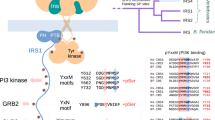
Abstract
We present the NMR structure of the PTB domain of insulin receptor substrate-1 (IRS-1) complexed to a tyrosine-phosphorylated peptide derived from the IL-4 receptor. Despite the lack of sequence homology and different binding specificity, the overall fold of the protein is similar to that of the Shc PTB domain and closely resembles that of PH domains. However, the PTB domain of IRS-1 is smaller than that of She (110 versus 170 residues) and binds to phosphopeptides in a distinct manner. We explain the phosphopeptide binding specificity based on the structure of the complex and results of site-directed mutagenesis experiments.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Myers, M.G., Jr, Sun, X.J. & White, M.F. The IRS-1 signaling system. Trends Biochem. Sci. 192, 89–293 (1994).
Sun, X.J. et al. Structure of the insulin receptor substrate IRS-1 defines a unique signal transduction protein. Nature 352, 73–77 (1991).
Wang, L.-M. et al. IRS-1: Essential for insulin- and IL-4-stimulated mitogensis in hematopoietic cells. Science 261, 1591–1594 (1993).
Keegan, A.D. et al. An IL-4 receptor region containing an insulin receptor motif is important for IL-4-mediated IRS-1 phosphorylation and cell growth. Cell 76, 811–820 (1994).
Gustafson, T.A., He, W., Craparo, A., Schaub, C.D. & O'Neill, T.J. Phosphotyrosine-dependent interaction of She and insulin receptor substrate 1 with the NPEY motif of the insulin receptor via a novel non-SH2 domain. Mol. Cell. Biol. 15, 2500–2508 (1995).
Kavanaugh, W.M. & Williams, L.T. An alternative to SH2 domains for binding tyrosine-phosphorylated proteins. Science 266, 1862–1865 (1994).
Blaikie, P. et al. A region in She distinct from the SH2 domain can bind tyrosine-phosphorylated growth factor receptors. J. Biol. Chem. 269, 32031–32034 (1994).
van der Geer, P. et al. A conserved amino-terminal She domain binds to phosphotyrosine motifs in activated receptors and phosphopeptides. Current Biol. 5, 404–412 (1995).
Zhou, M.-M. et al. Structure and ligand recognition of the phosphotyrosine binding domain of Shc. Nature 378, 584–592 (1995).
Trüb, T. et al. Specificity of the PTB domain of She for β turn forming pentapeptide motifs amino-terminal to phosphotyrosine. J. Biol. Chem. 270, 18205–18208 (1995).
Songyang, Z., Margolis, B., Chaudhuri, M., Shoelson, S.E. & Cantley, L.C. The phosphotyrosine interaction domain of She recognizes tyrosine-phosphorylated NPXY motif. J. Biol. Chem. 270, 14863–18466 (1995).
Wolf, G. et al. PTB domains of IRS-1 and She have distinct but overlapping binding specificities. J. Biol. Chem. 270, 27407–27410 (1995).
Sun, X.J. et al. Role of IRS-2 in insulin and cytokine signalling. Nature 377, 173–177 (1995).
Kavanaugh, W.M., Turck, C.W. & Williams, L.T. PTB domain binding to signaling proteins through a sequence motif containing phosphotyrosine. Science 268, 1177–1180 (1995).
Yoon, H.S. et al. Solution structure of a pleckstrin-homology domain. Nature 369, 672–675 (1994).
Macias, M.J. et al. Structure of the pleckstrin homology domain from β-spectrin. Nature 369, 675–677 (1994).
Downing, A.K. et al. Three-dimensional solution structure of the pleckstrin homology domain from dynamin. Curr. Biol. 4, 884–891 (1994).
Ferguson, K.M., Lemmon, M.A., Schlessinger, J. & Sigler, P.B. Crystal structure at 2.2 Å resolution of the pleckstrin homology domain from human dynamin. Cell 79, 199–209 (1994).
Timm, D. et al. Crystal structure of the pleckstrin homology domain from dynamin Nature Struct. Biol. 1, 782–788 (1994).
Fushman, D., Cahill, S., Lemmon, M.A., Schlessinger, J. & Cowburn, D. Solution structure of pleckstrin homology domain of dynamin by heteronuclear NMR spectroscopy. Proc. Natl. Acad. Sci. USA 92, 816–820 (1995).
Zhang, P., Talluri, S., Deng, H., Branton, D. & Wagner, G. Solution structure of the pleckstrin homology domain of Drosophila β-spectrin. Structure 3. 1185–1195 (1995).
Ferguson, K.M., Lemmon, M.A., Schlessinger, J. & Sigler, P.B. Structure of the high affinity complex of inositol trisphosphate with a phospholipase C pleckstrin homology domain. Cell 83, 1037–1046 (1995).
Harlan, J.E., Hajduk, P.J., Yoon, H.S. & Fesik, S.W. Pleckstrin homology domains bind to phosphatidylinosital 4,5-bisphosphate. Nature 371, 168–170 (1994).
Myers, M.G. Jr. et al. The pleckstrin homology domain in insulin receptor substrate-1 sensitizes insulin signaling. J. Biol. Chem. 270, 11715–11718 (1995).
Touhara, K. et al. Binding of G protein βγ-subunits to pleckstrin homology domains. J. Biol. Chem. 269, 10217–10220 (1994).
Yamazaki, T., Lee, W., Arrowsmith, C.H., Muhandiram, D.R. & Kay, L.E. A suite of triple resonances NMR experiments for the backbone assignment of 15N, 13C, 2H labeled proteins with high sensitivity. J. Am. Chem. Soc. 116, 11655–11666 (1994).
Gore, G.M. & Gronenborn, A.M. Multidimensional heteronuclear nuclear magnetic resonance of proteins. Methods Enzymol. 239, 349–363 (1994).
Neri, D., Szyperski, T., Otting, G., Senn, H. & Wüthrich, K. Biochemistry 28, 7510–7516 (1989).
Nilges, M., Clore, G.M. & Gronenborn, A.M. Determination of three-dimensional structures of proteins from interproton distance data by hybrid distance goemetry-dynamical simulated annealing calculations. FEBS Lett. 229, 317–324 (1988).
Kuszewski, J., Nilges, M. & Brünger, A.T .A sampling and efficacy of metric matrix distance geometry: a novel partial metrization algorithm. J. Biomol. NMR 2, 33–56 (1992).
Brünger, A.T. X-PLOR Manual, Yale Univ.Press, Cambridge, MA, version 3.1 (1992).
Carson, M. Ribbons. J. Molec. Graphics 5, 103–106 (1987).
Brooks, B.R. et al. CHARMM: a program for macromolecular energy minimization, and dynamics calculations. J. Comput. Chem. 4, 187–217 (1983).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zhou, MM., Huang, B., Olejniczak, E. et al. Structural basis for IL-4 receptor phosphopeptide recognition by thelRS-1 PTB domain. Nat Struct Mol Biol 3, 388–393 (1996). https://doi.org/10.1038/nsb0496-388
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nsb0496-388
This article is cited by
-
Orchestration of signaling by structural disorder in class 1 cytokine receptors
Cell Communication and Signaling (2020)
-
Non-histone protein methylation as a regulator of cellular signalling and function
Nature Reviews Molecular Cell Biology (2015)
-
Phosphotyrosine recognition domains: the typical, the atypical and the versatile
Cell Communication and Signaling (2012)
-
Structural basis for the recognition of nucleophosmin-anaplastic lymphoma kinase oncoprotein by the phosphotyrosine binding domain of Suc1-associated neurotrophic factor-induced tyrosine-phosphorylated target-2
Journal of Structural and Functional Genomics (2010)
-
Structural systems biology: modelling protein interactions
Nature Reviews Molecular Cell Biology (2006)