Abstract
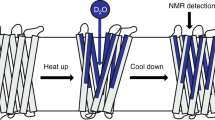
We have determined the three-dimensional fold of the 19 kDa (177 residues) transmembrane domain of the outer membrane protein A of Escherichia coli in dodecylphosphocholine (DPC) micelles in solution using heteronuclear NMR. The structure consists of an eight-stranded β-barrel connected by tight turns on the periplasmic side and larger mobile loops on the extracellular side. The solution structure of the barrel in DPC micelles is similar to that in n-octyltetraoxyethylene (C8E4) micelles determined by X-ray diffraction. Moreover, data from NMR dynamic experiments reveal a gradient of conformational flexibility in the structure that may contribute to the membrane channel function of this protein.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
References
Bax, A. Curr. Op. Struct. Biol. 4, 738–744 (1994).
Gardner, K. H. & Kay, L.E. Annu. Rev. Biomol. Struct . 27, 357–406 (1998).
MacKenzie, K.R., Prestegard J.H. & Engelman, D.M. Science 276, 131–3 (1997).
Rastogi, V.K. & Girvin, M.E. Nature 402, 263–8 (1999).
Ketchem, R.R., Hu, W. & Cross, T.A. Science 261, 1457–1460 (1993).
Opella, S.J. et al. Nature Struct. Biol. 6, 374–379 (1999).
Pervushin, K., Riek, R., Wider, G. & Wüthrich, K. Proc. Natl. Acad. Sci. USA 94, 12366–12371 (1997).
Kay, L.E. & Gardner, K.H. Curr. Opin. Struct. Biol. 7, 722–731 (1997).
Goto, N.K. & Kay, L.E. Curr. Opin. Struct. Biol. 10, 585–592 (2000).
Tjandra, N. & Bax, A. Science 278, 1111–1114 (1997).
Battiste, J.L. & Wagner, G. Biochemistry 39, 5355–5365 (2000).
Grisshammer, R., Averbeck, P. & Sohal, A.K. Biochem. Soc. Trans. 27, 899–903 (1999).
Curran, A.R., Templer, R.H. & Booth, P.L. Biochemistry 38, 9328–9336 (1999).
Sanders, C.R. & Nagy, J.K. Curr. Opin. Struct. Biol. 10, 438–442 (2000).
Kleinschmidt, J.H., den Blaauwen, T., Driessen, A.J.M. & Tamm, L.K. Biochemistry 38, 5006–5016 (1999).
Kleinschmidt, J.H., Wiener, M. & Tamm, L.K. Protein Sci. 8, 2065–2071 (1999).
Arora, A., Rinehart, D., Szabo, G. & Tamm, L.K. J. Biol. Chem. 275, 1594–1600 (2000).
Saint, N., El Hamel, C., De, E. & Molle, G. FEMS Microbiol. Lett. 190, 261–265 (2000).
Belaaouaj, A., Kim, K.S. & Shapiro, S.D. Science 289, 1185–1188 (2000).
Soulas, C. et al. J. Immunol. 165, 2335–2340 (2000).
Pautsch, A. & Schulz, G.E. Nature Struct. Biol. 5, 1013–1017 (1998).
Pautsch, A. & Schulz, G.E. J. Mol. Biol. 298, 273–282 (2000).
Schweizer, M., Hindennach, I., Garten, W. & Henning, U. Eur. J. Biochem. 82, 211–217 (1978).
Dornmair, K., Kiefer, H. & Jähnig, F. J. Biol. Chem. 265, 18907–18911 (1990).
Yang, D. & Kay, L.E. J. Biomol. NMR 13, 3–9 (1999).
Yang, D. & Kay, L.E. J. Am. Chem. Soc. 121, 2571–2575 (1999).
Powers, R. et al. J. Mol. Biol. 221, 1081–1090 (1991).
Zhu, G., Kong, X. M., Sze, K.H. J. Biomol. NMR 13, 77–81 (1999).
Ikura, M., Bax, A., Clore, G.M. & Gronenborn, A.M. J. Am. Chem. Soc. 112, 9020–9022 (1990).
Liu, W.M. J. Mol. Biol. 275, 541–545 (1998).
Caffrey, M. et al. J. Magn. Reson. 135, 368–372 (1998).
Vogel, H., Nilsson, L., Rigler, R., Voges, K.P. & Jung, G. Proc. Natl. Acad. Sci. USA 85, 5067–5071 (1988).
Brown, M.F., Seelig, J. & Häberlen, U. J. Chem. Phys. 70, 5045–5053 (1979).
Waugh, D. J. Biomol. NMR 8, 184–192 (1996).
Muchmore, D.C., McIntosh, L.P., Russell, C.B., Anderson, D.E. & Dahlquist, F.W. Methods Enzymol. 177, 44–73 (1989).
Cornilescu, G., Delaglio, F. & Bax, A. J. Biomol. NMR, 13, 289–302 (1999).
Gardner K.H., Rosen, M.K. & Kay, L.E. Biochemistry 36, 1389–1401 (1997).
Güntert, P., Mumenthaler, C. & Wüthrich, K. J. Mol. Biol . 273, 283–298 (1997).
Luginbühl, P., Güntert, P., Billeter, M. & Wüthrich, K. J. Biomol. NMR 8, 136–146 (1996).
Koradi, R., Billeter, M. & Wüthrich, K. J. Mol. Graphics 14, 51–55 (1996).
Farrow, N.A. et al. Biochemistry 33, 5984–6003 (2000).
Laskowski, R.A., Rullmann, J.A.C., MacArthur, M.W., Kaptein, R. & Thornton, J.M. J. Biomol. NMR 8, 477–486 (1996).
Acknowledgements
This work was supported by a grant from the NIH. The experiments at 750 MHz were carried out at the National Magnetic Resonance Facility at Madison, which is supported by the NIH and NSF.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Arora, A., Abildgaard, F., Bushweller, J. et al. Structure of outer membrane protein A transmembrane domain by NMR spectroscopy. Nat Struct Mol Biol 8, 334–338 (2001). https://doi.org/10.1038/86214
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/86214
This article is cited by
-
Analyzing protein topology based on Laguerre tessellation of a pore-traversing water network
Scientific Reports (2018)
-
Combating virulence of Gram-negative bacilli by OmpA inhibition
Scientific Reports (2017)
-
Comparative study of the structure and interaction of the pore helices of the hERG and Kv1.5 potassium channels in model membranes
European Biophysics Journal (2017)
-
The Burkholderia cenocepacia OmpA-like protein BCAL2958: identification, characterization, and detection of anti-BCAL2958 antibodies in serum from B. cepacia complex-infected Cystic Fibrosis patients
AMB Express (2016)