Key Points
-
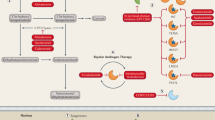
The antiandrogen withdrawal syndrome (AAWS) is characterized by tumour regression and declines in serum PSA levels on discontinuation of antiandrogens or other hormonal agents
-
Initially reported in relation to flutamide, withdrawal responses have been described with other antiandrogens including bicalutamide and enzalutamide, and hormonal agents including oral corticosteroids and spironolactone
-
Mutations activating the androgen receptor (AR), AR amplification and AR coactivator overexpression can all alter ligand specificity, leading to the acquisition of partial agonist properties that account for withdrawal responses
-
Therapeutic steroids, given singly or as concomitant treatment with abiraterone and taxanes, might also be associated with treatment resistance, potentially by activation of mutant AR variants or the glucocorticoid receptor
-
The discovery of the Phe876Leu mutation in the AR has raised the possibility of a withdrawal response to newer antiandrogens such as enzalutamide and ARN-509, but clinical evidence is lacking
-
Structural molecular modelling techniques have been proposed that can identify agents that retain antitumour activity in the presence of these AR mutations
Abstract
The antiandrogen withdrawal syndrome (AAWS) is characterized by tumour regression and a decline in serum PSA on discontinuation of antiandrogen therapy in patients with prostate cancer. This phenomenon has been best described with the withdrawal of the nonsteroidal antiandrogens, bicalutamide and flutamide, but has also been reported with a wide range of hormonal agents. Mutations that occur in advanced prostate cancer and induce partial activation of the androgen receptor (AR) by hormonal agents have been suggested as the main causal mechanism of the AAWS. Corticosteroids, used singly or in conjunction with abiraterone, docetaxel and cabazitaxel might also be associated with the AAWS. The discovery of the Phe876Leu mutation in the AR, which is activated by enzalutamide, raises the possibility of withdrawal responses to novel hormonal agents. This Review focusses on the molecular mechanisms responsible for withdrawal responses, the role of AR mutations in the development of treatment resistance, and the evidence for the sequential use of antiandrogens in prostate cancer therapy. The implications of AR mutations for the development of novel drugs that target the AR are discussed, as are the challenges associated with redefining the utility of older treatments in the current therapeutic landscape.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Ferlay, J. et al. GLOBOCAN 2012 v1.0, Cancer Incidence and Mortality Worldwide: IARC CancerBase No. 11. IARC [online], (2013).
Heidenreich, A. et al. EAU guidelines on prostate cancer. part 1: screening, diagnosis, and local treatment with curative intent-update 2013. Eur. Urol. 65, 124–137 (2014).
Yap, T. A., Zivi, A., Omlin, A. & de Bono, J. S. The changing therapeutic landscape of castration-resistant prostate cancer. Nat. Rev. Clin. Oncol. 8, 597–610 (2011).
Heidenreich, A. et al. EAU guidelines on prostate cancer. Part II: Treatment of advanced, relapsing, and castration-resistant prostate cancer. Eur. Urol. 65, 467–479 (2014).
Denmeade, S. R. & Isaacs, J. T. Development of prostate cancer treatment: the good news. Prostate 58, 211–224 (2004).
Crawford, E. D. et al. A controlled trial of leuprolide with and without flutamide in prostatic carcinoma. N. Engl. J. Med. 321, 419–424 (1989).
Eisenberger, M. A. et al. Bilateral orchiectomy with or without flutamide for metastatic prostate cancer. N. Engl. J. Med. 339, 1036–1042 (1998).
Maximum androgen blockade in advanced prostate cancer: an overview of the randomised trials. Prostate Cancer Trialists' Collaborative Group. Lancet 355, 1491–1498 (2000).
Ryan, C. J. et al. Abiraterone in metastatic prostate cancer without previous chemotherapy N. Engl. J. Med. 368, 138–148 (2013).
Bono, J. De & Logothetis, C. Abiraterone and increased survival in metastatic prostate cancer. N. Engl. J. Med. 1995–2005 (2011).
Scher, H. I. et al. Increased survival with enzalutamide in prostate cancer after chemotherapy. N. Engl. J. Med. 367, 1187–1197 (2012).
Singh, S. M., Gauthier, S. & Labrie, F. Androgen receptor antagonists (antiandrogens): structure-activity relationships. Curr. Med. Chem. 7, 211–247 (2000).
Yuan, X., Cai, C., Chen, S., Yu, Z. & Balk, S. P. Androgen receptor functions in castration-resistant prostate cancer and mechanisms of resistance to new agents targeting the androgen axis. Oncogene 33, 2815–2825 (2014).
Hodgson, M. C., Shen, H. C., Hollenberg, A. N. & Balk, S. P. Structural basis for nuclear receptor corepressor recruitment by antagonist-liganded androgen receptor. Mol. Cancer Ther. 7, 3187–3194 (2008).
Schellhammer, P. F. et al. Clinical benefits of bicalutamide compared with flutamide in combined androgen blockade for patients with advanced prostatic carcinoma: final report of a double-blind, randomized, multicenter trial. Casodex Combination Study Group. Urology 50, 330–336 (1997).
Tran, C. et al. Development of a second-generation antiandrogen for treatment of advanced prostate cancer. Science 324, 787–790 (2009).
Beer, T. M. et al. Enzalutamide in metastatic prostate cancer before chemotherapy. N. Engl. J. Med. 371, 424–433 (2014).
Clegg, N. J. et al. ARN-509: a novel antiandrogen for prostate cancer treatment. Cancer Res. 72, 1494–1503 (2012).
Rathkopf, D. E. et al. A phase I study of the androgen signaling inhibitor ARN 509 in patients with metastatic castration resistant prostate cancer (mCRPC). J. Clin. Oncol. 30 (Suppl.), a4548 (2012).
Chen, Y., Clegg, N. J. & Scher, H. I. Anti-androgens and androgen-depleting therapies in prostate cancer: new agents for an established target. Lancet Oncol. 10, 981–991 (2009).
Joseph, J. D. et al. A clinically relevant androgen receptor mutation confers resistance to second-generation antiandrogens enzalutamide and ARN-509. Cancer Discov. 3, 1020–1029 (2013).
Korpal, M. et al. An F876L mutation in androgen receptor confers genetic and phenotypic resistance to MDV3100 (enzalutamide). Cancer Discov. 3, 1030–1043 (2013).
Sartor, A. O. et al. Antiandrogen withdrawal in castrate-refractory prostate cancer: a Southwest Oncology Group trial (SWOG 9426). Cancer 112, 2393–2400 (2008).
Scher, H. I. & Kelly, W. K. Flutamide withdrawal syndrome: its impact on clinical trials in hormone-refractory prostate cancer. J. Clin. Oncol. 11, 1566–1572 (1993).
Akakura, K., Akimoto, S., Furuya, Y. & Ito, H. Incidence and characteristics of antiandrogen withdrawal syndrome in prostate cancer after treatment with chlormadinone acetate. Eur. Urol. 33, 567–571 (1998).
Huan, S. D., Gerridzen, R. G., Yau, J. C. & Stewart, D. J. Antiandrogen withdrawal syndrome with nilutamide. Urology 49, 632–634 (1997).
Kelly, W. K. & Scher, H. I. Prostate specific antigen decline after antiandrogen withdrawal: the flutamide withdrawal syndrome. J. Urol. 149, 607–609 (1993).
Taplin, M. E. et al. Selection for androgen receptor mutations in prostate cancers treated with androgen antagonist. Cancer Res. 59, 2511–2515 (1999).
Gaddipati, J. P. et al. Frequent detection of codon 877 mutation in the androgen receptor gene in advanced prostate cancers. Cancer Res. 54, 2861–2864 (1994).
Fenton, M. a. et al. Functional characterization of mutant androgen receptors from androgen-independent prostate cancer. Clin. Cancer Res. 3, 1383–1388 (1997).
Scher, H. I. et al. Bicalutamide for advanced prostate cancer: the natural versus treated history of disease. J. Clin. Oncol. 15, 2928–2938 (1997).
Joyce, R. et al. High dose bicalutamide for androgen independent prostate cancer: effect of prior hormonal therapy. J. Urol. 159, 149–153 (1998).
Small, E. J. & Srinivas, S. The antiandrogen withdrawal syndrome. Experience in a large cohort of unselected patients with advanced prostate cancer. Cancer 76, 1428–1434 (1995).
Akakura, K., Akimoto, S., Furuya, Y. & Ito, H. Incidence and characteristics of antiandrogen withdrawal syndrome in prostate cancer after treatment with chlormadinone acetate. Eur. Urol. 33, 567–571 (1998).
Nieh, P. T. Withdrawal phenomenon with the antiandrogen casodex. J. Urol. 153, 1070–1073 (1995).
Bissada, N. K. & Kaczmarek, A. T. Complete remission of hormone refractory adenocarcinoma of the prostate in response to withdrawal of diethylstilbestrol. J. Urol. 153, 1944–1945 (1995).
Chen, C. D. et al. Molecular determinants of resistance to antiandrogen therapy. Nat. Med. 10, 33–39 (2004).
US National Library of Medicine. ClinicalTrials.gov [online], (2014).
Trial from ANZCTR. Australian New Zealand Clinical Trials Registry [online], (2014).
Gelmann, E. P. Molecular biology of the androgen receptor. J. Clin. Oncol. 20, 3001–3015 (2002).
Mostaghel, E. A., Plymate, S. R. & Montgomery, B. Molecular Pathways: Targeting resistance in the androgen receptor for therapeutic benefit. Clin. Cancer Res. 20, 791–798 (2014).
Barbieri, C. E. et al. The Mutational Landscape of Prostate Cancer. Eur. Urol. 64, 567–576 (2013).
Montgomery, R. B. et al. Maintenance of intratumoral androgens in metastatic prostate cancer: a mechanism for castration-resistant tumor growth. Cancer Res. 68, 4447–4454 (2008).
Dehm, S. M., Schmidt, L. J., Heemers, H. V, Vessella, R. L. & Tindall, D. J. Splicing of a novel androgen receptor exon generates a constitutively active androgen receptor that mediates prostate cancer therapy resistance. Cancer Res. 68, 5469–5477 (2008).
Taplin, M. E. et al. Mutation of the androgen-receptor gene in metastatic androgen-independent prostate cancer. N. Engl. J. Med. 332, 1393–1398 (1995).
Tilley, W. D., Buchanan, G., Hickey, T. E. & Bentel, J. M. Mutations in the androgen receptor gene are associated with progression of human prostate cancer to androgen independence. Clin. Cancer Res. 2, 277–285 (1996).
Gelmann, E. P. Androgen receptor mutations in prostate cancer. Cancer Treat. Res. 87, 285–302 (1996).
Tan, J. et al. Dehydroepiandrosterone activates mutant androgen receptors expressed in the androgen-dependent human prostate cancer xenograft CWR22 and LNCaP cells. Mol. Endocrinol. 11, 450–459 (1997).
Culig, Z. et al. Mutant androgen receptor detected in an advanced-stage prostatic carcinoma is activated by adrenal androgens and progesterone. Mol. Endocrinol. 7, 1541–1550 (1993).
Zhao, X. Y. et al. Glucocorticoids can promote androgen-independent growth of prostate cancer cells through a mutated androgen receptor. Nat. Med. 6, 703–706 (2000).
Céraline, J. et al. Constitutive activation of the androgen receptor by a point mutation in the hinge region: a new mechanism for androgen-independent growth in prostate cancer. Int. J. Cancer 108, 152–157 (2004).
Matias, P. M. et al. Structural evidence for ligand specificity in the binding domain of the human androgen receptor. Implications for pathogenic gene mutations. J. Biol. Chem. 275, 26164–26171 (2000).
Bergerat, J.-P. & Céraline, J. Pleiotropic functional properties of androgen receptor mutants in prostate cancer. Hum. Mutat. 30, 145–157 (2009).
Wurtz, J. M. et al. A canonical structure for the ligand-binding domain of nuclear receptors. Nat. Struct. Biol. 3, 206 (1996).
Buchanan, G. et al. Mutations at the boundary of the hinge and ligand binding domain of the androgen receptor confer increased transactivation function. Mol. Endocrinol. 15, 46–56 (2001).
Gottlieb, B., Beitel, L. K., Nadarajah, A., Paliouras, M. & Trifiro, M. The androgen receptor gene mutations database: 2012 update. Hum. Mutat. 33, 887–894 (2012).
Culig, Z. et al. Activation of two mutant androgen receptors from human prostatic carcinoma by adrenal androgens and metabolic derivatives of testosterone. Cancer Detect. Prev. 20, 68–75 (1996).
Veldscholte, J. et al. A mutation in the ligand binding domain of the androgen receptor of human LNCaP cells affects steroid binding characteristics and response to anti-androgens. Biochem. Biophys. Res. Commun. 173, 534–540 (1990).
Veldscholte, J. et al. The androgen receptor in LNCaP cells contains a mutation in the ligand binding domain which affects steroid binding characteristics and response to antiandrogens. J. Steroid Biochem. Mol. Biol. 41, 665–669 (1992).
Monge, A. et al. Unfaithfulness and promiscuity of a mutant androgen receptor in a hormone-refractory prostate cancer. Cell. Mol. Life Sci. 63, 487–497 (2006).
Poukka, H., Aarnisalo, P., Karvonen, U., Palvimo, J. J. & Jänne, O. A. Ubc9 interacts with the androgen receptor and activates receptor-dependent transcription. J. Biol. Chem. 274, 19441–19446 (1999).
Chmelar, R., Buchanan, G., Need, E. F., Tilley, W. & Greenberg, N. M. Androgen receptor coregulators and their involvement in the development and progression of prostate cancer. Int. J. Cancer 120, 719–733 (2007).
Culig, Z. et al. Switch from antagonist to agonist of the androgen receptor bicalutamide is associated with prostate tumour progression in a new model system. Br. J. Cancer 81, 242–251 (1999).
Yoshida, T. et al. Antiandrogen bicalutamide promotes tumor growth in a novel androgen-dependent prostate cancer xenograft model derived from a bicalutamide-treated patient. Cancer Res. 65, 9611–9616 (2005).
Visakorpi, T. et al. In vivo amplification of the androgen receptor gene and progression of human prostate cancer. Nat. Genet. 9, 401–406 (1995).
Masiello, D., Cheng, S., Bubley, G. J., Lu, M. L. & Balk, S. P. Bicalutamide functions as an androgen receptor antagonist by assembly of a transcriptionally inactive receptor. J. Biol. Chem. 277, 26321–26326 (2002).
Gregory, C. W. et al. A mechanism for androgen receptor-mediated prostate cancer recurrence after androgen deprivation therapy. Cancer Res. 61, 4315–4319 (2001).
Miyamoto, H., Yeh, S., Wilding, G. & Chang, C. Promotion of agonist activity of antiandrogens by the androgen receptor coactivator, ARA70, in human prostate cancer DU145 cells. Proc. Natl Acad. Sci. USA 95, 7379–7384 (1998).
Dupont, A., Gomez, J. L., Cusan, L., Koutsilieris, M. & Labrie, F. Response to flutamide withdrawal in advanced prostate cancer in progression under combination therapy. J. Urol. 150, 908–913 (1993).
Herrada, J., Dieringer, P. & Logothetis, C. J. Characterization of patients with androgen-independent prostatic carcinoma whose serum prostate specific antigen decreased following flutamide withdrawal. J. Urol. 155, 620–623 (1996).
Small, E. J. & Carroll, P. R. Prostate-specific antigen decline after casodex withdrawal: evidence for an antiandrogen withdrawal syndrome. Urology 43, 408–410 (1994).
Sella, A., Flex, D., Sulkes, A. & Baniel, J. Antiandrogen withdrawal syndrome with cyproterone acetate. Urology 52, 1091–1093 (1998).
Small, E. J. et al. Antiandrogen withdrawal alone or in combination with ketoconazole in androgen-independent prostate cancer patients: a phase III trial (CALGB 9583). J. Clin. Oncol. 22, 1025–1033 (2004).
Dawson, N. A. & McLeod, D. G. Dramatic prostate specific antigen decrease in response to discontinuation of megestrol acetate in advanced prostate cancer: expansion of the antiandrogen withdrawal syndrome. J. Urol. 153, 1946–1947 (1995).
Nishiyama, T. & Terunuma, M. Prostate specific antigen and prostate acid phosphatase declines after estramustine phosphate withdrawal: a case report. Int. J. Urol. 1, 355–356 (1994).
Akakura, K., Akimoto, S., Ohki, T. & Shimazaki, J. Antiandrogen withdrawal syndrome in prostate cancer after treatment with steroidal antiandrogen chlormadinone acetate. Urology 45, 700–705 (1995).
Longmore, L., Foley, J. P., Rozanski, T. A, Higgins, B. & Thompson, I. M. Prolonged prostate-specific antigen response in flutamide withdrawal syndrome despite disease progression. South. Med. J. 91, 573–575 (1998).
Schellhammer, P. F. et al. Prostate specific antigen decreases after withdrawal of antiandrogen therapy with bicalutamide or flutamide in patients receiving combined androgen blockade. J. Urol. 157, 1731–1735 (1997).
Morote, J. & Bellmunt, J. Bone alkaline phosphatase serum level predicts the response to antiandrogen withdrawal. Eur. Urol. 41, 257–261 (2002).
Figg, W. D. et al. Prostate Specific Antigen Decline Following the Discontinuation of Flutamide in Patients with Stage D2 Prostate Cancer 98, 412–414 (1995).
Terada, N. et al. Antiandrogen withdrawal syndrome and alternative antiandrogen therapy associated with the W741C mutant androgen receptor in a novel prostate cancer xenograft. Prostate 70, 252–61 (2010).
Suzuki, H. et al. Alternative nonsteroidal antiandrogen therapy for advanced prostate cancer that relapsed after initial maximum androgen blockade. J. Urol. 180, 921–927 (2008).
Kojima, S. et al. Alternative antiandrogens to treat prostate cancer relapse after initial hormone therapy. J. Urol. 171, 679–683 (2004).
Okihara, K. et al. Clinical efficacy of alternative antiandrogen therapy in Japanese men with relapsed prostate cancer after first-line hormonal therapy. Int. J. Urol. 14, 128–132 (2007).
Okegawa, T., Nutahara, K. & Higashihara, E. Alternative antiandrogen therapy in patients with castration-resistant prostate cancer: a single-center experience. Int. J. Urol. 17, 950–955 (2010).
Takada, T. et al. [Alternative antiandrogen therapy with flutamide in patients with castration-resistant prostate cancer: a single center experience]. Hinyokika Kiyo. 57, 291–295 (2011).
Choi, J., Il Kim, Y. B., Yang, S. O., Lee, J. K. & Jung, T. Y. Efficacy of alternative antiandrogen therapy for prostate cancer that relapsed after initial maximum androgen blockade. Korean J. Urol. 52, 461–465 (2011).
Kamiya, N. et al. Development of nomogram to non-steroidal antiandrogen sequential alternation in prostate cancer for predictive model. Jpn J. Clin. Oncol. 44, 263–269 (2014).
Balbas, M. D. et al. Overcoming mutation-based resistance to antiandrogens with rational drug design. Elife 2 [online], (2013).
Thadani-Mulero, M. et al. Androgen receptor splice variants determine taxane sensitivity in prostate cancer. Cancer Res. 74, 2270–2282 (2014).
Von Klot, C. A., Kuczyk, M. A. & Merseburger, A. S. No androgen withdrawal syndrome for enzalutamide: a report of disease dynamics in the postchemotherapy setting. Eur. Urol. 65, 258–259 (2014).
Rodriguez-Vida, A. et al. Is there an anti-androgen withdrawal effect with enzalutamide? [abstract] J. Clin. Oncol. 32 (Suppl. 4), a200 (2014).
Venkitaraman, R. et al. Efficacy of low-dose dexamethasone in castration-refractory prostate cancer. BJU Int. 101, 440–443 (2008).
De Bono, J. S. et al. Abiraterone and increased survival in metastatic prostate cancer. N. Engl. J. Med. 364, 1995–2005 (2011).
Ryan, C. J. et al. Abiraterone in metastatic prostate cancer without previous chemotherapy. N. Engl. J. Med. 368, 138–148 (2013).
Arora, V. K. et al. Glucocorticoid receptor confers resistance to antiandrogens by bypassing androgen receptor blockade. Cell 155, 1309–1322 (2013).
Montgomery, B. et al. Impact of Baseline Corticosteroids on Survival and Steroid Androgens in Metastatic Castration-resistant Prostate Cancer: Exploratory Analysis from COU-AA-301. Eur. Urol. http://dx.doi.org/10.1016/j.eururo.2014.06.042 (2014).
Scher, H. I. et al. Impact of on-study corticosteroid use on efficacy and safety in the phase III AFFIRM study of enzalutamide (ENZA), an androgen receptor inhibitor [abstract]. J. Clin. Oncol. 31 (Suppl. 6), a6 (2013).
Venkitaraman, R. et al. A randomized phase II trial of dexamethasone versus prednisolone as secondary hormonal therapy in CRPC [abstract]. J. Clin. Oncol. 31 (Suppl. 6), a123 (2013).
Attard, G. et al. Selective inhibition of CYP17 with abiraterone acetate is highly active in the treatment of castration-resistant prostate cancer. J. Clin. Oncol. 27, 3742–3748 (2009).
Richards, J. et al. Interactions of abiraterone, eplerenone, and prednisolone with wild-type and mutant androgen receptor: a rationale for increasing abiraterone exposure or combining with MDV3100. Cancer Res. 72, 2176–2182 (2012).
Sahu, B. et al. FoxA1 specifies unique androgen and glucocorticoid receptor binding events in prostate cancer cells. Cancer Res. 73, 1570–1580 (2012).
De Bono, J. S. et al. Abiraterone and increased survival in metastatic prostate cancer. N. Engl. J. Med. 364, 1995–2005 (2011).
Sundar, S. & Dickinson, P. D. Spironolactone, a possible selective androgen receptor modulator, should be used with caution in patients with metastatic carcinoma of the prostate. BMJ Case Rep. 2012 http://dx.doi.org/10.1136/bcr.11.2011.5238 (2012).
Attard, G. et al. Phase I clinical trial of a selective inhibitor of CYP17, abiraterone acetate, confirms that castration-resistant prostate cancer commonly remains hormone driven. J. Clin. Oncol. 26, 4563–4571 (2008).
US National Library of Medicine. ClinicalTrials.gov [online], (2014).
Leibowitz-Amit, R. et al. Retreatment of men with metastatic castrate-resistant prostate cancer with abiraterone. Prostate 74, 1462–1464 (2014).
Lorente, D. et al. Tumour responses following a steroid switch from prednisone to dexamethasone in castration-resistant prostate cancer patients progressing on abiraterone. Br. J. Cancer http://dx.doi.org/10.1038/bjc.2014.531 (2014).
Schmid, S. et al. Abiraterone—What Is Wrong With the Adrenal Glands? Clin. Genitourin. Cancer http://dx.doi.org/10.1016/j.clgc.2014.01.008 (2014).
Caffo, O. et al. Biochemical and objective response to abiraterone acetate withdrawal: incidence and clinical relevance of a new scenario for castration-resistant prostate cancer. Urology 82, 1090–1093 (2013).
Scher, H. I. et al. Design and end points of clinical trials for patients with progressive prostate cancer and castrate levels of testosterone: recommendations of the Prostate Cancer Clinical Trials Working Group. J. Clin. Oncol. 26, 1148–1159 (2008).
Attard, G., Reid, A. H. M. & de Bono, J. S. Abiraterone acetate is well tolerated without concomitant use of corticosteroids. J. Clin. Oncol. 28, e560–e562 (2010).
Sweeney, C. et al. Impact on overall survival (OS) with chemohormonal therapy versus hormonal therapy for hormone-sensitive newly metastatic prostate cancer (mPrCa): An ECOG-led phase III randomized trial [abstract LBA2]. J. Clin. Oncol. 32 (Suppl.), 5s (2014).
Halabi, S. et al. Prostate-specific antigen changes as surrogate for overall survival in men with metastatic castration-resistant prostate cancer treated with second-line chemotherapy. J. Clin. Oncol. 31, 3944–3950 (2013).
Josan, J. S. & Katzenellenbogen, J. A. Designer antiandrogens join the race against drug resistance. Elife 2 [online], (2013).
Lonergan, P. E. and Tindall, D. J. Androgen receptor signaling in prostate cancer development and progression. J. Carcinog. 10, http://dx.doi.org/10.4103/1477-3163.83937 (2011).
Taplin, M. E. et al. Androgen receptor mutations in androgen-independent prostate cancer: Cancer and Leukemia Group B Study 9663. J. Clin. Oncol. 21, 2673–2678 (2003).
Hara, T. et al. Novel mutations of androgen receptor: a possible mechanism of bicalutamide withdrawal syndrome. Cancer Res. 63, 149–153 (2003).
Chen, G. et al. Androgen receptor mutants detected in recurrent prostate cancer exhibit diverse functional characteristics. Prostate 63, 395–406 (2005).
Duff, J. & McEwan, I. J. Mutation of histidine 874 in the androgen receptor ligand-binding domain leads to promiscuous ligand activation and altered p160 coactivator interactions. Mol. Endocrinol. 19, 2943–2954 (2005).
Korpal, M. et al. An F876L mutation in androgen receptor confers genetic and phenotypic resistance to MDV3100 (enzalutamide). Cancer Discov. 3, 1030–1043 (2013).
Sartor, O. et al. Surprising activity of flutamide withdrawal, when combined with aminoglutethimide, in treatment of “hormone-refractory” prostate cancer. J. Natl Cancer Inst. 86, 222–227 (1994).
Sartor, O. et al. Antiandrogen withdrawal in castrate-refractory prostate cancer: a Southwest Oncology Group trial (SWOG 9426). Cancer 112, 2393–2400 (2008).
Small, E. J. et al. Antiandrogen withdrawal alone or in combination with ketoconazole in androgen-independent prostate cancer patients: a phase III trial (CALGB 9583). J. Clin. Oncol. 22, 1025–1033 (2004).
Acknowledgements
D.L., J.M., Z.Z., A.D.S., R.F. and J.S.d.B., are employees of the Section of Medicine, which is supported by a Cancer Research UK programme grant and an Experimental Cancer Medical Centre (ECMC) grant from Cancer Research UK and the Department of Health (Ref: C51/A7401). D.L. is supported by a grant from the Spanish Society for Medical Oncology 'Beca SEOM para la Investigación Traslacional en el Extranjero'. J.M. is funded by the Medical Research Council and Prostate Cancer UK-Movember Foundation and Z.Z. is supported by a grant from the Hellenic Society of Medical Oncology.
Author information
Authors and Affiliations
Contributions
All authors contributed equally to data research, discussions of content, writing the article and editing the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
J.S.d.B. has received consulting fees from Ortho Biotech Oncology Research and Development (a unit of Cougar Biotechnology), consulting fees and travel support from Amgen, Astellas, AstraZeneca, Boehringer Ingelheim, Bristol-Myers Squibb, Dendreon, Enzon, Exelixis, Genentech, GlaxoSmithKline, Medivation, Merck, Novartis, Pfizer, Roche, Sanofi–Aventis, Supergen, and Takeda, and grant support from AstraZeneca and Genentech. D.L., J.M., Z.Z., A.D.S., S.S. and R.F. declare no competing interests.
Rights and permissions
About this article
Cite this article
Lorente, D., Mateo, J., Zafeiriou, Z. et al. Switching and withdrawing hormonal agents for castration-resistant prostate cancer. Nat Rev Urol 12, 37–47 (2015). https://doi.org/10.1038/nrurol.2014.345
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrurol.2014.345
This article is cited by
-
Clinical implications of genomic alterations in metastatic prostate cancer
Prostate Cancer and Prostatic Diseases (2021)
-
Resistance to the Androgen Receptor Centred Therapies: Biology and Management
SN Comprehensive Clinical Medicine (2021)
-
Neurotensin and its receptors mediate neuroendocrine transdifferentiation in prostate cancer
Oncogene (2019)
-
Functional analysis of androgen receptor mutations that confer anti-androgen resistance identified in circulating cell-free DNA from prostate cancer patients
Genome Biology (2016)
-
Alterations in androgen deprivation enhanced prostate-specific membrane antigen (PSMA) expression in prostate cancer cells as a target for diagnostics and therapy
EJNMMI Research (2015)