 272, 12116–12121 (1997).
Morelock, M.M., Pargellis, C.A., Graham, E.T., Lamarre, D. & Jung, G. J. Med. Chem. 38, 1751–1761 (1995).
Pargellis, C.A. et al. Biochemistry 33, 12527–12534 (1994).
Kaplan, A.P. & Bartlett, P.A. Biochemistry 30, 8165–8170 (1991).
Cha, S. Biochem. Pharmacol. 24, 2177–2185 (1975).
Hubbard, S.R., Wei, L., Ellis, L., & Hendrickson, W.A. Nature 372, 746–754 (1994).
Hubbard, S.R., Mohammadi, M., & Schlessinger, J. J. Biol. Chem. 273, 11987–11990 (1998).
Schindler, T. et al. Science 289, 1938–1942 (2000).
Hanks, S.K., Quinn, A.M. & Hunter, T. Science 241, 42–52 (1988).
Branger, J. et al. J. Immunol. In the press (2002).
Cirillo, P.F., Gilmore, T.A., Hickey, E., Regan, J. & Zhang, L.H. Aromatic Heterocyclic Compounds as Antiinflammatory Agents. World Patent Application WO-00043384 (2000).
Pav, S. et al. Protein Sci. 6, 242–245 (1997).
Brünger, A.T. et al. Acta Crystallogr. D 54, 905–921 (1998).
Jones, T.A., Zou, J.Y., Cowan, S.W. & Kjeldgaard, M. Acta Crystallogr. A 47, 110–119 (1991).
Raingeaud, J., Whitmarsh, A.J., Barrett, T., Derijard, B. & Davis, R.J. Mol. Cell. Biol. 16, 1247–1255 (1996).
Han, J. et al. J. Biol. Chem. 271, 2886–2891 (1996).
Lozanski, G., Ballou, S.P., & Kushner, I. J. Rheumatol. 19, 921–926 (1992).
Carson, M. J. Mol. Graph. 5, 103–106 (1987).
Evans, S.V. J. Mol. Graph. 11, 134–138 (1993).
Nicholls, A., Sharp, K A., & Honig, B. Proteins 11, 281–296 (1991).
Acknowledgements
We are indebted to the following people: J. Pelletier, A. Proto and M. Basso for assistance with biological assays; A. Swinamer, B. Klaus and M. Moriak for the preparation of various analogs of BIRB 796; M. Morelock for statistical analysis; L. Rondano for graphics support; and R. Winquist and P. Farina for useful discussions. We acknowledge Glaxo SmithKline Pharmaceuticals for the generous gift of SK&F 86002.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Pargellis, C., Tong, L., Churchill, L. et al. Inhibition of p38 MAP kinase by utilizing a novel allosteric binding site. Nat Struct Mol Biol 9, 268–272 (2002). https://doi.org/10.1038/nsb770
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsb770
This article is cited by
-
Characterization of p38α autophosphorylation inhibitors that target the non-canonical activation pathway
Nature Communications (2023)
-
In vitro and in silico studies of 7′′,8′′-buddlenol D anti-inflammatory lignans from Carallia brachiata as p38 MAP kinase inhibitors
Scientific Reports (2023)
-
Investigation of structure–activity relationship: In silico studies of [1, 2, 4]triazolo[4, 3-a]pyridine ureas as P38 kinase inhibitors
Structural Chemistry (2023)
-
p38 MAPK Is a Major Regulator of Amyloid Beta-Induced IL-6 Expression in Human Microglia
Molecular Neurobiology (2022)
-
Activated microglia–induced neuroinflammatory cytokines lead to photoreceptor apoptosis in Aβ-injected mice
Journal of Molecular Medicine (2021)