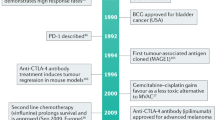
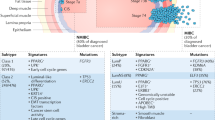
Key Points
-
Over the past 20 years, survival of patients with muscle-invasive bladder cancer has not improved
-
None of the targeted therapies that are approved for other cancers have been approved for the treatment of bladder cancer
-
The small number of clinical trials that have been performed in patients with bladder cancer had limited success owing to several limitations
-
Specific oncogenic mutations in different cell lines used in in vivo research might render these cell lines insensitive to the therapy that is being investigated
-
Inhibition of EGFR, FGFR-3, VEGF, mTOR, STAT3, the androgen receptor and CD24 resulted in inhibition of tumour growth in multiple in vivo studies
-
Careful patient selection in clinical trials based on the molecular profile of the tumour will be essential in demonstrating benefit of new targeted therapies
Abstract
Survival of patients with muscle-invasive bladder cancer is poor and new therapies are needed. Currently, none of the targeted agents that are approved for cancer therapy have been approved for the treatment of bladder cancer and the few clinical trials that have been performed had limited success, often owing to a lack of efficacy and toxic effects. However, many other novel targeted agents have been investigated in animal models of bladder cancer. EGFR, FGFR-3, VEGF, mTOR, STAT3, the androgen receptor and CD24 are molecular targets that could be efficiently inhibited, resulting in reduced tumour growth, and that have been investigated in multiple independent studies. Several other targets, for example COX-2, IL-12, Bcl-xL, livin and choline kinase α, have also been observed to inhibit tumour growth, but these findings have not been replicated to date. Limitations of several studies include the use of cell lines with mutations downstream of the target, providing resistance to the tested therapy. Furthermore, certain technologies, such as interfering RNAs, although effective in vitro, are not yet ready for clinical applications. Further preclinical research is needed to discover and evaluate other possible targets, but several validated targets are now available to be studied in clinical trials.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Kompier, L. C. et al. The development of multiple bladder tumour recurrences in relation to the FGFR3 mutation status of the primary tumour. J. Pathol. 218, 104–112 (2009).
Babjuk, M. et al. EAU guidelines on non-muscle-invasive urothelial carcinoma of the bladder: update 2013. Eur. Urol. 64, 639–653 (2013).
Kiemeney, L. A., Witjes, J. A., Heijbroek, R. P., Verbeek, A. L. & Debruyne, F. M. Predictability of recurrent and progressive disease in individual patients with primary superficial bladder cancer. J. Urol. 150, 60–64 (1993).
Sylvester, R. J. et al. Predicting recurrence and progression in individual patients with stage Ta T1 bladder cancer using EORTC risk tables: a combined analysis of 2596 patients from seven EORTC trials. Eur. Urol. 49, 466–477 (2006).
Surveillance, Epidemiology, and End Results Program (SEER). seer.cancer.gov [online], (2015).
Ploussard, G. et al. Conditional survival after radical cystectomy for bladder cancer: evidence for a patient changing risk profile over time. Eur. Urol. 66, 361–370 (2014).
International Collaboration of Trialists et al. International phase III trial assessing neoadjuvant cisplatin, methotrexate, and vinblastine chemotherapy for muscle-invasive bladder cancer: long-term results of the BA06 30894 trial. J. Clin. Oncol. 29, 2171–2177 (2011).
Visser, O. & Horenblas, S. Blaascarcinoom: Kankerzorg in beeld. Intergraal kankercentrum Nederland [online], (2014).
Abdollah, F. et al. Incidence, survival and mortality rates of stage-specific bladder cancer in United States: a trend analysis. Cancer Epidemiol. 37, 219–225 (2013).
Ferlay, J. et al. GLOBOCAN 2012 v1.0, Cancer Incidence and Mortality Worldwide: IARC CancerBase No. 11. Lyon, France: International Agency for Research on Cancer [online], (2013).
DeVita, V. T. Jr, & Chu, E. A history of cancer chemotherapy. Cancer Res. 68, 8643–8653 (2008).
Ghosh, M., Brancato, S. J., Agarwal, P. K. & Apolo, A. B. Targeted therapies in urothelial carcinoma. Curr. Opin. Oncol. 26, 305–320 (2014).
Gerlinger, M. et al. Intratumour heterogeneity in urologic cancers: from molecular evidence to clinical implications. Eur. Urol. 67, 729–737 (2015).
Diaz, L. A. Jr. et al. The molecular evolution of acquired resistance to targeted EGFR blockade in colorectal cancers. Nature 486, 537–540 (2012).
Philips, G. K. et al. A phase II trial of cisplatin, fixed dose-rate gemcitabine and gefitinib for advanced urothelial tract carcinoma: results of the Cancer and Leukaemia Group B 90102. BJU Int. 101, 20–25 (2007).
Meeks, J. J. et al. A systematic review of neoadjuvant and adjuvant chemotherapy for muscle-invasive bladder cancer. Eur. Urol. 62, 523–533 (2012).
Sausville, E. A. & Burger, A. M. Contributions of human xenografts to anticancer drug development. Cancer Res. 66, 3351–3354 (2006).
Jäger, W. et al. Patient-derived bladder cancer xenografts in the preclinical development of novel targeted therapies. Oncotarget 6, 21522–21532 (2015).
Jäger, W. et al. Hiding in plain view: genetic profiling reveals decades old cross contamination of bladder cancer cell line KU7 with HeLa. J. Urol. 190, 1404–1409 (2013).
Schlessinger, J. Cell signaling by receptor tyrosine kinases. Cell 103, 211–225 (2000).
Cancer Genome Atlas Research Network. Comprehensive molecular characterization of urothelial bladder carcinoma. Nature 507, 315–322 (2014).
Kim, P. H. et al. Genomic predictors of survival in patients with high-grade urothelial carcinoma of the bladder. Eur. Urol. 67, 198–201 (2015).
Black, P. C., Agarwal, P. K. & Dinney, C. P. Targeted therapies in bladder cancer—an update. Urol. Oncol. 25, 433–438 (2007).
Kassouf, W. et al. Distinctive expression pattern of ErbB family receptors signifies an aggressive variant of bladder cancer. J. Urol. 179, 353–358 (2008).
Gerber, D. E. Targeted therapies: a new generation of cancer treatments. Am. Fam. Physician 77, 311–319 (2008).
Krause, D. S. & Van Etten, R. A. Tyrosine kinases as targets for cancer therapy. N. Engl. J. Med. 353, 172–187 (2005).
Bhuvaneswari, R., Gan, Y. Y., Soo, K. C. & Olivo, M. Targeting EGFR with photodynamic therapy in combination with erbitux enhances in vivo bladder tumor response. Mol. Cancer 8, 94 (2009).
Li, F., Ye, Z. Q., Guo, D. S. & Yang, W. M. Suppression of bladder cancer cell tumorigenicity in an athymic mouse model by adenoviral vector-mediated transfer of LRIG1. Oncol. Rep. 26, 439–446 (2011).
Rebouissou, S. et al. EGFR as a potential therapeutic target for a subset of muscle-invasive bladder cancers presenting a basal-like phenotype. Sci. Transl. Med. 6, 244ra91 (2014).
Bhuvaneswari, R., Thong, P., Yuen, G. Y., Olivo, M. & Chee, S. K. Combined use of anti-VEGF and anti-EGFR monoclonal antibodies with photodynamic therapy suppresses tumor growth in an in vivo tumor model. J. Cancer Sci. Ther. 5, 100–105 (2013).
Quesnelle, K. M. & Grandis, J. R. Dual kinase inhibition of EGFR and HER2 overcomes resistance to cetuximab in a novel in vivo model of acquired cetuximab resistance. Clin. Cancer Res. 17, 5935–5944 (2011).
Catalogue of somatic mutations in cancer (COSMIC). cancer.sanger.ac.uk [online], (2015).
Halling-Brown, M. D. et al. canSAR: an integrated cancer public translational research and drug discovery resource. Nucleic Acids Res. 40, D947–D956 (2012).
Karapetis, C. S. et al. K-ras mutations and benefit from cetuximab in advanced colorectal cancer. N. Engl. J. Med. 359, 1757–1765 (2008).
Pruthi, R. S. et al. A phase II trial of neoadjuvant erlotinib in patients with muscle-invasive bladder cancer undergoing radical cystectomy: clinical and pathological results. BJU Int. 106, 349–354 (2010).
Philips, G. K. et al. A phase II trial of cisplatin (C), gemcitabine (G) and gefitinib for advanced urothelial tract carcinoma: results of Cancer and Leukemia Group B (CALGB) 90102. Ann. Oncol. 20, 1074–1079 (2009).
Hussain, M. et al. A randomized phase 2 trial of gemcitabine/cisplatin with or without cetuximab in patients with advanced urothelial carcinoma. Cancer 120, 2684–2693 (2014).
Petrylak, D. P. et al. Results of the Southwest Oncology Group phase II evaluation (study S0031) of ZD1839 for advanced transitional cell carcinoma of the urothelium. BJU Int. 105, 317–321 (2010).
US National Library of Medicine. ClinicalTrials.gov[online], (2015).
US National Library of Medicine. ClinicalTrials.gov[online], (2015).
US National Library of Medicine. ClinicalTrials.gov[online], (2013).
US National Library of Medicine. ClinicalTrials.gov[online], (2012).
US National Library of Medicine. ClinicalTrials.gov[online], (2015).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
Groenendijk, F. H. et al. ERBB2 mutations characterize a subgroup of muscle-invasive bladder cancers with excellent response to neoadjuvant chemotherapy. Eur. Urol. http://dx.doi.org/10.1016/j.eururo.2015.01.014.
van Rhijn, B. W. et al. Molecular grade (FGFR3/MIB-1) and EORTC risk scores are predictive in primary non-muscle-invasive bladder cancer. Eur. Urol. 58, 433–441 (2010).
Tomlinson, D. C., Baldo, O., Harnden, P. & Knowles, M. A. FGFR3 protein expression and its relationship to mutation status and prognostic variables in bladder cancer. J. Pathol. 213, 91–98 (2007).
Miyake, M. et al. 1-tert-butyl-3-[6-(3,5-dimethoxy-phenyl)-2-(4-diethylamino-butylamino)-pyrido[2,3-d]pyrimidin-7-yl]-urea (PD173074), a selective tyrosine kinase inhibitor of fibroblast growth factor receptor-3 (FGFR3), inhibits cell proliferation of bladder cancer carrying the FGFR3 gene mutation along with up-regulation of p27/Kip1 and G1/G0 arrest. J. Pharmacol. Exp. Ther. 332, 795–802 (2010).
Qing, J. et al. Antibody-based targeting of FGFR3 in bladder carcinoma and t(4;14)-positive multiple myeloma in mice. J. Clin. Invest. 119, 1216–1229 (2009).
Gust, K. M. et al. Fibroblast growth factor receptor 3 is a rational therapeutic target in bladder cancer. Mol. Cancer Ther. 12, 1245–1254 (2013).
Wu, Y. M. et al. Identification of targetable FGFR gene fusions in diverse cancers. Cancer Discov. 3, 636–647 (2013).
Lamont, F. R. et al. Small molecule FGF receptor inhibitors block FGFR-dependent urothelial carcinoma growth in vitro and in vivo. Br. J. Cancer 104, 75–82 (2011).
Cheng, T. et al. Fibroblast growth factor receptors-1 and -3 play distinct roles in the regulation of bladder cancer growth and metastasis: implications for therapeutic targeting. PLoS ONE 8, e57284 (2013).
Herrera-Abreu, M. T. et al. Parallel RNA interference screens identify EGFR activation as an escape mechanism in FGFR3-mutant cancer. Cancer Discov. 3, 1058–1071 (2013).
Milowsky, M. I. et al. Phase 2 trial of dovitinib in patients with progressive FGFR3-mutated or FGFR3 wild-type advanced urothelial carcinoma. Eur. J. Cancer 50, 3145–3152 (2014).
Galsky, M. D. et al. Phase Ib study of dovitinib in combination with gemcitabine plus cisplatin or gemcitabine plus carboplatin in patients with advanced solid tumors. Cancer Chemother. Pharmacol. 74, 465–471 (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2015).
Carmeliet, P. VEGF as a key mediator of angiogenesis in cancer. Oncology 69 (Suppl. 3), 4–10 (2005).
Feng, W. et al. siRNA-mediated knockdown of VEGF-A, VEGF-C and VEGFR-3 suppresses the growth and metastasis of mouse bladder carcinoma in vivo. Exp. Ther. Med. 1, 899–904 (2010).
Yang, H. et al. Soluble vascular endothelial growth factor receptor-3 suppresses lymphangiogenesis and lymphatic metastasis in bladder cancer. Mol. Cancer 10, 36 (2011).
Liang, P. H. et al. Construction of a DNA vaccine encoding Flk-1 extracellular domain and C3d fusion gene and investigation of its suppressing effect on tumor growth. Cancer Immunol. Immunother. 59, 93–101 (2010).
Chaudhary, U. B. et al. Phase II trial of neoadjuvant cisplatin, gemcitabine, and bevacizumab followed by radical cystectomy (RC) in patients with muscle-invasive transitional cell carcinoma (TCC) of the bladder. J. Clin. Oncol. 29 (Suppl. 7), abstract 276 (2011).
Siefker-Radtke, A. O. et al. Neoadjuvant chemotherapy with DD-MVAC and bevacizumab in high-risk urothelial cancer: results from a phase II trial at the M. D. Anderson Cancer Center. J. Clin. Oncol. 30 (Suppl. 5), abstract 261 (2012).
Balar, A. V. et al. Phase II study of gemcitabine, carboplatin, and bevacizumab in patients with advanced unresectable or metastatic urothelial cancer. J. Clin. Oncol. 31, 724–730 (2013).
Galsky, M. D. et al. Gemcitabine, cisplatin, and sunitinib for metastatic urothelial carcinoma and as preoperative therapy for muscle-invasive bladder cancer. Clin. Genitourin. Cancer 11, 175–181 (2013).
Hahn, N. M. et al. Phase II trial of cisplatin, gemcitabine, and bevacizumab as first-line therapy for metastatic urothelial carcinoma: Hoosier Oncology Group GU 04–75. J. Clin. Oncol. 29, 1525–1530 (2011).
Gallagher, D. J. et al. Phase II study of sunitinib in patients with metastatic urothelial cancer. J. Clin. Oncol. 28, 1373–1379 (2010).
Twardowski, P. et al. Phase II study of aflibercept (VEGF-Trap) in patients with recurrent or metastatic urothelial cancer, a California Cancer Consortium Trial. Urology 76, 923–926 (2010).
Pili, R. et al. A phase II safety and efficacy study of the vascular endothelial growth factor receptor tyrosine kinase inhibitor pazopanib in patients with metastatic urothelial cancer. Clin. Genitourin. Cancer 11, 477–483 (2013).
Necchi, A. et al. Pazopanib in advanced and platinum-resistant urothelial cancer: an open-label, single group, phase 2 trial. Lancet Oncol. 13, 810–816 (2012).
US National Library of Medicine. ClinicalTrials.gov[online], (2015).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2015).
Puzio-Kuter, A. M. et al. Inactivation of p53 and Pten promotes invasive bladder cancer. Genes Dev. 23, 675–680 (2009).
Makhlin, I. et al. The mTOR pathway affects proliferation and chemosensitivity of urothelial carcinoma cells and is upregulated in a subset of human bladder cancers. BJU Int. 108, E84–E90 (2011).
Seront, E. et al. PTEN deficiency is associated with reduced sensitivity to mTOR inhibitor in human bladder cancer through the unhampered feedback loop driving PI3K/Akt activation. Br. J. Cancer 109, 1586–1592 (2013).
Zhou, H. et al. Urothelial tumor initiation requires deregulation of multiple signaling pathways: Implications in target-based therapies. Carcinogenesis 33, 770–780 (2012).
US National Library of Medicine. ClinicalTrials.gov[online], (2012).
US National Library of Medicine. ClinicalTrials.gov[online], (2015).
US National Library of Medicine. ClinicalTrials.gov[online], (2015).
Seront, E. et al. Phase II study of everolimus in patients with locally advanced or metastatic transitional cell carcinoma of the urothelial tract: clinical activity, molecular response, and biomarkers. Ann. Oncol. 23, 2663–2670 (2012).
Gerullis, H. et al. A phase II trial of temsirolimus in second-line metastatic urothelial cancer. Med. Oncol. 29, 2870–2876 (2012).
Milowsky, M. I. et al. Phase II study of everolimus in metastatic urothelial cancer. BJU Int. 112, 462–470 (2013).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
Rodon, J. et al. Phase I dose-escalation and -expansion study of buparlisib (BKM120), an oral pan-class I PI3K inhibitor, in patients with advanced solid tumors. Invest. New Drugs 32, 670–681 (2014).
US National Library of Medicine. ClinicalTrials.gov[online], (2015).
Harada, Y. et al. Cell-permeable peptide DEPDC1-ZNF224 interferes with transcriptional repression and oncogenicity in bladder cancer cells. Cancer Res. 70, 5829–5839 (2010).
Kodaira, K. et al. Potent cytotoxic effect of a novel nuclear factor-κB inhibitor dehydroxymethylepoxyquinomicin on human bladder cancer cells producing various cytokines. Urology 75, 805–812 (2010).
Shanmugam, R. et al. A water soluble parthenolide analog suppresses in vivo tumor growth of two tobacco-associated cancers, lung and bladder cancer, by targeting NF-κB and generating reactive oxygen species. Int. J. Cancer 128, 2481–2494 (2011).
Sen, M. et al. Targeting Stat3 abrogates EGFR inhibitor resistance in cancer. Clin. Cancer Res. 18, 4986–4996 (2012).
Zhu, J. et al. Downregulation of integrin-linked kinase inhibits epithelial-to-mesenchymal transition and metastasis in bladder cancer cells. Cell Signal. 24, 1323–1332 (2012).
Shiota, M. et al. Androgen receptor signaling regulates cell growth and vulnerability to doxorubicin in bladder cancer. J. Urol. 188, 276–286 (2012).
Wu, J. T., Han, B. M., Yu, S. Q., Wang, H. P. & Xia, S. J. Androgen receptor is a potential therapeutic target for bladder cancer. Urology 75, 820–827 (2010).
Jitao, W. et al. Androgen receptor inducing bladder cancer progression by promoting an epithelial–mesenchymal transition. Andrologia 46, 1128–1133 (2014).
Overdevest, J. B. et al. CD24 offers a therapeutic target for control of bladder cancer metastasis based on a requirement for lung colonization. Cancer Res. 71, 3802–3811 (2011).
Ozawa, A. et al. Inhibition of bladder tumour growth by histone deacetylase inhibitor. BJU Int. 105, 1181–1186 (2010).
Miyamoto, H. et al. Promotion of bladder cancer development and progression by androgen receptor signals. J. Natl Cancer Inst. 99, 558–568 (2007).
Overdevest, J. B. et al. CD24 expression is important in male urothelial tumorigenesis and metastasis in mice and is androgen regulated. Proc. Natl Acad. Sci. USA 109, E3588–E3596 (2012).
Zhou, N. et al. The investigational aurora kinase A inhibitor MLN8237 induces defects in cell viability and cell-cycle progression in malignant bladder cancer cells in vitro and in vivo. Clin. Cancer Res. 19, 1717–1728 (2013).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
Knowles, M. What we could do now: molecular pathology of bladder cancer. Mol. Pathol. 54, 215–221 (2001).
Müllauer, L. et al. Mutations in apoptosis genes: a pathogenetic factor for human disease. Mutat. Res. 488, 211–231 (2001).
Madka, V. et al. p53-stabilizing agent CP-31398 prevents growth and invasion of urothelial cancer of the bladder in transgenic UPII-SV40T mice. Neoplasia 15, 966–974 (2013).
Zhang, Z. et al. Urothelium-specific expression of an oncogene in transgenic mice induced the formation of carcinoma in situ and invasive transitional cell carcinoma. Cancer Res. 59, 3512–3517 (1999).
Card, K. et al. A soluble single-chain T-cell receptor IL-2 fusion protein retains MHC-restricted peptide specificity and IL-2 bioactivity. Cancer Immunol. Immunother. 53, 345–357 (2004).
US National Library of Medicine. ClinicalTrials.gov[online], (2015).
US National Library of Medicine. ClinicalTrials.gov[online], (2015).
Yoshimine, S. et al. Prognostic significance of Bcl-xL expression and efficacy of Bcl-xL targeting therapy in urothelial carcinoma. Br. J. Cancer 11, 2312–2320 (2013).
Gazzaniga, P. et al. Expression and prognostic significance of LIVIN, SURVIVIN and other apoptosis-related genes in the progression of superficial bladder cancer. Ann. Oncol. 14, 85–90 (2003).
Liu, C. A. et al. Antisense oligonucleotide targeting livin induces apoptosis of human bladder cancer cell via a mechanism involving caspase 3. J. Exp. Clin. Cancer Res. 29, 63 (2010).
Wang, H. et al. Translation initiation factor eIF3b expression in human cancer and its role in tumor growth and lung colonization. Clin. Cancer Res. 19, 2850–2860 (2013).
Amit, D. & Hochberg, A. Development of targeted therapy for bladder cancer mediated by a double promoter plasmid expressing diphtheria toxin under the control of H19 and IGF2-P4 regulatory sequences. J. Transl. Med. 8, 134 (2010).
Amit, D., Tamir, S., Birman, T., Gofrit, O. N. & Hochberg, A. Development of targeted therapy for bladder cancer mediated by a double promoter plasmid expressing diphtheria toxin under the control of IGF2-P3 and IGF2-P4 regulatory sequences. Int. J. Clin. Exp. Med. 4, 91–102 (2011).
US National Library of Medicine. ClinicalTrials.gov[online], (2012).
Graff, J. R. et al. Therapeutic suppression of translation initiation factor eIF4E expression reduces tumor growth without toxicity. J. Clin. Invest. 117, 2638–2648 (2007).
Graff, J. R., Konicek, B. W., Carter, J. H. & Marcusson, E. G. Targeting the eukaryotic translation initiation factor 4E for cancer therapy. Cancer Res. 68, 631–634 (2008).
Hsieh, T. F. et al. Epidermal growth factor enhances androgen receptor-mediated bladder cancer progression and invasion via potentiation of AR transactivation. Oncol. Rep. 30, 2917–2922 (2013).
Hong, D. S. et al. A phase 1 dose escalation, pharmacokinetic, and pharmacodynamic evaluation of eIF-4E antisense oligonucleotide LY2275796 in patients with advanced cancer. Clin. Cancer Res. 17, 6582–6591 (2011).
Du, X. et al. FGFR3 stimulates stearoyl CoA desaturase 1 activity to promote bladder tumor growth. Cancer Res. 72, 5843–5855 (2012).
Hernando, E. et al. A critical role for choline kinase-α in the aggressiveness of bladder carcinomas. Oncogene 28, 2425–2435 (2009).
de la Cueva, A. et al. Combined 5-FU and ChoKα inhibitors as a new alternative therapy of colorectal cancer: evidence in human tumor-derived cell lines and mouse xenografts. PLoS ONE 8, e64961 (2013).
US National Library of Medicine. ClinicalTrials.gov[online], (2014).
Abolhassani, M. et al. Screening of well-established drugs targeting cancer metabolism: Reproducibility of the efficacy of a highly effective drug combination in mice. Invest. New Drugs 30, 1331–1342 (2012).
Guais, A. et al. Adding a combination of hydroxycitrate and lipoic acid (METABLOC™) to chemotherapy improves effectiveness against tumor development: experimental results and case report. Invest. New Drugs 30, 200–211 (2012).
Allen, M. D. et al. Prognostic and therapeutic impact of argininosuccinate synthetase 1 control in bladder cancer as monitored longitudinally by PET imaging. Cancer Res. 74, 896–907 (2014).
Parviainen, S. et al. CD40 ligand and tdTomato-armed vaccinia virus for induction of antitumor immune response and tumor imaging. Gene Ther. 21, 195–204 (2014).
Gomes-Giacoia, E., Miyake, M., Goodison, S. & Rosser, C. J. Targeting plasminogen activator inhibitor-1 inhibits angiogenesis and tumor growth in a human cancer xenograft model. Mol. Cancer Ther. 12, 2697–2708 (2013).
Thomas, S. et al. CD24 is an effector of HIF-1-driven primary tumor growth and metastasis. Cancer Res. 72, 5600–5612 (2012).
Yen, M. C. et al. A novel cancer therapy by skin delivery of indoleamine 2,3-dioxygenase siRNA. Clin. Cancer Res. 15, 641–649 (2009).
Liu, W., Cao, Y., Fernandez, M. I., Niu, H. & Xiu, Y. Additive antitumoral effect of interleukin-12 gene therapy and chemotherapy in the treatment of urothelial bladder cancer in vitro and in vivo. Int. Urol. Nephrol. 43, 721–727 (2011).
Kim, J. W., Tomita, Y., Trepel, J. & Apolo, A. B. Emerging immunotherapies for bladder cancer. Curr. Opin. Oncol. 27, 191–200 (2015).
Powles, T. et al. MPDL3280A (anti-PD-L1) treatment leads to clinical activity in metastatic bladder cancer. Nature 515, 3558–62 (2014).
Fenner, A. Could MPDL3280A offer a therapeutic breakthrough in metastatic bladder cancer? Nat. Rev. Urol. 12, 61 (2015).
U. S. Food and Drug Administration. fda.gov[online], (2015).
US National Library of Medicine. ClinicalTrials.gov[online], (2015).
US National Library of Medicine. ClinicalTrials.gov[online], (2015).
US National Library of Medicine. ClinicalTrials.gov[online], (2015).
US National Library of Medicine. ClinicalTrials.gov[online], (2015).
US National Library of Medicine. ClinicalTrials.gov[online], (2015).
US National Library of Medicine. ClinicalTrials.gov[online], (2015).
US National Library of Medicine. ClinicalTrials.gov[online], (2015).
US National Library of Medicine. ClinicalTrials.gov[online], (2015).
Fujita, T. et al. Combined therapeutic effects of adenoviral vector-mediated GLIPR1 gene therapy and radiotherapy in prostate and bladder cancer models. Urol. Oncol. 32, 92–100 (2014).
Wang, H. et al. Enhanced antitumor efficacy of integrin-targeted oncolytic adenovirus AxdAdB3-F/RGD on bladder cancer. Urology 83, 508.e513–508.e519 (2014).
Alemany, R. Viruses in cancer treatment. Clin. Transl. Oncol. 15, 182–188 (2013).
Coffin, R. S. From virotherapy to oncolytic immunotherapy: where are we now? Curr. Opin. Virol. 13, 93–100 (2015).
Bauzon, M. & Hermiston, T. Armed therapeutic viruses—a disruptive therapy on the horizon of cancer immunotherapy. Front. Immunol. 5, 1–10 (2014).
Burke, J. M. et al. A first human phase 1 study of CG0070, a GM-CSF expressing oncolytic adenovirus, for the treatment of nonmuscle invasive bladder cancer. J. Urol. 188, 2391–2397 (2012).
Wu, C. L., Ping, S. Y., Yu, C. P. & Yu, D. S. Tyrosine kinase receptor inhibitor-targeted combined chemotherapy for metastatic bladder cancer. Kaohsiung J. Med. Sci. 28, 194–203 (2012).
Li, X. et al. The differential expression of EphB2 and EphB4 receptor kinases in normal bladder and in transitional cell carcinoma of the bladder. PloS One 9, e105326 (2014).
Zhang, B., Lu, Z., Hou, Y., Hu, J. & Wang, C. The effects of STAT3 and survivin silencing on the growth of human bladder carcinoma cells. Tumour Biol. 35, 5401–5407 (2014).
van der Horst, G. et al. Targeting of α-v integrins reduces malignancy of bladder carcinoma. PLoS ONE 9, e108464 (2014).
Kilkenny, C., Browne, W. J., Cuthill, I. C., Emerson, M. & Altman, D. G. Improving bioscience research reporting: the ARRIVE guidelines for reporting animal research. PLoS Biol. 8, e1000412 (2010).
Baker, D., Lidster, K., Sottomayor, A. & Amor, S. Two years later: journals are not yet enforcing the ARRIVE guidelines on reporting standards for pre-clinical animal studies. PLoS Biol. 12, e1001756 (2014).
Carter, A. J. & Nguyen, C. N. A comparison of cancer burden and research spending reveals discrepancies in the distribution of research funding. BMC Public Health 12, 526 (2012).
Joung, Y. H. et al. Combination of AG490, a Jak2 inhibitor, and methylsulfonylmethane synergistically suppresses bladder tumor growth via the Jak2/STAT3 pathway. Int. J. Oncol. 44, 883–895 (2014).
Qin, J. et al. In vitro and in vivo inhibitory effect evaluation of cyclooxygenase-2 inhibitors, antisense cyclooxygenase-2 cDNA, and their combination on the growth of human bladder cancer cells. Biomed. Pharmacother. 63, 241–248 (2009).
Chen, S. C., Henry, D. O., Hicks, D. G., Reczek, P. R. & Wong, M. K. Intravesical administration of plasminogen activator inhibitor type-1 inhibits in vivo bladder tumor invasion and progression. J. Urol. 181, 336–342 (2009).
Bhattacharya, A. et al. Allyl isothiocyanate-rich mustard seed powder inhibits bladder cancer growth and muscle invasion. Carcinogenesis 31, 2105–2110 (2010).
Bhattacharya, A., Li, Y., Geng, F., Munday, R. & Zhang, Y. The principal urinary metabolite of allyl isothiocyanate, N-acetyl-S-(N-allylthiocarbamoyl)cysteine, inhibits the growth and muscle invasion of bladder cancer. Carcinogenesis 33, 394–398 (2012).
Gao, J. et al. Small interfering RNA targeting integrin-linked kinase inhibited the growth and induced apoptosis in human bladder cancer cells. Int. J. Biochem. Cell Biol. 43, 1294–1304 (2011).
Cheng, H. et al. shRNA targeting PLCε inhibits bladder cancer cell growth in vitro and in vivo. Urology 78, e7–e11 (2011).
Wu, C. T., Chang, Y. H., Lin, P., Chen, W. C. & Chen, M. F. Thrombomodulin expression regulates tumorigenesis in bladder cancer. BMC Cancer 14, 375 (2014).
Garg, M. et al. Heat-shock protein 70–72 (HSP70–2) expression in bladder urothelial carcinoma is associated with tumour progression and promotes migration and invasion. Eur. J. Cancer 46, 207–215 (2010).
Niedworok, C. et al. The impact of the receptor of hyaluronan-mediated motility (RHAMM) on human urothelial transitional cell cancer of the bladder. PLoS ONE 8, e75681 (2013).
Mansure, J. J. et al. A novel mechanism of PPAR γ induction via EGFR signalling constitutes rational for combination therapy in bladder cancer. PLoS ONE 8, e55997 (2013).
Shim, J. S. et al. Effect of nitroxoline on angiogenesis and growth of human bladder cancer. J. Natl Cancer Inst. 102, 1855–1873 (2010).
Shimada, K., Anai, S., Marco, D. A., Fujimoto, K. & Konishi, N. Cyclooxygenase 2-dependent and independent activation of Akt through casein kinase 2 contributes to human bladder cancer cell survival. BMC Urol. 11, 8 (2011).
Matsui, Y. et al. Intravesical combination treatment with antisense oligonucleotides targeting heat shock protein-27 and HTI-286 as a novel strategy for high-grade bladder cancer. Mol. Cancer Ther. 8, 2402–2411 (2009).
Cho, S. D. et al. Activation of nerve growth factor-induced B α by methylene-substituted diindolynmethanes in bladder cancer cells induces apoptosis and inhibits tumor growth. Mol. Pharmacol. 77, 396–404 (2010).
Jeong, K. C. et al. Intravesical instillation of c-MYC inhibitor KSI-3716 suppresses orthotopic bladder tumor growth. J. Urol. 191, 510–518 (2014).
Ding, S. et al. A potent chemotherapeutic strategy for bladder cancer: (S)-methoxy-trityl-L-cystein, a novel Eg5 inhibitor. J. Urol. 184, 1175–1181 (2010).
Seo, H. K. et al. Development of replication-competent adenovirus for bladder cancer by controlling adenovirus E1a and E4 gene expression with the survivin promoter. Oncotarget 5, 5615–5623 (2014).
Seth, S. et al. RNAi-based therapeutics targeting survivin and PLK1 for treatment of bladder cancer. Mol. Ther. 19, 928–935 (2011).
Shimada, K. et al. Role of syndecan-1 (CD138) in cell survival of human urothelial carcinoma. Cancer Sci. 101, 155–160 (2010).
Belgorosky, D. et al. Inhibition of nitric oxide is a good therapeutic target for bladder tumors that express iNOS. Nitric Oxide 36, 11–18 (2014).
Han, C. et al. Target expression of staphylococcus enterotoxin A from an oncolytic adenovirus suppresses mouse bladder tumor growth and recruits CD3+ T cell. Tumor Biol. 34, 2863–2869 (2013).
Fan, Y. et al. TGF-β-induced upregulation of malat1 promotes bladder cancer metastasis by associating with suz12. Clin. Cancer Res. 20, 1531–1541 (2014).
Lin, C. C. et al. A novel adjuvant Ling Zhi-8 enhances the efficacy of DNA cancer vaccine by activating dendritic cells. Cancer Immunol. Immunother. 60, 1019–1027 (2011).
Acquaviva, J. et al. FGFR3 translocations in bladder cancer: differential sensitivity to HSP90 inhibition based on drug metabolism. Mol. Cancer Res. 12, 1042–1054 (2014).
Liu, Y. & Kwiatkowski, D. J. Combined CDKN1A/TP53 mutation in bladder cancer is a therapeutic target. Mol. Cancer Ther. 14, 174–182 (2015).
Levitt, J. M., Yamashita, H., Jian, W., Lerner, S. P. & Sonpavde, G. Dasatinib is preclinically active against Src-overexpressing human transitional cell carcinoma of the urothelium with activated Src signaling. Mol. Cancer Ther. 9, 1128–1135 (2010).
Marra, E. et al. Growth delay of human bladder cancer cells by prostate stem cell antigen downregulation is associated with activation of immune signaling pathways. BMC Cancer 10, 129 (2010).
Liu, A. G. et al. RNA interference targeting adrenomedullin induces apoptosis and reduces the growth of human bladder urothelial cell carcinoma. Med. Oncol. 30, 616 (2013).
Wei, J. et al. The inhibition of human bladder cancer growth by calcium carbonate/CaIP6 nanocomposite particles delivering AIB1 siRNA. Biomaterials 34, 1246–1254 (2013).
Chen, H. et al. MicroRNA-449a acts as a tumor suppressor in human bladder cancer through the regulation of pocket proteins. Cancer Lett. 320, 40–47 (2012).
Zhou, H. et al. Development and characterization of a potent immunoconjugate targeting the Fn14 receptor on solid tumor cells. Mol. Cancer Ther. 10, 1276–1288 (2011).
Liang, P. Y. et al. Overexpression of immunoglobulin G prompts cell proliferation and inhibits cell apoptosis in human urothelial carcinoma. Tumor Biol. 34, 1783–1791 (2013).
Zhao, W. et al. Steroid receptor coactivator-3 regulates glucose metabolism in bladder cancer cells through coactivation of hypoxia inducible factor 1α. J. Biol. Chem. 289, 11219–11229 (2014).
Mao, L. et al. Replication-competent adenovirus expressing TRAIL synergistically potentiates the antitumor effect of gemcitabine in bladder cancer cells. Tumour Biol. 35, 5937–5944 (2014).
Li, C. et al. BCMab1, a monoclonal antibody against aberrantly glycosylated integrin α3β1, has potent antitumor activity of bladder cancer in vivo. Clin. Cancer Res. 20, 4001–4013 (2014).
Said, N., Sanchez-Carbayo, M., Smith, S. C. & Theodorescu, D. RhoGDI2 suppresses lung metastasis in mice by reducing tumor versican expression and macrophage infiltration. J. Clin. Invest. 122, 1503–1518 (2012).
Foulks, J. M. et al. A small-molecule inhibitor of PIM kinases as a potential treatment for urothelial carcinomas. Neoplasia 16, 403–412 (2014).
Zhao, F. et al. Knockdown of a novel lincRNA AATBC suppresses proliferation and induces apoptosis in bladder cancer. Oncotarget 6, 1064–1078 (2015).
Acknowledgements
We would like to acknowledge W. M. Bramer for his contribution to the literature search and database management. This study was funded by an Erasmus MC 2012 grant.
Author information
Authors and Affiliations
Contributions
E.C.Z., K.E.M.v.K., T.C.M.Z. and A.R.A. researched data for the article. E.C.Z., K.E.M.v.K., T.C.M.Z. and J.L.B. substantially contributed to discussion of the content. E.C.Z., K.E.M.v.K. and J.L.B. wrote article. All authors contributed to review and editing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Table 1
In vivo studies using KU-7 animal models (PDF 90 kb)
Supplementary Table 2
In vivo studies investigating targeting of receptor tyrosine kinases and angiogenesis (PDF 110 kb)
Supplementary Table 3
In vivo studies investigating targeting of signal transduction pathways (PDF 114 kb)
Supplementary Table 4
In vivo studies investigating targeting of cell cycle, apoptosis, translation and metabolism (PDF 95 kb)
Supplementary Table 5
In vivo studies investigating targeting of metastasis, the immune system and other strategies (PDF 107 kb)
Rights and permissions
About this article
Cite this article
van Kessel, K., Zuiverloon, T., Alberts, A. et al. Targeted therapies in bladder cancer: an overview of in vivo research. Nat Rev Urol 12, 681–694 (2015). https://doi.org/10.1038/nrurol.2015.231
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrurol.2015.231
This article is cited by
-
Phase II trial of afatinib in patients with advanced urothelial carcinoma with genetic alterations in ERBB1-3 (LUX-Bladder 1)
British Journal of Cancer (2024)
-
RNA-seq reveals novel mechanistic targets of Livin in bladder cancer
BMC Urology (2023)
-
Annexin A1 promotes the progression of bladder cancer via regulating EGFR signaling pathway
Cancer Cell International (2022)
-
SBSN drives bladder cancer metastasis via EGFR/SRC/STAT3 signalling
British Journal of Cancer (2022)
-
A network pharmacology perspective for deciphering potential mechanisms of action of Solanum nigrum L. in bladder cancer
BMC Complementary Medicine and Therapies (2021)