Abstract
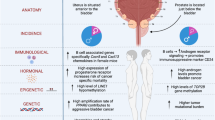
Early studies documented the existence of sexual dimorphism in bladder cancer occurrence and progression, with a greater bladder cancer incidence in males than females. However, the progression of bladder cancer after diagnosis is much quicker in females than males. These findings can be explained by the effects of female hormones (predominantly oestrogens) and their binding receptors, including oestrogen receptor 1 (ESR1; also known as ERα), oestrogen receptor 2 (ESR2; also known as ERβ), and GPR30 protein on bladder cancer incidence and progression. Results from studies using various in vitro cell lines and in vivo mouse models demonstrate differential roles of oestrogen receptors in cancer initiation and progression. ERα suppresses bladder cancer initiation and invasion, whereas ERβ promotes bladder cancer initiation and progression. Mechanistic studies suggest that ERα and ERβ exert these effects via modulation of the AKT pathway and DNA replication complex, respectively. Targeting these signalling pathways—for example, with ERα agonists, ERβ antagonists, or selective oestrogen receptor modulators such as 4-[2-phenyl-5,7-bis(trifluoromethyl)pyrazolo[1,5-a]pyrimidin-3-yl]phenol (also known as PHTPP)—could lead to the development of new therapeutic approaches for controlling bladder cancer progression.
Key Points
-
Although bladder cancer incidence is greater in men, bladder cancer tends to be more aggressive in women (greater muscle invasion rate), suggesting that oestrogen inhibits tumour initiation but promotes invasion
-
Differential expression of oestrogen receptor 1 (ESR1; also known as ERα) and oestrogen receptor 2 (ESR2; also known as ERβ) in human bladder cancers implicates different roles in bladder tumorigenesis
-
Reported inconsistencies in ERα and ERβ expression—and their correlation with bladder cancer stage, grade, and patient survival—can be explained by differences in study design and disease complexity
-
Studies of mouse models and human cell lines suggest that ERα suppresses initiation and invasion, whereas ERβ promotes initiation and progression
-
Inducible knockout mouse studies (with selective deletion of ERα, ERβ, or GPR30 in urothelial cells at specific stages of tumorigenesis) could elucidate the role of oestrogens in bladder cancer development
-
New therapeutic approaches might selectively target ERα or ERβ (or their downstream target genes); for example, a selective ERα agonist or ERβ antagonist could have therapeutic effect
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Jemal, A. et al. Global cancer statistics. CA Cancer J. Clin. 61, 69–90 (2011).
Siegel, R., Naishadham, D. & Jemal, A. Cancer statistics, 2012. CA Cancer J. Clin. 62, 10–29 (2012).
Scelo, G. & Brennan, P. The epidemiology of bladder and kidney cancer. Nat. Clin. Pract. Urol. 4, 205–217 (2007).
Brennan, P. et al. Cigarette smoking and bladder cancer in men: a pooled analysis of 11 case-control studies. Int. J. Cancer 86, 289–294 (2000).
Freedman, N. D., Silverman, D. T., Hollenbeck, A. R., Schatzkin, A. & Abnet, C. C. Association between smoking and risk of bladder cancer among men and women. JAMA 306, 737–745 (2011).
Richardson, K., Band, P. R., Astrakianakis, G. & Le, N. D. Male bladder cancer risk and occupational exposure according to a job-exposure matrix-a case-control study in British Columbia, Canada. Scand. J. Work Environ. Health 33, 454–464 (2007).
Skov, T. et al. Risk for cancer of the urinary bladder among hairdressers in the Nordic countries. Am. J. Ind. Med. 17, 217–223 (1990).
Sorahan, T. Bladder cancer risks in workers manufacturing chemicals for the rubber industry. Occup. Med. (Lond.) 58, 496–501 (2008).
Tremblay, C., Armstrong, B., Theriault, G. & Brodeur, J. Estimation of risk of developing bladder cancer among workers exposed to coal tar pitch volatiles in the primary aluminum industry. Am. J. Ind. Med. 27, 335–348 (1995).
Hall, P. Radiation-associated urinary bladder cancer. Scand. J. Urol. Nephrol. Suppl. 218, 85–88 (2008).
Nieder, A. M., Porter, M. P. & Soloway, M. S. Radiation therapy for prostate cancer increases subsequent risk of bladder and rectal cancer: a population based cohort study. J. Urol. 180, 2005–2010 (2008).
Chaturvedi, A. K. et al. Second cancers among 104,760 survivors of cervical cancer: evaluation of long-term risk. J. Natl Cancer Inst. 99, 1634–1643 (2007).
Kantor, A. F. et al. Urinary tract infection and risk of bladder cancer. Am. J. Epidemiol. 119, 510–515 (1984).
Michaud, D. S. Chronic inflammation and bladder cancer. Urol. Oncol. 25, 260–268 (2007).
Hardin, B. M., White, W. M., Clark, C. T. & Maclennan, G. T. Urinary tract schistosomiasis. J. Urol. 184, 2136–2137 (2010).
Parkin, D. M. The global health burden of infection-associated cancers in the year 2002. Int. J. Cancer 118, 3030–3044 (2006).
Lynch, C. F., Davila, J. A. and Platz, C. E. in Cancer of the urinary bladder 181–192 (National Cancer Institute, Bethesda, MD, 2007). [Series eds Ries, L. A. G., Young, J. L., Keel, G. E., Eisner, M. P., Lin, Y. D., & Horner, M.-J. Surveillance Epidemiology and End Results (SEER) Survival Monograph].
Vaidya, A., Soloway, M. S., Hawke, C., Tiguert, R. & Civantos, F. De novo muscle invasive bladder cancer: is there a change in trend? J. Urol. 165, 47–50 (2001).
Mungan, N. A. et al. Gender differences in stage-adjusted bladder cancer survival. Urology 55, 876–880 (2000).
Castelao, J. E. et al. Gender- and smoking-related bladder cancer risk. J. Natl Cancer Inst. 93, 538–545 (2001).
Mori, T., Bern, H. A., Mills, K. T. & Young, P. N. Long-term effects of neonatal steroid exposure on mammary gland development and tumorigenesis in mice. J. Natl Cancer Inst. 57, 1057–1062 (1976).
Warner, M. R. & Warner, R. L. Effects of exposure of neonatal mice to 17beta-estradiol on subsequent age-incidence and morphology of carcinogen-induced mammary dysplasia. J. Natl Cancer Inst. 55, 289–298 (1975).
Cavalieri, E. L. et al. Molecular origin of cancer: catechol estrogen-3, 4-quinones as endogenous tumor initiators. Proc. Natl Acad. Sci. USA 94, 10937–10942 (1997).
Stack, D. E., Byun, J., Gross, M. L., Rogan, E. G. & Cavalieri, E. L. Molecular characteristics of catechol estrogen quinones in reactions with deoxyribonucleosides. Chem. Res. Toxicol. 9, 851–859 (1996).
Bolton, J. L. & Thatcher, G. R. Potential mechanisms of estrogen quinone carcinogenesis. Chem. Res. Toxicol. 21, 93–101 (2008).
Chen, Y. et al. A metabolite of equine estrogens, 4-hydroxyequilenin, induces DNA damage and apoptosis in breast cancer cell lines. Chem. Res. Toxicol. 13, 342–350 (2000).
Liu, X. et al. Oxidative DNA damage induced by equine estrogen metabolites: role of estrogen receptor alpha. Chem. Res. Toxicol. 15, 512–519 (2002).
Okajima, E., Hiramatsu, T., Iriya, K., Ijuin, M. & Matsushima, S. Effects of sex hormones on development of urinary bladder tumours in rats induced by N-butyl-N-(4-hydroxybutyl) nitrosamine. Urol. Res. 3, 73–79 (1975).
Tanahashi, N. K., Suzawa, N. & Azuma, C. Effects of sex hormones on oncogenesis in rat urinary bladder by N-butyl-N-(4-hydroxybutyl)-nitrosamine. Int. J. Clin. Pharmacol. Biopharm. 15, 101–105 (1977).
McGrath, M., Michaud, D. S. & De Vivo, I. Hormonal and reproductive factors and the risk of bladder cancer in women. Am. J. Epidemiol. 163, 236–244 (2006).
Dietrich, K. et al. Parity, early menopause and the incidence of bladder cancer in women: a case-control study and meta-analysis. Eur. J. Cancer 47, 592–599 (2010).
Cantor, K. P., Lynch, C. F. & Johnson, D. Bladder cancer, parity, and age at first birth. Cancer Causes Control 3, 57–62 (1992).
Wolpert, B. J. et al. Estrogen exposure and bladder cancer risk in Egyptian women. Maturitas 67, 353–357 (2010).
Davis-Dao, C. A. et al. Lower risk in parous women suggests that hormonal factors are important in bladder cancer etiology. Cancer Epidemiol. Biomarkers Prev. 20, 1156–1170 (2011).
Huang, A. T. et al. Bladder cancer and reproductive factors among women in Spain. Cancer Causes Control 20, 1907–1913 (2009).
Cantwell, M. M., Lacey, J. V., Jr, Schairer, C., Schatzkin, A. & Michaud, D. S. Reproductive factors, exogenous hormone use and bladder cancer risk in a prospective study. Int. J. Cancer 119, 2398–2401 (2006).
Carrera-Gonzalez, M. D. et al. Putative relationship between hormonal status and serum pyrrolidone carboxypeptidase activity in pre- and post- menopausal women with breast cancer. Breast 6, 751–754 (2012).
Li, Y. et al. FSH stimulates ovarian cancer cell growth by action on growth factor variant receptor. Mol. Cell Endocrinol. 267, 26–37 (2007).
Silva, E. G. et al. Elevated luteinizing hormone in serum, breast cancer tissue, and normal breast tissue from breast cancer patients. Breast Cancer Res. Treat. 76, 125–130 (2002).
Marquez, D. C., Lee, J., Lin, T. & Pietras, R. J. Epidermal growth factor receptor and tyrosine phosphorylation of estrogen receptor. Endocrine 16, 73–81 (2001).
Lindberg, M. K. et al. Estrogen receptor specificity for the effects of estrogen in ovariectomized mice. J. Endocrinol. 174, 167–178 (2002).
Couse, J. F. et al. Estrogen receptor-alpha knockout mice exhibit resistance to the developmental effects of neonatal diethylstilbestrol exposure on the female reproductive tract. Dev. Biol. 238, 224–238 (2001).
Spencer-Segal, J. L. et al. Estradiol acts via estrogen receptors alpha and beta on pathways important for synaptic plasticity in the mouse hippocampal formation. Neuroscience 202, 131–146 (2011).
Hill, L., Jeganathan, V., Chinnasamy, P., Grimaldi, C. & Diamond, B. Differential roles of estrogen receptors alpha and beta in control of B-cell maturation and selection. Mol. Med. 17, 211–220 (2010).
Wang, C. et al. GPR30 contributes to estrogen-induced thymic atrophy. Mol. Endocrinol. 22, 636–648 (2008).
Sharma, G. & Prossnitz, E. R. Mechanisms of estradiol-induced insulin secretion by the G protein-coupled estrogen receptor GPR30/GPER in pancreatic beta-cells. Endocrinology 152, 3030–3039 (2011).
Lin, B. C. et al. Stimulating the GPR30 estrogen receptor with a novel tamoxifen analogue activates SF-1 and promotes endometrial cell proliferation. Cancer Res. 69, 5415–5423 (2009).
Ignatov, T. et al. Role of GPR30 in endometrial pathology after tamoxifen for breast cancer. Am. J. Obstet. Gynecol. 203, 595 e9–e16 (2010).
Tuygun, C. et al. Sex-specific hormone receptors in urothelial carcinomas of the human urinary bladder: a comparative analysis of clinicopathological features and survival outcomes according to receptor expression. Urol. Oncol. 29, 43–51 (2009).
Shen, S. S. et al. Expression of estrogen receptors-alpha and -beta in bladder cancer cell lines and human bladder tumor tissue. Cancer 106, 2610–2616 (2006).
Miyamoto, H. et al. Expression of androgen and oestrogen receptors and its prognostic significance in urothelial neoplasm of the urinary bladder. BJU Int. 109, 1716–1726 (2012).
Bolenz, C., Lotan, Y., Ashfaq, R. & Shariat, S. F. Estrogen and progesterone hormonal receptor expression in urothelial carcinoma of the bladder. Eur. Urol. 56, 1093–1095 (2009).
Croft, P. R., Lathrop, S. L., Feddersen, R. M. & Joste, N. E. Estrogen receptor expression in papillary urothelial carcinoma of the bladder and ovarian transitional cell carcinoma. Arch. Pathol. Lab. Med. 129, 194–199 (2005).
Teng, J., Wang, Z. Y., Jarrard, D. F. & Bjorling, D. E. Roles of estrogen receptor alpha and beta in modulating urothelial cell proliferation. Endocr. Relat. Cancer 15, 351–364 (2008).
Lokeshwar, V. B., Merseburger, A. S. & Hautmann, S. H. Bladder tumors: molecular aspects and clinical management 43–79 (Humana Press, New York, 2011).
Kirkali, Z. et al. Bladder cancer: epidemiology, staging and grading, and diagnosis. Urology 66, 4–34 (2005).
Schned, A. R. et al. Histological classification and stage of newly diagnosed bladder cancer in a population-based study from the Northeastern United States. Scand. J. Urol. Nephrol. 42, 237–242 (2008).
Bolenz, C., Lotan, Y., Ashfaq, R. & Shariat, S. F. Estrogen and progesterone hormonal receptor expression in urothelial carcinoma of the bladder. Eur. Urol. 56, 1093–1095 (2009).
Kontos, S., Kominea, A., Melachrinou, M., Balampani, E. & Sotiropoulou-Bonikou, G. Inverse expression of estrogen receptor-beta and nuclear factor-kappaB in urinary bladder carcinogenesis. Int. J. Urol. 17, 801–809 (2010).
Basakci, A. et al. Prognostic significance of estrogen receptor expression in superficial transitional cell carcinoma of the urinary bladder. Eur. Urol. 41, 342–345 (2002).
Hammond, M. E. et al. American Society of Clinical Oncology/College of American Pathologists guideline recommendations for immunohistochemical testing of estrogen and progesterone receptors in breast cancer. Arch. Pathol. Lab. Med. 134, 907–922 (2010).
Qiu, J. et al. Effect of delayed formalin fixation on estrogen and progesterone receptors in breast cancer: a study of three different clones. Am. J. Clin. Pathol. 134, 813–819 (2010).
Khoury, T. et al. Delay to formalin fixation effect on breast biomarkers. Mod. Pathol. 22, 1457–1467 (2009).
Skliris, G. P. et al. Evaluation of seven oestrogen receptor beta antibodies for immunohistochemistry, western blotting, and flow cytometry in human breast tissue. J. Pathol. 197, 155–162 (2002).
Shaaban, A. M., O'Neill, P. A. & Foster, C. S. Re: Skliris et al. Evaluation of seven oestrogen receptor beta antibodies for immunohistochemistry, western blotting, and flow cytometry in human breast tissue. J. Pathol. 2002; 196, 155–162. J. Pathol. 199, 130–131 (2003).
Hermansen, S. K., Christensen, K. G., Jensen, S. S. & Kristensen, B. W. Inconsistent immunohistochemical expression patterns of four different CD133 antibody clones in glioblastoma. J. Histochem. Cytochem. 59, 391–407 (2011).
Wong-You-Cheong, J. J., Woodward, P. J., Manning, M. A. & Sesterhenn, I. A. From the archives of the AFIP: neoplasms of the urinary bladder: radiologic-pathologic correlation. Radiographics 26, 553–580 (2006).
Iosif, C. S., Batra, S., Ek, A. & Astedt, B. Estrogen receptors in the human female lower uninary tract. Am. J. Obstet. Gynecol. 141, 817–820 (1981).
Blakeman, P. J., Hilton, P. & Bulmer, J. N. Oestrogen and progesterone receptor expression in the female lower urinary tract, with reference to oestrogen status. BJU Int. 86, 32–38 (2000).
Wilson, P. D., Barker, G., Barnard, R. J. & Siddle, N. C. Steroid hormone receptors in the female lower urinary tract. Urol. Int. 39, 5–8 (1984).
Chao, K. S. C., Perez, C. A. & Brady, L. W. Radiation oncology: management decisions 483–493 (Lippincott Williams & Wilkins, Philadelphia, USA, 2002).
Seidal, T., Balaton, A. J. & Battifora, H. Interpretation and quantification of immunostains. Am. J. Surg. Pathol. 25, 1204–1207 (2001).
Leong, F. J. & Leong, A. S. Digital imaging in pathology: theoretical and practical considerations, and applications. Pathology 36, 234–241 (2004).
Lejeune, M. et al. Quantification of diverse subcellular immunohistochemical markers with clinicobiological relevancies: validation of a new computer-assisted image analysis procedure. J. Anat. 212, 868–878 (2008).
Zhang, Y., Zhu, C., Curado, M. P., Zheng, T. & Boyle, P. Changing patterns of bladder cancer in the USA: evidence of heterogeneous disease. BJU Int. 109, 52–56 (2011).
Guey, L. T. et al. Genetic susceptibility to distinct bladder cancer subphenotypes. Eur. Urol. 57, 283–292 (2009).
Speirs, V., Green, C. A. & Shaaban, A. M. Oestrogen receptor beta immunohistochemistry: time to get it right? J. Clin. Pathol. 61, 1150–1152 (2008).
Liu, A. Laser capture microdissection in the tissue biorepository. J. Biomol. Tech. 21, 120–125 (2010).
Cho-Vega, J. H. et al. Combined laser capture microdissection and serial analysis of gene expression from human tissue samples. Mod. Pathol. 18, 577–584 (2005).
Tincello, D. G., Taylor, A. H., Spurling, S. M. & Bell, S. C. Receptor isoforms that mediate estrogen and progestagen action in the female lower urinary tract. J. Urol. 181, 1474–1482 (2009).
Stauffer, S. R. et al. Pyrazole ligands: structure-affinity/activity relationships and estrogen receptor-alpha-selective agonists. J. Med. Chem. 43, 4934–4947 (2000).
Compton, D. R. et al. Pyrazolo[1, 5-a]pyrimidines: estrogen receptor ligands possessing estrogen receptor beta antagonist activity. J. Med. Chem. 47, 5872–5893 (2004).
Sonpavde, G. et al. Efficacy of selective estrogen receptor modulators in nude mice bearing human transitional cell carcinoma. Urology 69, 1221–1226 (2007).
Kim, H. T. et al. Raloxifene, a mixed estrogen agonist/antagonist, induces apoptosis through cleavage of BAD in TSU-PR1 human cancer cells. J. Biol. Chem. 277, 32510–32515 (2002).
Reznikoff, C. A., Loretz, L. J., Christian, B. J., Wu, S. Q. & Meisner, L. F. Neoplastic transformation of SV40-immortalized human urinary tract epithelial cells by in vitro exposure to 3-methylcholanthrene. Carcinogenesis 9, 1427–1436 (1988).
Christian, B. J., Loretz, L. J., Oberley, T. D. & Reznikoff, C. A. Characterization of human uroepithelial cells immortalized in vitro by simian virus 40. Cancer Res. 47, 6066–6073 (1987).
Fedele, C. G. et al. Inositol polyphosphate 4-phosphatase II regulates PI3K/Akt signaling and is lost in human basal-like breast cancers. Proc. Natl Acad. Sci. USA 107, 22231–22236 (2010).
Hodgson, M. C. et al. Decreased expression and androgen regulation of the tumor suppressor gene INPP4B in prostate cancer. Cancer Res. 71, 572–582 (2011).
Gewinner, C. et al. Evidence that inositol polyphosphate 4-phosphatase type II is a tumor suppressor that inhibits PI3K signaling. Cancer Cell 16, 115–125 (2009).
Hopwood, B. & Dalton, S. Cdc45p assembles into a complex with Cdc46p/Mcm5p, is required for minichromosome maintenance, and is essential for chromosomal DNA replication. Proc. Natl Acad. Sci. USA 93, 12309–12314 (1996).
Afanasyeva, E. A. et al. MicroRNA miR-885-5p targets CDK2 and MCM5, activates p53 and inhibits proliferation and survival. Cell Death Differ. 18, 974–984 (2011).
Teng, J., Wang, Z. Y., Prossnitz, E. R. & Bjorling, D. E. The G protein-coupled receptor GPR30 inhibits human urothelial cell proliferation. Endocrinology 149, 4024–4034 (2008).
Sarkar, S. et al. Different combinations of genetic/epigenetic alterations inactivate the p53 and pRb pathways in invasive human bladder cancers. Cancer Res. 60, 3862–3871 (2000).
Zhang, Z. T., Pak, J., Shapiro, E., Sun, T. T. & Wu, X. R. Urothelium-specific expression of an oncogene in transgenic mice induced the formation of carcinoma in situ and invasive transitional cell carcinoma. Cancer Res. 59, 3512–3517 (1999).
Nagao, M., Suzuki, E., Yasuo, K., Yahagi, T. & Seino, Y. Mutagenicity of N-butyl-N-(4-hydroxybutyl)nitrosamine, a bladder carcinogen, and related compounds. Cancer Res. 37, 399–407 (1977).
Miyamoto, H. et al. Promotion of bladder cancer development and progression by androgen receptor signals. J. Natl Cancer Inst. 99, 558–568 (2007).
Fukushima, S., Hirose, M., Tsuda, H., Shirai, T. & Hirao, K. Histological classification of urinary bladder cancers in rats induced by N-butyl-n-(4-hydroxybutyl)nitrosamine. Gann. 67, 81–90 (1976).
Puzio-Kuter, A. M. et al. Inactivation of p53 and Pten promotes invasive bladder cancer. Genes Dev. 23, 675–680 (2009).
Martensson, U. E. et al. Deletion of the G protein-coupled receptor 30 impairs glucose tolerance, reduces bone growth, increases blood pressure, and eliminates estradiol-stimulated insulin release in female mice. Endocrinology 150, 687–698 (2009).
Yates, M. A., Li, Y., Chlebeck, P. J. & Offner, H. GPR30, but not estrogen receptor-alpha, is crucial in the treatment of experimental autoimmune encephalomyelitis by oral ethinyl estradiol. BMC Immunol. 11, 20 (2010).
Chu, H. A. & Crawford-Brown, D. Inorganic arsenic in drinking water and bladder cancer: a meta-analysis for dose-response assessment. Int. J. Environ. Res. Public Health 4, 340–341 (2007).
Waalkes, M. P., Liu, J., Ward, J. M. & Diwan, B. A. Enhanced urinary bladder and liver carcinogenesis in male CD1 mice exposed to transplacental inorganic arsenic and postnatal diethylstilbestrol or tamoxifen. Toxicol. Appl. Pharmacol. 215, 295–305 (2006).
Waalkes, M. P., Liu, J., Ward, J. M., Powell, D. A. & Diwan, B. A. Urogenital carcinogenesis in female CD1 mice induced by in utero arsenic exposure is exacerbated by postnatal diethylstilbestrol treatment. Cancer Res. 66, 1337–1345 (2006).
Botelho, M. C. et al. Inactivation of estrogen receptor by Schistosoma haematobium total antigen in bladder urothelial cells. Oncol. Rep. 27, 356–362 (2011).
Botelho, M. C., Machado, J. C., Brindley, P. J. & Correia da Costa, J. M. Targeting molecular signaling pathways of Schistosoma haemotobium infection in bladder cancer. Virulence 2, 267–279 (2011).
Davey, J. C., Bodwell, J. E., Gosse, J. A. & Hamilton, J. W. Arsenic as an endocrine disruptor: effects of arsenic on estrogen receptor-mediated gene expression in vivo and in cell culture. Toxicol. Sci. 98, 75–86 (2007).
Anghel, A. et al. Estrogen receptor alpha polymorphisms and the risk of malignancies. Pathol. Oncol. Res. 16, 485–496 (2010).
Amonkar, P., Murali, G. & Krishnamurthy, S. Schistosoma induced squamous cell carcinoma of the bladder. Indian J. Pathol. Microbiol. 44, 363–364 (2001).
Ghisletti, S., Meda, C., Maggi, A. & Vegeto, E. 17beta-estradiol inhibits inflammatory gene expression by controlling NF-kappaB intracellular localization. Mol. Cell Biol. 25, 2957–2968 (2005).
Galea, E. et al. Estrogen inhibits NF kappa B-dependent inflammation in brain endothelium without interfering with I kappa B degradation. Neuroreport 13, 1469–1472 (2002).
Giraud, S. N., Caron, C. M., Pham-Dinh, D., Kitabgi, P. & Nicot, A. B. Estradiol inhibits ongoing autoimmune neuroinflammation and NFkappaB-dependent CCL2 expression in reactive astrocytes. Proc. Natl Acad. Sci. USA 107, 8416–8421 (2010).
Martinez-Ferrer, M. et al. Role of nicotinic and estrogen signaling during experimental acute and chronic bladder inflammation. Am. J. Pathol. 172, 59–67 (2008).
Steele, R. J., Eremin, O., Brown, M. & Hawkins, R. A. Oestrogen receptor concentration and macrophage infiltration in human breast cancer. Eur. J. Surg. Oncol. 12, 273–276 (1986).
Chavey, C. et al. Oestrogen receptor negative breast cancers exhibit high cytokine content. Breast Cancer Res. 9, R15 (2007).
Volodko, N., Reiner, A., Rudas, M. & Jakesz, R. Tumour-associated macrophages in breast cancer and their prognostic correlations. The Breast 7, 99–105 (1998).
Netto, G. J. Molecular biomarkers in urothelial carcinoma of the bladder: are we there yet? Nat. Rev. Urol. 9, 41–51 (2011).
Esrig, D. et al. p53 nuclear protein accumulation correlates with mutations in the p53 gene, tumor grade, and stage in bladder cancer. Am. J. Pathol. 143, 1389–1397 (1993).
Esrig, D. et al. Accumulation of nuclear p53 and tumor progression in bladder cancer. N. Engl. J. Med. 331, 1259–1264 (1994).
Sayeed, A. et al. Estrogen receptor alpha inhibits p53-mediated transcriptional repression: implications for the regulation of apoptosis. Cancer Res. 67, 7746–7755 (2007).
Konduri, S. D. et al. Mechanisms of estrogen receptor antagonism toward p53 and its implications in breast cancer therapeutic response and stem cell regulation. Proc. Natl Acad. Sci. USA 107, 15081–15086 (2010).
Liu, G., Schwartz, J. A. & Brooks, S. C. Estrogen receptor protects p53 from deactivation by human double minute-2. Cancer Res. 60, 1810–1814 (2000).
Berger, C. E., Qian, Y., Liu, G., Chen, H. & Chen, X. p53, a target of estrogen receptor (ER) alpha, modulates DNA damage-induced growth suppression in ER-positive breast cancer cells. J. Biol. Chem. 287, 30117–30127 (2012).
Menendez, D., Inga, A. & Resnick, M. A. Estrogen receptor acting in cis enhances WT and mutant p53 transactivation at canonical and noncanonical p53 target sequences. Proc. Natl Acad. Sci. USA 107, 1500–1505 (2010).
Lewandowski, S. A. et al. Opposite effects of estrogen receptors alpha and beta on MCF-7 sensitivity to the cytotoxic action of TNF and p53 activity. Oncogene 24, 4789–4798 (2005).
Kaufmann, O., Baume, H. & Dietel, M. Detection of oestrogen receptors in non-invasive and invasive transitional cell carcinomas of the urinary bladder using both conventional immunohistochemistry and the tyramide staining amplification (TSA) technique. J. Pathol. 186, 165–168 (1998).
Han, B., Cui, D., Jing, Y., Hong, Y. & Xia, S. Estrogen receptor beta (ERbeta) is a novel prognostic marker of recurrence survival in non-muscle-invasive bladder cancer potentially by inhibiting cadherin switch. World J. Urol. 6, 861–867 (2013).
Acknowledgements
The authors were partly supported in this work by the National Cancer Institute grant CA137474.
Author information
Authors and Affiliations
Contributions
I. Hsu and S. Yeh researched, wrote, edited, and discussed the article with colleagues. S. Vitkus made substantial contributions to discussions of content and reviewed the article prior to submission. J. Da researched data for the article and also made substantial contributions to discussions of content.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Hsu, I., Vitkus, S., Da, J. et al. Role of oestrogen receptors in bladder cancer development. Nat Rev Urol 10, 317–326 (2013). https://doi.org/10.1038/nrurol.2013.53
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrurol.2013.53
This article is cited by
-
The sex gap in bladder cancer survival — a missing link in bladder cancer care?
Nature Reviews Urology (2024)
-
Geschlechtsspezifische Unterschiede spezieller Tumorerkrankungen
Die Innere Medizin (2023)
-
Sex differences in bladder cancer: emerging data and call to action
Nature Reviews Urology (2022)
-
Expression profiles, biological functions and clinical significance of circRNAs in bladder cancer
Molecular Cancer (2021)
-
ERα-mediated alterations in circ_0023642 and miR-490-5p signaling suppress bladder cancer invasion
Cell Death & Disease (2019)