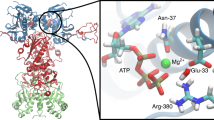
Abstract
Determination of the structure of the substrate binding domain of the Escherichia coli Hsp70 chaperone, DnaK, and the biochemical characterisation of the motif it recognizes within substrates provide insights into the principles governing Hsp70 interaction with polypeptide chains. DnaK recognizes extended peptide strands composed of up to five consecutive hydrophobic residues within and positively charged residues outside the substrate binding cavity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Morimoto, R.I., Tissieres, A. & Georgopoulos, C. The biology of heat shock proteins and molecular chaperones. Cold Spring Harbor Laboratory Press (1994)
Hartl, F.U. Molecular chaperones in cellular protein folding. Nature 381, 571–580 (1996)
Gething, M.-J.H. & Sambrook, J.F. Protein folding in the cell. Nature 355, 33–45 (1992)
Langer, T., Lu, C., Echols, H., Flanagan, J., Hayer, M.K. & Hartl, F.U. Successive action of DnaK, DnaJ and GroEL along the pathway of chaperone-mediated protein folding. Nature 356, 683–689 (1992)
Frydman, J., Nimmesgern, E., Ohtsuka, K. & Hartl, F.U. Folding of nascent polypeptide chains in a high molecular mass assembly with molecular chaperones. Nature 370, 111–117 (1994)
Buchberger, A., Schröder, H., Hesterkamp, T., Schönfeld, H.-J. & Bukau, B. Substrate shuttling between the DnaK and GroEL systems indicates a chaperone network promoting protein folding. J. Mol. Biol. 261, 328–333 (1996)
Freeman, B.C. & Morimoto, R.I. The human cytosolic molecular chaperones hsp90, hsp70 (hsc70) and hdj-1 have distinct roles in recognition of a non-native protein and protein refolding. EMBO J. 15, 2969–2979 (1996)
Bukau, B., Hesterkamp, H. & Luirink, J. Growing up in a dangerous enviroment: a network of multiple targeting and folding pathways for nascent polypetides in the cytosol. Trends Cell Biol. 6, 480–486 (1996)
Craig, E.A., Gambill, B.D. & Nelson, R.J. Heat shock proteins: molecular chaperones of protein synthesis. Microbiological Reviews 57, 402–414 (1993)
Bult, C.J., White, O., Olsen, G.J., Zhou, L., Fleischmann, R.D., Sutton, G.G., Blake, J.A., M., F.L., A., C.R., al., e. & Venter, J.C. Complete genome sequence of the methanogenic archaeon, Methanococcus jannaschii. Science 273, 1058–1073 (1996)
Georgopoulos, C. The emergence of the chaperone machines. TIBS 17, 295–299 (1992)
James, P., Pfund, C. & Craig, E.C. Functional specificity among Hsp70 molecular chaperones. Science 275, 387–389 (1997)
Schatz, G. & Dobberstein, B. Common principles of protein translocation across membranes. Science 271, 1519–1526 (1996)
Georgopoulos, C., Liberek, K., Zylicz, M. & Ang, D. Properties of the heat shock proteins of Escherichia coli and the autoregulation of the heat shock response. In The biology of heat shock proteins and molecular chaperones (R. I. Morimoto, A. Tissieres & C. Georgopoulos, eds.) 209–250 (Cold Spring Harbor Laboratory Press; 1994)
Chappell, T.G., Welch, W., Schlossman, D.M., Palter, K.B., Schlesinger, M.J. & Rothman, J.E. Uncoating ATPase is a member of the 70 kilodalton family of stress proteins. Cell 45, 3–13 (1986)
Sherman, M. & Goldberg, A.L. Involvement of the chaperon DnaK in the rapid degradation of a mutant protein in Escherichia coli. EMBO J. 11, 71–77 (1992)
Straus, D.B., Walter, W.A. & Gross, C.A. Escherichia coli heat shock gene mutants are defective in proteolysis. Genes & Dev. 2, 1851–1858 (1988)
Straus, D., Walter, W. & Gross, C. DnaK, DnaJ, and GrpE heat shock proteins negatively regulate heat shock gene expression by controlling the synthesis and stability of σ32. Genes & Dev. 4, 2202–2209 (1990)
Craig, E.A. & Gross, C.A. Is hsp70 the cellular thermometer? TIBS 16, 135–140 (1991)
Wickner, S., Hoskins, J. & McKenney, K. Monomerization of RepA dimers by heat shock proteins activates binding to DMA replication origins. Proc. Natl. Acad. Sci. USA 88, 7903–7907 (1991)
Blaszczak, A., Zylicz, M., Georgopoulos, C. & Liberek, K. Both ambient temperature and the DnaK chaperone machine modulate the heat shock response in Escherichia coli by regulating the switch between σ70 and σ32 factors assembled with RNA polymerase. EMBO J. 14, 5085–5093 (1995)
Gamer, J., Multhaup, G., Tomoyasu, T., McCarty, J.S., Rüdiger, S., Schönfeld, H.-J., Schirra, C., Bujard, H. & Bukau, B. A cycle of binding and release of the DnaK, DnaJ and GrpE chaperones regulates activity of the E. coli heat shock transcription factor σ32. EMBO J. 15, 607–617 (1996)
Schröder, H., Langer, T., Hartl, F.-U. & Bukau, B. Dnak, DnaJ, GDnaK, DnaJ, GrpE form a cellular chaperone machinery capable of repairing heat-induced protein damage. EMBO J. 12, 4137–4144 (1993)
Skowyra, D., Georgopoulos, C. & Zylicz, M. The E. coli dnaK gene product, the Hsp70 homolog, can reactivate heat-inactivated RNA polymerase in an ATP hydrolysis-dependent manner. Cell 62, 939–944 (1990)
Bukau, B. Regulation of the E. coli heat shock response. Molec. Microbiol. 9, 671–680 (1993)
Flynn, G.C., Chappell, T.G. & Rothman, J.E. Peptide binding and release by proteins implicated as catalysts of protein assembly. Science 245, 385–390 (1989)
Flynn, G.C., Pohl, J., Flocco, M.T. & Rothman, J.E. Peptide-binding specificity of the molecular chaperone BiP. Nature 353, 726–730 (1991)
McCarty, J.S., Buchberger, A., Reinstein, J. & Bukau, B. The role of ATP in the functional cycle of the DnaK chaperone system. J. Mol. Biol. 249, 126–137 (1995)
Liberek, K., Marszalek, J., Ang, D., Georgopoulos, C. & Zylicz, M. Escherichia coli DnaJ and GrpE heat shock proteins jointly stimulate ATPase activity of DnaK. Proc. Natl. Acad. Sci. USA 88, 2874–2878 (1991)
Jordan, R. & McMacken, R. Modulation of the ATPase Activity of the Molecular Chaperone DnaK by Peptides and the DnaJ and GrpE Heat Shock Proteins. J. Biol. Chem. 270, 4563–4569 (1995)
Höhfeld, J., Minami, Y. & Hartl, F.U. Hip, a novel cochaperone involved in the eukaryotic Hsc70/Hsp40 reaction cycle. Cell 83, 589–598 (1995)
Cyr, D.M., Lu, X. & Douglas, M.G. Regulation of Hsp70 function by a eukaryotic DnaJ homolog. J. Biol. Chem. 29, 20927–20931 (1992)
Levy, E.J., McCarty, J., Bukau, B. & Chirico, W.J. Conserved ATPase and luciferase refolding activities between bacteria and yeast Hsp70 chaperones and modulator. FEBS Lett. 368, 435–440 (1995)
Wang, T.-F., Chang, J. & Wang, C. Identification of the peptide binding domain of Hsc70. J. Biol. Chem. 268, 26049–26051 (1993)
Flaherty, K.M., Deluca-Flaherty, C. & McKay, D.B. Three-dimensional structure of the ATPase fragment of a 70K heat-shock cognate protein. Nature (London) 346, 623–628 (1990)
Zhu, X., Zhao, X., Burkholder, W.F., Gragerov, A., Ogata, C.M., Gottesman, M. & Hendrickson, W.A. Structural analysis of substrate binding by the molecular chaperone DnaK. Science 272, 1606–1614 (1996)
Burkholder, W.F., Zhao, X., Zhu, X. & Hendrickson, W.A. Mutations in the C-terminal fragment of DnaK affecting peptide binding. Proc. Nat. Acad. Sci. USA 93, 10632–10637 (1996)
Landry, S.J., Jordan, R., McMacken, R. & Gierasch, L.M. Different conformations for the same polypeptide bound to chaperones DnaK and GroEL. Nature 355, 455–457 (1992)
Richarme, G. & Kohiyama, M. Specificity of the Escherichia coli chaperone DnaK (70-kDa heat shock protein) for hydrophobic amino acids. J. Biol. Chem. 268, 24074–24077 (1993)
Wickner, S., Hoskins, J. & McKenny, K. Function of DnaJ and DnaK as chaperones of origin specific DMA binding by RepA. Nature (London) 350, 165–167 (1991)
Gragerov, A., Zeng, L., Zhao, X., Burkholder, W. & Gottesman, M.E. Specificity of DnaK-peptide binding. J. Mol. Biol. 235, 848–854 (1994)
Kramer, A., Schuster, A., Reineke, U., Malin, R., Volkmer-Engert, R., Landgraf, C. & Schneider-Mergener, J. Combinatorial cellulose-bound peptide libraries: screening tools for the identification of peptidesthat bind ligands with predefined specificity. Methods (Comp. Meth. Enzymol.) 6, 388–395 (1994)
Rüdiger, S., Germeroth, L., Schneider-Mergener, J. & Bukau, B. Substrate specificity of the DnaK chaperone determined by screening of cellulose-bound peptide libraries. EMBO J. 16, 1501–1507 (1997)
Blond-Elguindi, S., Cwirla, S.E., Dower, W.J., Lipshutz, R.J., Sprang, S.R., Sambrook, J.F. & Gething, M.-J.H. Affinity panning of a library of peptides displayed on bacteriophages reveals the binding specificity of BiP. Cell 75, 717–728 (1993)
Alfano, C. & McMacken, R. Ordered assembly of nucleoprotein structures at the bacteriophage λ, replication origin during the initiation of DNA replication. J. Biol. Chem. 264, 10699–10708 (1989)
Gamer, J., Bujard, H. & Bukau, B. Physical interaction between heat shock proteins DnaK, DnaJ, GrpE and the bacterial heat shock transcriptional factor σ32. Cell 69, 833–842 (1992)
Liberek, K., Galitski, T.P., Zylicz, M. & Georgopoulos, C. The DnaK chaperone modulates the heat shock response of Escherichia coli by binding to the σ32 transcription factor. Proc. Natl. Acad. Sci. USA 89, 3516–3520 (1992)
Fourie, A.M., Sambrook, J.F. & Gething, M.-J.H. Common and divergent peptide binding specificities of Hsp70 molecular chaperones. J. Biol. Chem. 269, 30470–30478 (1994)
Gragerov, A. & Gottesman, M.E. Different peptide binding specificities of Hsp70 family members. J. Mol. Biol. 241, 133–135 (1994)
Schmid, D., Baici, A., Gehring, H. & Christen, P. Kinetics of molecular chaperone action. Science 263, 971–973 (1994)
McCarty, J.S., Rüdiger, S., Schönfeld, H.-J., Schneider-Mergener, J., Nakahigashi, K., Yura, T. & Bukau, B. Regulatory region C of the E. coli heat shock transcription factor, σ32, constitutes a DnaK binding site and is conserved among eubacteria. J. Mol. Biol. 256, 829–837 (1996)
Buchberger, A., Valencia, A., McMacken, R., Sander, C. & Bukau, B. The chaperone function of DnaK requires the coupling of ATPase activity with substrate binding through residue E171. EMBO J. 13, 1687–1695 (1994)
Greene, L.E., Zinner, R., Naficy, S. & Eisenberg, E. Effect of nucleotide on the binding of peptides to 70-kDa heat shock protein. J. Biol. Chem. 270, 2967–2973 (1995)
Takeda, S. & McKay, D.B. Kinetics of Peptide Binding to the Bovine 70 kDa Heat Shock Cognate Protein, a Molecular Chaperone. Biochemistry 35, 4636–4644 (1996)
Palleros, D.R., Reid, K.L., Shi, L., Welch, W.J. & Fink, A.L. ATP-induced protein-Hsp70 complex dissociation requires K+ but not ATP hydrolysis. Nature 365, 664–666 (1993)
Theyssen, H., Schuster, H.-P., Bukau, B. & Reinstein, J. SThe second step of ATP Binding to DnaK Induces Peptide Release. J. Mol. Biol. 263, 657–670 (1996)
Munro, S. & Pelham, H.R.B. An Hsp70-like protein in the ER: identity with the 78 kDa glucose-regulated protein and immunoglobulin heavy chain binding protein. Cell 46, 291–300 (1986)
Liberek, K., Skowyra, D., Zylicz, M., Johnson, C. & Georgopoulos, C. The Escherichia coli DnaK chaperone, the 70-kDa heat shock protein eukaryotic equivilant, changes conformation upon ATP hydrolysis, thus triggering its dissociation from a bound target protein. J. Biol. Chem. 266, 14491–14496 (1991)
Wei, J., Gaut, J.R. & Hendershot, L.M. In vitro Dissociation of BiP-Peptide Complexes Requires a Conformational Change in BiP after ATP Binding but Does Not Require ATP Hydrolysis. J. Biol. Chem. 270, 26677–26682 (1995)
Hoffmann, H.J., Lyman, S.K., Lu, C., Petit, M.A. & Echols, H. Activity of the Hsp70 chaperone complex-DnaK, DnaJ, and GrpE-in initiating phage λ DNA replication by sequestering and releasing λ P protein. Proc. Natl. Acad. Sci. USA 89, 12108–12111 (1992)
Wall, D., Zylicz, M. & Georgopoulos, C. The NH2-terminal 108 amino acids of the Escherichia coli DnaJ protein stimulate the ATPase activity of DnaK and are sufficient for λ replication. J. Biol. Chem. 269, 5446–5451 (1994)
Szabo, A., Langer T., Schröder, H., Flanagan, J., Bukau, B. & Hartl, F.U. The ATP hydrolysis-dependent reaction cycle of the Escherichia coli Hsp70 system-DnaK, DnaJ and GrpE. Proc. Natl. Acad. Sci. USA 91, 10345–10349 (1994)
Banecki, B. & Zylicz, M. Real Kinetics of the DnaK/DnaJ/GrpE Molecular Chaperone Machine Action. J. Biol. Chem. 271, 6137–6143 (1996)
Ha, J.-H. & McKay, D.B. Kinetics of Nucleotide-lnduced Changes in the Tryptophane Fluorescence of the Molecular Chaperone Hsc70 and Its Subfragments Suggest the ATP-lnduced Conformational Change Follows Initial ATP Binding. Biochemistry 34, 11635–11644 (1995)
Banecki, B., Zylicz, M., Bertoli, E. & Tanfani, F. Structural and functional relationships in DnaK and DnaK756 heat-shock proteins from Escherichia coli. J. Biol. Chem. 267, 25051–25058 (1992)
Buchberger, A., Theyssen, H., Schröder, H., McCarty, J.S., Virgallita, G., Milkereit, P., Reinstein, J. & Bukau, B. Nucleotide-induced Conformational changes in the ATPase and substrate binding domains of the DnaK chaperone provide evidence for interdomaincommunication. J. Biol. Chem. 270, 16903–16910 (1995)
Shi, L., Kataoka, M. & Fink, A.L. Conformational Characterization of DnaK and Its Complexes by Small-Angle X-ray Scattering. Biochemistry 35, 3297–3308 (1996)
Wilbanks, S.M., Chen, L., Tsuruta, H., Hodgson, K.O. & McKay, D.B. Solution small-angle X-ray scattering study of the molecular chaperone hsc70 and its subfragments. Biochem. 34, 12095–12106 (1995)
Fung, K.L., Hilgenberg, L., Wang, N.M. & Chirico, W.J. Conformations of the Nucleotide and Polypeptide Binding Domains of a Cytosolic Hsp70 Molecular Chaperone Are Coupled. J. Biol. Chem. 271, 21559–21565 (1996)
Wilbanks, S.M. & McKay, D.B. How Potassium Affects the Activity of the Molecular Chaperone Hsc70. II. Potassium Binds Specifically in the ATPase Active Site. J. Biol. Chem. 270, 2251–2257 (1995)
Kamath-Loeb, A.S., Lu, S.Z., Suh, W.-C., Lonetto, M.A. & Gross, C.A. Analysis of Three DnaK Mutant Proteins Suggests That Progression through the ATPase Cycle Requires Conformational Changes. J. Biol. Chem. 270, 30051–30059 (1995)
Holmes, K.C., Sander, C. & Valencia, A. A new ATP-binding fold in actin, hexokinase and Hsc70. Trends Cell Biol. 3, 53–59 (1993)
Tirion, M.M. & ben-Avraham, D. Normal mode analysis of G-actin. J. Mol. Biol. 230, 186–195 (1993).349
Harrison, C.J., Hayer-Hartl, M., Diliberto, M., Hartl, F.U. & Kuriyan, J. Crystal structure of the nucleotide exchange factor GrpE bound to the ATPase domain of the molecular chaperone DnaK. Science in the press.
Packschier, L. et al. The molecular chaperone GrpE accelerates nucleotide exchange of DnaK by an associative displacement mechanism. Biochemistry in the press.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rüdiger, S., Buchberger, A. & Bukau, B. Interaction of Hsp70 chaperones with substrates. Nat Struct Mol Biol 4, 342–349 (1997). https://doi.org/10.1038/nsb0597-342
Issue Date:
DOI: https://doi.org/10.1038/nsb0597-342
This article is cited by
-
A first-in-class inhibitor of Hsp110 molecular chaperones of pathogenic fungi
Nature Communications (2023)
-
Expression of a Heat Shock Protein 70 from the Brown Alga Ectocarpus sp. Imparts Salinity Stress Tolerance in Arabidopsis thaliana
Journal of Applied Phycology (2023)
-
DnaK response to expression of protein mutants is dependent on translation rate and stability
Communications Biology (2022)
-
A Review: Molecular Chaperone-mediated Folding, Unfolding and Disaggregation of Expressed Recombinant Proteins
Cell Biochemistry and Biophysics (2021)
-
The functions and regulation of heat shock proteins; key orchestrators of proteostasis and the heat shock response
Archives of Toxicology (2021)