Abstract
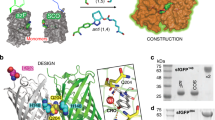
The transfer of electrons between proteins is an essential step in biological energy production. Two protein redox partners are often artificially crosslinked to investigate the poorly understood mechanism by which they interact. To better understand the effect of crosslinking on electron transfer rates, we have constructed dimers of azurin by crosslinking the monomers. The measured electron exchange rates, combined with crystal structures of the dimers, demonstrate that the length of the linker can have a dramatic effect on the structure of the dimer and the electron transfer rate. The presence of ordered water molecules in the protein–protein interface may considerably influence the electronic coupling between redox centers.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Bendall, D.S. In Protein electron transfer (ed. Bendall, D.S.) 43–68 (Bios Scientific Publishers, Oxford; 1996).
Castro, G., Boswell, C.A. & Northrup, S.H. J. Biomol. Struct. Dyn. 16, 413–424 (1998).
Liang, Z.X., Nocek, J.M., Kurnikov, I.V., Beratan, D.N. & Hoffman, B.M. J. Am. Chem. Soc. 122, 3552–3553 (2000).
Nocek, J.M. et al. Chem. Rev. 96, 2459–2489 (1996).
Ubbink, M. & Bendall, D.S. Biochemistry 36, 6326–6335 (1997).
Qin, L. & Kostic, N.M. Biochemistry 32, 6073–6080 (1993).
Hippler, M., Drepper, F., Haehnel, W. & Rochaix, J.D. Proc. Natl. Acad. Sci. USA 95, 7339–7344 (1998).
Malatesta, F. et al. Biochem. J. 315, 909–916 (1996).
Peerey, L.M., Brothers, H.M., Hazzard, J.T., Tollin, G. & Kostic, N.M. Biochemistry 30, 9297–9304 (1991).
Nar, H., Messerschmidt, A., Huber, R., van de Kamp, M. & Canters, G.W. J. Mol. Biol. 221, 765–772 (1991).
Nar, H., Messerschmidt, A., Huber, R., van de Kamp, M. & Canters, G.W. J. Mol. Biol. 218, 427–447 (1991).
Mikkelsen, K.V., Skov, L.K., Nar, H. & Farver, O. Proc. Natl. Acad. Sci. USA 90, 5443–5445 (1993).
van de Kamp, M., Floris, R., Hali, F.C. & Canters, G.W. J. Am. Chem. Soc. 112, 907–908 (1990).
van Amsterdam, I.M.C. et al. Chem. Eur. J. 7, 2398–2406 (2001).
Canters, G.W., Hill, H.A.O., Kitchen, N.A. & Adman, E.T. J. Magn. Reson. 57, 1–23 (1984).
Bain, A.D. & Duns, G.J. Can. J. Chem. 74, 819–824 (1996).
Page, C.C., Moser, C.C., Chen, X. & Dutton, P.L. Nature 402, 47–52 (1999).
Beratan, D.N. & Onuchic, J.N. In Biological electron transfer. (ed. Bendall, D.S.) 23–42 (Bios Scientific Publishers, Oxford; 1996).
Gray, H.B., Malmström, B.G. & Williams, R.J.P. J. Biol. Inorg. Chem. 5, 551–559 (2000).
Kurnikov, I.V., Wenzel, W. & Beratan, D.N. HARLEM http://www.kurnikov.org/harlem_main.html (Biomolecular Modeling package, University of Pittsburgh; 2000)
Ponce, A., Gray, H.B. & Winkler, J.R. J. Am. Chem. Soc. 122, 8187–8191 (2000).
Tezcan, F.A., Crane, B.R., Winkler, J.R. & Gray, H.B. Proc. Natl. Acad. Sci. USA 98, 5002–5006 (2001).
Navaza, J. Acta Crystallogr. A 50, 157–163 (1994).
Brünger, A.T. et al. Acta Crystallogr. D 54, 905–921 (1998).
Jones, T.A., Zhou, J.Y., Cowan, S.W. & Kjeldgaard, M. Acta Crystallogr. A 47, 110–119 (1991).
Hubbard, S.J. & Thornton, J.M. NACCESS (Department of Biochemistry and Molecular Biology, University College London; 1993).
Kraulis, P. J. J. Appl. Crystallogr. 24, 946–950 (1991).
Esnouf, R. M. J. Mol. Graph. Model. 15, 132–& (1997).
Merritt, E. A. & Bacon, D. J. Methods Enzymol. 277, 505–524 (1997).
DeLano, W.L. The PyMOL Molecular Graphics System. http://pymol.sourceforge.net/ (DeLano Scientific, San Carlos, California; 2001)
Acknowledgements
We thank M.Ph. Verbeet and L.J.C. Jeuken for their help with the mutagenesis, C. Erkelens for his technical assistance during NMR measurements, A.D. Bain for his advice in using the MEX/MEXICO programs, C.C. Moser and P.L. Dutton for providing the ET Rates software and I.V. Kurnikov for his help in using the HARLEM program. This work is supported in part by grants of the EU, the National Research Council of Italy (to A.Mer., D.C. and G.R.) and the Deutsche Forschungsgemeinschaft (to O.E. and A.Mes.).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
van Amsterdam, I., Ubbink, M., Einsle, O. et al. Dramatic modulation of electron transfer in protein complexes by crosslinking. Nat Struct Mol Biol 9, 48–52 (2002). https://doi.org/10.1038/nsb736
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsb736
This article is cited by
-
Light-driven progesterone production by InP–(M. neoaurum) biohybrid system
Bioresources and Bioprocessing (2022)
-
Long distance electron transfer through the aqueous solution between redox partner proteins
Nature Communications (2018)
-
A further investigation of the cytochrome b 5–cytochrome c complex
JBIC Journal of Biological Inorganic Chemistry (2003)