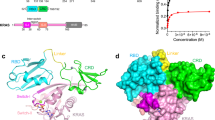
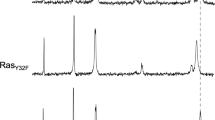
Abstract
Members of the Ras subfamily of small GTP-binding proteins have been shown to be promiscuous towards a variety of putative effector molecules such as the protein kinase c-Raf and the Ral-specif guanine nucleotide exchange factor (Ral-GEF). To address the question of specificity of interactions we have introduced the mutations E30D and K31E into Rap and show biochemically, by X-ray structure analysis and by transfection in vivo that the identical core effector region of Ras and Rap (residues 32–40) is responsible for molecular recognition, but that residues outside this region are responsible for the specificity of the interaction. The major determinant for the switch in specificity is the opposite charge of residue 31—Lys in Rap, Glu in Ras—which creates a favourable complementary interface for the Ras–Raf interaction.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Nassar, N. et al. The 2.2 Å crystal structure of the Ras-binding domain of the serine/threonine kinase c-Raf1 in complex with RaplA and a GTP analogue. Nature 375, 554–560 (1995).
Vijay-Kumar, S., Bugg, C.E. & Cook, W.J. Structure of ubiquitin refined at 1·8 Å resolution. J. Mol. Biol. 194, 531–544 (1987).
Weber, P.L., Brown, S.C. & Muller, L. Sequential 1H NMR assignments and secondary structure identification of human ubiquitin. Biochemistry 26, 7282–7290 (1987).
Di Stefano, D.L. & Wand, A.J. Two-dimensional 1H NMR study of human ubiquitin: a main chain directed assignment and structure analysis. Biochemistry 26, 7272–7281 (1987).
Derrick, J.P. & Wigley, D.B. The third IgG-binding domain from streptococcal protein G. J. Mol. Biol. 243, 906–918 (1994).
Orengo, C.A., Jones, D.T. & Thornton, J.M. Protein superfamilies and domain superfolds. Nature 372, 631–634 (1994).
Tsukihara, T. Structure of the [2Fe-2S] Ferredoxin I from the blue-green Alga Aphanothece sacrum at 2. 2 Å resolution. J. Mol. Biol. 216, 399–410 (1990).
Derrick, J.P. & Wigley, D.B. Crystal structure of a streptococcal protein G domain bound to an Fab fragment. Nature 359, 752–754 (1992).
Lian, L.Y., Barsukov, I.L., Derrick, J.P. & Roberts, G.C.K. Mapping the interactions between streptococcal protein G and the Fab fragment of IgG in solution. Nature Struct. Biol. 1, 355–357 (1994).
Pizon, V., Chardin, P., Lerosey,I., Olofsson, B. & Tavitian, A. Human cDNAs rap1 and rap2 homologous to the Drosophila gene Dras3 encode proteins closely related to ras in the ‘effector’ region. Oncogene 3, 201–204 (1988).
Kawata, M. et al. A novel small molecular weight GTP-binding protein with the same putative effector domain as the ras proteins in bovine brain membranes. J Biol. Chem. 263, 18965–18971 (1988).
Ohmori, T., Kikuchi, A., Yamamoto, K., Kirn, S. & Takai, Y. Small molecular weight GTP-binding proteins in human platelet membranes. J. Biol. Chem. 264, 1877–1881 (1989).
Zhang, X.-f. et al. Normal and oncogenic p21ras proteins bind to the amino-terminal regulatory domain of c-Raf-1. Nature 364, 308–313 (1993).
Hofer, F., Fields, S., Schneider, D. & Martin, G.S. Activated Ras interacts with the Ral guanine nucleotide dissociation stimulator. Proc. Natl. Acad. Sci. USA 91, 11089–11093 (1994).
Kikuchi, A., Demo, S.D., Ye Zhi-Hai Chen, Y.-W. & Williams, L.T. RalGDS family members interact with the effector loop of ras p21. Mol. Cell. Biol. 14, 7483–7491 (1994).
Spaargaren, M. & Bischoff, J.R. Identification of the guanine nucleotide dissociation stimulator for Ral as a putative effector molecule of R-ras, H-ras, K-ras, and Rap. Proc. Natl. Acad. Sci. USA 91, 12609–12613 (1994).
Kitayama, H., Sugimoto, Y., Matsuzaki, T., Ikawa, Y. & Noda, M. A ras-related gene with transformation suppressor activity. Cell 56, 77–84 (1989).
Zhang, K., Noda, M., Vass, W.C., Papageorge, A.G. & Lowy, D.R. Identification of small clusters of divergent amino acids that mediate the opposing effects of ras and Krev-1. Science 249, 162–165 (1990).
Cook, S.J., Rubinfeld, B., Albert, I. & McCormick, F. RapV12 antagonizes Ras-dependent activation of ERK1 and ERK2 by LPA and EGF in Rat-1 fibroblasts. EMBO J. 12, 3475–3485 (1993).
Marshall, M.S. The effector interactions of p21ras. Trends Biochem. Sci. 18, 250–254 (1993).
Herrmann, C., Horn, G., Spaargaren, M. & Wittinghofer, A. Differential interaction of the Ras family GTP-binding proteins H-Ras, Rap1 A and R-Ras with the putative effector molecules Raf-kinase and Ral-guanine nucleotide exchange factor. J. Biol. Chem. 271, 6794–6800 (1996).
Marshall, M.S. et al. Identification of amino acid residues required for ras p21 target activation. Mol. Cell. Biol. 11, 3997–4004 (1991).
Zhang, K. et al. Heterogeneous amino acids in ras and raplA specifying sensitivity to GAP proteins. Science 254, 1630–1634 (1991).
Shirouzu, M. et al. A glutamic acid residue at position 31 of Ras protein is essential to the signal transduction for neurite outgrowth of PC12 cells and the stimulation of GTPase activity by GAPRAS. Oncogene 7, 475–480 (1992).
Pai, E.F. et al. Refined crystal structure of the triphosphate conformation of H-ras p21 at 1.35Å resolution: implications for the mechanism of GTP hydrolysis. EMBO J. 9, 2351–2359 (1990).
Lee, B. & Richards, F.M. The interpretation of protein structures: estimation of static accessibility. J. Mol. Biol. 55, 379–400 (1971).
Programs for Protein Crystallography. Acta Crystallogr. D50 760–763 (1994).
Lawrence, M.C. & Colman, P.M. Shape complementarity at protein/protein interfaces. J. Mol. Biol. 234, 946–950 (1993).
Block, C., Janknecht, R., Herrmann, C., Nassar, N. & Wittinghofer, A. Quantitative structure-activity analysis correlating Ras/Raf interaction in vitro to Raf activation in vivo. Nature Struct. Biol. 3, 244–251 (1996).
Hu, C.-D. et al. Cysteine-rich region of Raf-1 interacts with activator domain of post-translationally modified Ha-Ras. J. Biol. Chem. 270, 30274–30277 (1995).
Drugan, J.K. et al. Ras interaction with two distinct binding domains in Raf-1 may be required for Ras transformation. J. Biol. Chem. 271, 233–237 (1996).
Nur-E-Kamal, M.S.A. & Maruta, H. The role of Gin61 and Glu63 of ras GTPases in their activation by NF1 and ras GAP. Mol. Biol. Cell 3, 1437–1442 (1992).
White, M.A. et al. Multiple Ras functions can contribute to mammalian cell transformation. Cell 80, 1–20 (1995).
Joneson, T., White, M.A., Wigler, M.H. & Bar-Sagi, D. Stimulation of membrane ruffling and MAP kinase activtion by distinct effectors of RAS. Science 271, 810–812 (1996).
Akasaka, K. et al. Differential structural requirements for interaction of Ras protein with its distinct downstream effectors. J. Biol. Chem. 271, 5353–5360 (1996).
Amano, M. et al. Identification of a putative target for Rho as the serine-threonine kinase protein kinase N. Science 271, 648–659 (1996).
Nonaka, H. et al. A downstream target of RHO1 small GTP-binding protein is PKC1, a homolog of protein kinase C, which leads to activation of the MAP kinase cascade in Saccharomyces cerevisiae. EMBO J. 14, 5931–5938 (1995).
Diekmann, D., Nobes, C.D., Burbelo, P.O., Abo, A. & Hall, A. Rac GTPase interacts with GAPs and target proteins through multiple effector sites. EMBO. J. 14, 5297–5305 (1995).
Dorseuil, O., Reibel, L., Bokoch, G.M., Camonis, J. & Gacon, G. The Rac target NADPH oxidase p67phox interacts preferentially with Rac2 rather than Rac1. J. Biol. Chem. 271, 83–88 (1996)
Herrmann, C., Martin, G. & Wittinghofer, A. Quantitative analysis of the complex between p21ras and the Ras-binding domain of the human Raf-1 protein kinase, J. Biol. Chem. 270, 2901–2905 (1995).
Kabsch, W. Automatic processing of rotation diffraction data from crystals of initially unknown symmetry and cell constants. J. Appl. Crystallogr. 26, 795–800 (1993).
Brünger, T.A. XPLOR version 3.1, Manual, Yale Univ.(1993).
Jones, T.A., Zou, J.Y., Cowan, S.W. & Kjeldgaard, M. Improved methods for binding protein models in electron density maps and the location of errors in these models. Acta Crystallogr. A47, 110–119 (1991).
Luzzati, P.V. Traitements statistique des erreurs dans la dètermination des structures cristallines. Acta Crystallogr. 5, 802–810 (1952).
Read, R.J. Improved Fourier coefficients for maps using phases from partial structures with errors. Acta Crystallogr. A42, 140–149 (1986).
Laskowski, R.A., MacArthur, M.W., Moss, D.S. & Thornton, J.M. PROCHECK: a program to check the stereochemical quality of protein structures. J. Appl. Crystallog. 26, 283–291 (1993).
Nicholls, A., Sharp, K.A. & Honig, B. Proteinfolding and association: insights from the interfacial and thermodynamic properties of hydrocarbons. Proteins Struct. Fund. Genet. 11, 281 (1991).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nassar, N., Horn, G., Herrmann, C. et al. Ras/Rap effector specificity determined by charge reversal. Nat Struct Mol Biol 3, 723–729 (1996). https://doi.org/10.1038/nsb0896-723
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nsb0896-723