Abstract
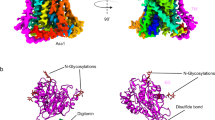
The crystal structure of phenylalanyl-tRNA synthetase from Thermus thermophilus, solved at 2.9 Å resolution, displays (αβ)2 subunit organization. Unexpectedly, both the catalytic α- and the non-catalytic β-subunits comprise the characteristic fold of the class II active-site domains. The αβ heterodimer contains most of the building blocks so far identified in the class II synthetases. The presence of an RNA-binding domain, similiar to that of the U1A spliceosomal protein, in the β-subunit is indicative of structural relationships among different families of RNA-binding proteins. The structure suggests a plausible catalytic mechanism which explains why the primary site of tRIMA aminoacylation is different from that of the other class II enzymes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Eriani, G., Delarue, M., Poch, O., Gangloff, J. & Moras, D. Partition of aminoacyl-tRNA synthetases in two classes on the basis of mutually exclusive sets of sequence motifs. Nature 347, 203–206 (1990).
Rossmann, M.G., Moras, D. & Olsen, K.W. Chemical and biological evolution of a nucleotide-binding protein. Nature 250, 194–199 (1974).
Cusack, S., Berthet-Colominas, C., Hartlein, M., Nassar, N. & Leberman, R. A second class of synthetase structure revealed by X-ray analysis of E.coli seryl-tRNA synthetase at 2.5 Å. Nature 347, 249–255 (1990).
Fraser, T.H. & Rich, A. Amino acids are not initially attached to the same position on tRNA molecules. Proc. natn. Acad. Sci. U.S.A. 72, 3044–3048 (1975).
Sprinzl, M. & Cramer, F. Site of aminoacylation of tRNAs from E. coli with respect to the 2′- or 3′-hydroxyl group of the terminal adenosine. Proc. natn. Acad. Sci. U.S.A. 72, 3049–3053 (1975).
Brunie, S., Zelwer, C. & Risler, J.L. Crystallographic study of the interaction of methionyl-tRNA synthetase from E. coli with ATP. J. molec. Biol. 216, 411–424 (1990).
Rould, M.A., Perona, J.J., Soll, D. & Steitz, T.A. Crystal structure of glutaminyl-tRNA synthetase-tRNAGlu complex. Science 246, 1135–1142 (1989).
Brick, P., Bhat, T.N. & Blow, D.M. Structure of tyrosyl-tRNA synthetase refined at 2.3 Å resolution. J. molec. Biol. 208, 83–98 (1989).
Ruff, M. et al. Class II aminoacyl-tRNA synthetases: crystal structure of yeast aspartyl-tRNA synthetase complexed with tRNAAsp. Science 252 1682–1689 (1991).
Kreutzer, R., Kruft, V., Bobkova, E., Lavrik, O. & Sprinzl, M. Structure of phenylalanyl-tRNA synthetase genes from T. thermophilus and their expression in E. coli. Nucleic Acids Res., 20, 4173–4178 (1992).
Khodyreva, S., Moor, N., Ankilova, V. & Lavrik, O. Phenylalanyl-tRNA synthetase from E. coli MRE-600: analysis of the active site distribution on the enzyme subunits by affinity labelling. Bioch. biophys. Acta 830, 206–212 (1985).
Chernaya, M., Reshetnikova, L., Korolev, S. & Safro, M. Preliminary crystallographic study of the phenylanyl-tRNA synthetase from T. thermophilus HB8. J. molec. Biol. 198, 555–556 (1987).
Reshetnikova, L. et al. Three-dimensional structure of phenylanyl-tRNA synthetase from T. thermophilus HB8 at 0.6-nm resolution. Eur. J. Biochem. 208, 411–417 (1992).
Richardson, J. The anatomy and taxonomy of protein structure. In: Adv. prot. Chem. 34, 167–339 (1981).
Jasin, M., Regan, L. & Schimmel, P. Modular arrangement of functional domains along the sequence of an aminoacyl-tRNA synthetase. Nature 306, 441–447 (1983).
Belrharli, H. et al. Crystal structures at 2.5 Å resolution of seryl-tRNA synthetase complexed with two analogs of seryl adenylate. Science 263, 1432–1436 (1994).
Cavarelli, J. et al. The active site of yeast aspartyl-tRNA synthetase: structural and functional aspects of the aminoacylation reaction. EMBO J. 13 327–337 (1994).
Cavarelli, J., Rees, B., Ruff, M., Thierry, J.-C. & Moras, D. Yeast tRNAAsp recognition by its cognate class II aminoacyl-tRNA synthetase. Nature 362, 181–184 (1993).
Biou, V., Yaremchuk, A., Tukalo, M. & Cusack, S. The 2.9 Å crystal structure of T. thermophilus seryl-tRNA synthetase complexed with tRNASer. Science 263, 1404–1410 (1994).
Schultz, S.C., Shields, G.C. & Steitz, T.A. Crystal structure of a CAP-DNA complex: the DNA is bent by 90°. Science 253, 1001–1007 (1991).
Hynes, T.R. & Fox, R.O. The crystal structure of staphylococcal nuclease refined at 1.7 Å resolution. Proteins 10, 92–105 (1991).
Orengo, C. & Thornton, J. Alpha plus beta folds revisited: some favoured motifs. Structure 1, 105–120 (1993).
Koch, C.A., Anderson, D., Moran, M.F., Ellis, C., Pawson, T. SH2 and SH3 domains: elements that control interactions of cytoplasmic signaling proteins. Science 252, 668–674 (1991).
Nagai, K., Outbridge, C., Jessen, T.H., Li, J. & Evans, P.R. Crystal structure of the RNA-binding domain of the U1 small nuclear ribonucleoprotein A. Nature 348, 515–520 (1990).
Wittekind, M., Gorlach, M., Friedrichs, M., Dreyfuss, G. & Mueller, L. 1H, 13C and 15N NMR assignments and global folding pattern of the RNA-binding domain of the human hnRNP C proteins. Biochemistry 31, 6254–6265 (1992).
Burd, C.G. & Dreyfuss, G. Conserved structures and diversity of functions of RNA-binding proteins. Science 265, 615–621 (1994).
Oubridge, C., Ito, N., Evans, P.R., Teo, C.-H. & Nagai, K. Crystal structure at 1.92 Å resolution of the RNA-binding domain of the U1A spliceosomal protein complexed with an RNA hairpin. Nature 372, 432–438 (1994).
Ibba, M., Kast, P. & Hennecke, H. Substrate specificity is determined by amino acid pocket size in E. coli phenylalanyl-tRNA synthetase. Biochemistry, 33, 7107–7112 (1994).
Eriani, G., Cavarelli, J., Martin, F., Dirheimer, G., Moras, D. & Gangloff, J. Role of dimerization in yeast aspartyl-tRNA synthetase and importance of class II invariant proline. Proc. natn. Acad. Sci. U.S.A. 90, 10816–10820 (1993).
Sanny, A., Walter, P., Boulanger, Y., Ebel, J.-P. & Fasiolo, F. Evolution of aminoacyl-tRNA synthetase quaternary structure and activity: Saccharomyces cerevisae mitochondrial phenylalanyl-tRNA synthetase. Proc. natn. Acad. Sci. U.S.A. 88, 8387–8391 (1991).
Delarue, M., Poterszman, A., Nikonov, S., Garber, M., Moras, D. & Thierry, J.C. Crystal structure of a procaryotic aspartyl-tRNA synthetase. EMBO J. 13, 3219–3229 (1994).
Cusack, S. Sequence, structure and evolutionary relationships between class II aminoacyl-tRNA synthetases: an update. Biochimie 75, 1077–1081 (1993).
Webster, T.A., Gibson, B.W., Keng, T., Biemann, K. & Schimmel, P. Primary structures of both subunits of Escherichia coli glycyl-tRNA synthetase. J biol. Chem. 256, 10637–10641 (1983).
Chou, P.Y. & Fasman, G.D. Prediction of the secondary structure of proteins from their amino acid sequence in Adv. Enzymol., 47, 45–148 (1978).
Wilson, K.P., Shewchuk, L.M., Brennan, R.G., Otsuka, A.J. & Matthews, B.W. Escherichia coli biotin holoenzyme synthetase/bio repressor crystal structure delineates the biotin- and DNA-binding domains. Proc. natn. Acad. Sci. U.S.A. 89, 9257–9261 (1992).
Artymiuk, P.J., Rice, D.W., Poirette, A.R., Willet, P. A tale of two synthetases. Nature struct. Biol. 1, 758–760 (1994).
Leslie, A.G.W. . in Joint CCP4 and EFS-EACMB Newsletter on Protein Crystallography No. 26 (Daresbury Laboratory, Warrington UK, 1992).
CCP4: A suite of pro grams for protein crystallography (SERC collaborative computing project No. 4, Daresbury Laboratory, Warrington UK, 1979).
Otwinowski, Z. In: Isomorphous Replacement and Anomalous scattering 80–86 (Daresbury Laboratory, Warrington UK, 1991).
Wang, B.-C. Resolution of phase ambiguity in macromolecular crystallography. Meths. Enzymol. 115, 90–112 (1985).
Leslie, A.G.W. A reciprocal-space method for calculating a molecular envelope using the algorithm of B.C. Wang. Acta crystallogr. A 43, 134–135 (1987).
Rould, M.A., Perona, J.J. & Steitz, T.A. Improving multiple isomorphous replacement phasing by heavy-atom refinement using solvent-flattened phases. Acta crystallogr. A 48, 751–756 (1992).
Cura, V., Krishnaswamy, S. & Podjarny, A.D. Heavy-atom refinement against solvent-flattened phases. Acta crystallogr. A 48, 756–764 (1992).
Zhang, K.Y.J. & Main, P. The use of Sayre's equation with solvent flattening and histogram matching for phase extenuation and refinement of protein structures. Acta crystallogr. A 46, 377–381 (1990).
Mosyak, L. & Safro, M. Phenylalanyl-tRNA synthetase from T. thermophilus has four antiparallel folds of which only two are catalytically active. Biochimie 75, 1091–1098 (1993).
Jones, T.A. A graphics model building and refinement system for macromolecules. J appl. crystallogr. 11, 268–272 (1978).
Jones, T.A., Zou, J.-Y., Cowan, S.W. & Kjeldgaard, M. Improved methods for building protein models in electron density maps and the location of errors in these maps. Acta crystallogr. A 47. 110–119 (1991).
Read, R.J., Fourier coefficients for maps using phases from partial structures with errors. Acta crystallogr. A 42, 140–149 (1986).
Brünger, A.T., Kuriyan, J. & Karplus, M., Crystallographic, R factor refinement by molecular dynamics. Science 235, 458–460 (1987).
Brünger, A.T. X-PLOR Version 3.1 Manual (Yale Univ., New Haven, 1993).
Hodel, A., Kim, S.-H. & Brünger, A.T. Model bias in macromolecular crystal structures. Acta crystallogr. A 48, 851–858 (1992).
Laskowski, R.A., MacArthur, M.W., Moss, D.S. & Thornton, J.M. PROCHECK: a program to check the stereochemical quality of protein structures. J. appl. crystallogr. 26, 283–291 (1993).
Brünger, A.T. Free R value: a novel statistical quantity for assessing the accuracy of crystal structures. Nature 335, 472–475 (1992).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mosyak, L., Reshetnikova, L., Goldgur, Y. et al. Structure of phenylalanyl-tRNA synthetase from Thermus thermophilus. Nat Struct Mol Biol 2, 537–547 (1995). https://doi.org/10.1038/nsb0795-537
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nsb0795-537
This article is cited by
-
Fatal systemic disorder caused by biallelic variants in FARSA
Orphanet Journal of Rare Diseases (2022)
-
The distinction of CPR bacteria from other bacteria based on protein family content
Nature Communications (2019)
-
The mechanistic and evolutionary aspects of the 2′- and 3′-OH paradigm in biosynthetic machinery
Biology Direct (2013)
-
The crystal structures of the α-subunit of the α2β2 tetrameric Glycyl-tRNA synthetase
Journal of Structural and Functional Genomics (2012)
-
Structural insights into the first step of RNA-dependent cysteine biosynthesis in archaea
Nature Structural & Molecular Biology (2007)