Abstract
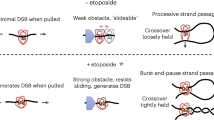
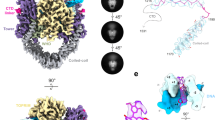
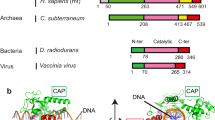
Type II DNA topoisomerases mediate the passage of one DNA duplex through a transient break in another, an event essential for chromosome segregation and cell viability. The active sites of the type II topoisomerase dimer associate covalently with the DNA break-points and must separate by at least the width of the second DNA duplex to accommodate transport. A new structure of the Saccharomyces cerevisiae topoisomerase II DNA-binding and cleavage core suggests that in addition to conformational changes in the DNA-opening platform, a dramatic reorganization of accessory domains may occur during catalysis. These conformational differences have implications for both the DNA-breaking and duplex-transport events in the topo II reaction mechanism, suggest a mechanism by which two distinct drug-resistance loci interact, and illustrate the scope of structural changes in the cycling of molecular machines.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Wang, J.C. in Annu. Rev. Biochem. vol. 65, pp. 635–692 (1996).
Lynn, R., Giaver, G., Swanberg, S.L. & Wang, J.C. Science 233, 647–649 (1986).
Wigley, D.B., Davies, G.J., Dodson, E.J., Maxwell, A. & Dodson, G. Nature 351, 624–629 (1991).
Roca, J.R. & Wang, J.C. Cell 71, 833–840 (1992).
Ali, J.A., Jackson, A.P., Howells, A.J. & Maxwell, A. Biochemistry 32, 2717–2724 (1993).
Horowitz, D.S. & Wang, J.C. J. Biol. Chem. 262, 5339–5344 (1987).
Worland, S.T. & Wang, J.C. J. Biol. Chem. 264, 4412–4416 (1989).
Yoshida, H., Bogaki, M., Nakamura, M., Ya, L.M. & Nakamura, S. Antimicrob. Agents Chemother. 35, 1647–1650 (1991).
Wasserman, R.A. & Wang, J.C. J. Biol. Chem. 269, 20943–20951 (1994).
Reece, R.J. & Maxwell, A. J. Biol. Chem. 264, 19648–19653 (1989).
Berger, J.M., Gamblin, S.J., Harrison, S.C. & Wang, J.C. Nature 379, 225–232 (1996).
Cabral, J.H.M. et al. Nature 388, 903–906 (1997).
Branden, C. & Tooze, J. Introduction to protein structure. (Garland Publishing, New York; 1991).
Berger, J.M., Fass, D., Wang, J.C. & Harrison, S.C. Proc. Natl. Acad. Sci. USA 95, 7876–7881 (1998).
Schultz, S.C., Shields, G.C. & Steitz, T.A. Science 253, 1001–1007 (1991).
Lindsley, J.E. & Wang, J.C. Proc. Nat. Acad. Sci. USA 88, 10485–10489 (1991).
Li, W. & Wang, J.C. J. Biol. Chem. 272, 31190–31195 (1997).
Lima, C.D., Wang, J.C. & Mondragón, A. Nature 367, 138–146 (1994).
Caron, P.R. & Wang, J.C. Molecular biology of DNA topoisomerases and its application to chemotherapy. (eds. Andoh, T., Ikeda, H. & Oguro, M.) 1–18 (CRC Press, Boca Raton; 1993).
Xu, Z., Horwich, A.L. & Sigler, P.B. Nature 388, 741–750 (1997).
Vonrhein, C., Schlauderer, G.J. & Schulz, G.E. Structure 3, 483–490 (1995).
Holmes, K.C. Curr. Opin. Struct. Biol. 6, 781–789 (1996).
Otwinowski, Z. & Minor, W. Methods Enzymol. 276, 472–494 (1997).
Collaborative Computational Project Number 4, Acta Crystallogr. D 50, 760–763 (1994).
Fortelle, E.d.-L. & Bricogne, G. Methods Enzymol. 276, 472–494 (1997).
Abrahams, J.P. & Leslie, A.G.W. Acta Crystallogr. D 52, 30–42 (1996).
Navaza, J. Acta Cryst. D 50, 1507–1516 (1994).
Jones, T.A., Zou, J.Y., Cowan, S.W. & Kjeldgaard, M. Acta Crystallogr. A 47, 110–119 (1991).
Brünger, A.T. XPLOR (Version 3.1): A system for X-ray Crystallography and NMR (Yale University Press, New Haven, Connecticut; 1993).
Laskowski, R.A., MacArthur, M.W., Moss, D.S. & Thornton, J.M. J. Appl. Crystallogr. 26, 283 (1993).
Kraulis, P. J. Appl. Crystallogr. 24, 946–950 (1991).
Carson, M. J. Appl. Crystallogr. 24, 958–961 (1991).
Nicholls, A., Sharp, K.A. & Honig, B. Proteins Struct. Funct. Genet. 11, 281–296 (1991).
Acknowledgements
We thank J.E. Lindsley, A.S. Lynch, P.S. Kim, S.J. Gamblin, S.C. Harrison, and J.C. Wang for discussions on topoisomerase mechanism. We are grateful to C. Ogata of the Brookhaven National Synchrotron Light Source beamline X4A and members of the laboratory of P.S.K. for assistance with data collection, and to S.C.H., J.C.W., D. Minor and M.D. Nichols for critical reading of the manuscript. The authors thank A. Maxwell and R.C. Liddington for providing coordinates of the GyrA breakage-reunion domain, and D.B. Wigley for providing coordinates of the GyrB ATPase domain. D.F. is supported by an NIH Grant to P.S. Kim. J.M.B. acknowledges support from the W.M. Keck Foundation and the Whitehead Institute.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fass, D., Bogden, C. & Berger, J. Quaternary changes in topoisomerase II may direct orthogonal movement of two DNA strands. Nat Struct Mol Biol 6, 322–326 (1999). https://doi.org/10.1038/7556
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/7556
This article is cited by
-
Evolutionary History of TOPIIA Topoisomerases in Animals
Journal of Molecular Evolution (2022)
-
Structural insights into the gating of DNA passage by the topoisomerase II DNA-gate
Nature Communications (2018)
-
A novel and unified two-metal mechanism for DNA cleavage by type II and IA topoisomerases
Nature (2010)
-
In front of and behind the replication fork: bacterial type IIA topoisomerases
Cellular and Molecular Life Sciences (2010)
-
Dynamics of strand passage catalyzed by topoisomerase II
European Biophysics Journal (2010)