Abstract
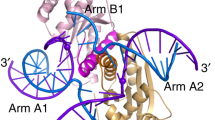
We have solved the crystal structure of the Holliday junction resolving enzyme T7 endonuclease I at 2.1 Å resolution using the multiwavelength anomalous dispersion (MAD) technique. Endonuclease I exhibits strong structural specificity for four-way DNA junctions. The structure shows that it forms a symmetric homodimer arranged in two well-separated domains. Each domain, however, is composed of elements from both subunits, and amino acid side chains from both protomers contribute to the active site. While no significant structural similarity could be detected with any other junction resolving enzyme, the active site is similar to that found in several restriction endonucleases. T7 endonuclease I therefore represents the first crystal structure of a junction resolving enzyme that is a member of the nuclease superfamily of enzymes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
References
Holliday, R. Genet. Res, 5, 282–304 (1964).
Orr-Weaver, T.L., Szostak, J.W. & Rothstein, R.J. Proc. Natl. Acad. Sci. USA 78, 6354–6358 (1981).
Potter, H. & Dressler, D. Proc. Natl. Acad. Sci. USA 73, 3000–3004 (1976).
Schwacha, A. & Kleckner, N. Cell 83, 783–791 (1995).
Duckett, D.R., Giraud-Panis, M.-J.E. & Lilley, D.M.J. J. Mol. Biol. 246, 95–107 (1995).
Pöhler, J.R.G., Giraud-Panis, M.-J.E. & Lilley, D.M.J. J. Mol. Biol. 260, 678–696 (1996).
White, M.F. & Lilley, D.M.J. J. Mol. Biol. 257, 330–341 (1996).
Studier, F.W. Virology 39, 562–574 (1969).
de Massey, B., Studier, F.W., Dorgai, L., Appelbaum, F. & Weisberg, R.A. Cold Spring Harbor Symp. Quant. Biol. 49, 715–726 (1984).
de Massey, B., Weisberg, R.A. & Studier, F.W. J. Mol. Biol. 193, 359–376 (1987).
Dickie, P., McFadden, G. & Morgan, A.R. J. Biol. Chem. 262, 14826–14836 (1987).
Kerr, C. & Sadowski, P.D. Virology 65, 281–285 (1975).
Powling, A. & Knippers, R. Mol. Gen. Genet. 149, 63–71 (1976).
Tsujimoto, Y. & Ogawa, H. J. Mol. Biol. 125, 255–273 (1978).
Parkinson, M.J. & Lilley, D.M.J. J. Mol. Biol. 270, 169–178 (1997).
Ariyoshi, M. et al. Cell 78, 1063–1072 (1994).
Raaijmakers, H. et al. EMBO J. 18, 1447–1458 (1999).
Terwilliger, T.C. & Berendzen, J. Acta Crystallogr. D 53, 571–579 (1997).
Collaborative Computational Project, Number 4. Acta Crystallogr. D 50, 760–763 (1994).
Rafferty, J.B. et al. Science 274, 415–421 (1996).
Guo, F., Gopaul, D.N. & Van Duyne, G.D. Nature 389, 40–46 (1997).
Parkinson, M J., Pöhler, J.R.G. & Lilley, D.M.J. Nucleic Acids Res. 27, 682–689 (1999).
Newman, M. et al. EMBO J. 17, 5466–5476 (1998).
Winkler, F.K. et al. EMBO J. 12, 1781–1795 (1993).
Kim, Y., Grable, J.C., Love, R., Greene, P.J. & Rosenberg, J.M. Science 249, 1307–1309 (1990).
Wah, D.A., Hirsch, J.A., Dorner, L.F., Schildkraut, I. & Aggarwal, A.K. Nature 388, 97–100 (1997).
Pingould, A. & Jeltsch, A. Eur. J. Biochem. 246, 1–22 (1997).
Lee, J., Jayaram, M. & Grainge, I. EMBO J. 18, 784–791 (1999).
Holm, L. & Sander, C. J. Mol. Biol. 233, 123–138 (1993).
Deibert, M., Grazulis, S., Janulaitis, A., Siksnys, V. & Huber, R. EMBO J. 18, 5805–5816 (1999).
Bozic, D., Grazulis, S., Siksnys, V. & Huber, R. J. Mol. Biol. 255, 176–186 (1996).
Hickman, A.B. et al. Mol. Cell. 5, 1025–1034 (2000).
Hargreaves, D. et al. Nature Struct. Biol. 5, 441–445 (1998).
Eichman, B.F., Vargason, J.M., Mooers, B.H.M. & Ho, P.S. Proc. Natl. Acad. Sci. USA 97, 3971–3976 (2000).
Giraud-Panis, M.-J.E. & Lilley, D.M.J. J. Biol. Chem. 271, 33148–33155 (1996).
Kvaratskhelia, M., Wardleworth, B.N., Norman, D.G. & White, M.F. J. Biol. Chem. 275, 25540–25546 (2000).
Ban, C. & Yang, W. EMBO J. 17, 1526–1534 (1998).
Kovall, R. & Matthews, B.W. Science 277, 1824–1827 (1997).
Lilley, D.M.J. & White, M.F. Proc. Natl. Acad. Sci. USA 97, 9351–9353 (2000).
LeMaster, D.M. & Richards, F.M. Biochemistry 24, 7263–7268 (1985).
Graber, P. et al. Eur. J. Biochem. 212, 751–755 (1993).
Leslie, A.G.W. MOSFLM. (MRC Laboratory of Molecular Biology, Cambridge, UK; 1996).
Jones, T.A., Zou, J.-Y., Cowan, S.W. & Kjeldgaard, M. Acta Crystallogr. A 47, 110–119 (1991).
Brünger, A.T. X-PLOR 3.1: a system for crystallography and NMR (Yale University Press, New Haven, Conneticut; 1993).
Brünger, A.T. et al. Acta Crystallogr. D 54, 905–921 (1998).
Duckett, D.R. et al. Cell 55, 79–89 (1988).
Laskwoski, R.A., MacArthur, M.W., Moss, D.W. & Thornton, J.M. J. Appl. Crystallogr. 26, 283–291 (1993).
Christopher, J.A. The Spock Homepage: http://quorum.tamu.edu/spock/ (1998).
Acknowledgements
We would like to thank G. Leonard, S. Azrt, R. Ravelli, and S. McSweeney for help with data collection at the European Synchrotron Radiation Facility and the staff of the Daresbury SRS for assistance with preliminary studies. We are grateful to the Wellcome Trust and the Cancer Research Campaign for financial support and for facilities provided by the BBSRC-funded North of England Structural Biology Centre (NESBIC).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hadden, J., Convery, M., Déclais, AC. et al. Crystal structure of the Holliday junction resolving enzyme T7 endonuclease I. Nat Struct Mol Biol 8, 62–67 (2001). https://doi.org/10.1038/83067
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/83067
This article is cited by
-
Search and processing of Holliday junctions within long DNA by junction-resolving enzymes
Nature Communications (2022)
-
Structural insights into sequence-dependent Holliday junction resolution by the chloroplast resolvase MOC1
Nature Communications (2020)
-
Junction resolving enzymes use multivalency to keep the Holliday junction dynamic
Nature Chemical Biology (2019)
-
Fast design of arbitrary length loops in proteins using InteractiveRosetta
BMC Bioinformatics (2018)
-
Realm of PD-(D/E)XK nuclease superfamily revisited: detection of novel families with modified transitive meta profile searches
BMC Structural Biology (2007)