Abstract
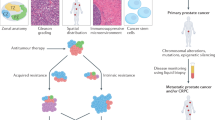
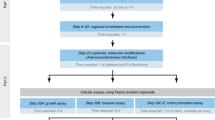
Although an array of new therapeutics exist for prostate cancer, the development of agents that can improve outcomes for men with prostate cancer remains inefficient, costly, and frustratingly slow. A major impediment to the clinical translation of research findings is the lack of preclinical models that can accurately predict the clinical efficacy of new drugs and, therefore, enable the selection of agents that are most suitable for clinical trials. An approach that is gaining popularity in the prostate cancer community is ex vivo culture of primary human tissues, which retains the native tissue architecture, hormone responsiveness, and cell-to-cell signalling of the tumour microenvironment in a dynamic and manipulable state. Ex vivo culture systems recapitulate the structural complexity and heterogeneity of human prostate cancers in a laboratory setting, making them an important adjunct to current cell-line-based and animal-based models. When incorporated into preclinical studies, ex vivo cultured tissues enable robust quantitative evaluation of clinically relevant end points representing drug efficacy, investigation of therapy resistance, and biomarker discovery. By providing new clinically relevant insights into prostate carcinogenesis, it is likely that ex vivo culture will enhance drug development programmes and improve the translational 'hit rate' for prostate cancer research.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Kamb, A., Wee, S. & Lengauer, C. Why is cancer drug discovery so difficult? Nat. Rev. Drug Discov. 6, 115–120 (2007).
Huggins, C., Stephens, R. C. & Hodges, C. V. Studies on prostatic cancer: the effects of castration on advanced carcinoma of the prostate gland. Arch. Surg. 43, 209 (1941).
Tannock, I. F. et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N. Engl. J. Med. 351, 1502–1512 (2004).
de Bono, J. S. et al. Prednisone plus cabazitaxel or mitoxantrone for metastatic castration-resistant prostate cancer progressing after docetaxel treatment: a randomised open-label trial. Lancet 376, 1147–1154 (2010).
de Bono, J. S. et al. Abiraterone and increased survival in metastatic prostate cancer. N. Engl. J. Med. 364, 1995–2005 (2011).
Scher, H. I. et al. Increased survival with enzalutamide in prostate cancer after chemotherapy. N. Engl. J. Med. 367, 1187–1197 (2012).
Adams, D. J. The Valley of Death in anticancer drug development: a reassessment. Trends Pharmacol. Sci. 33, 173–180 (2012).
Swartz, M. A. et al. Tumour microenvironment complexity: emerging roles in cancer therapy. Cancer Res. 72, 2473–2480 (2012).
Ricke, E. A. et al. Androgen hormone action in prostatic carcinogenesis: stromal androgen receptors mediate prostate cancer progression, malignant transformation and metastasis. Carcinogenesis 33, 1391–1398 (2012).
Matrisian, L. M., Cunha, G. R. & Mohla, S. Epithelial–stromal interactions and tumour progression: meeting summary and future directions. Cancer Res. 61, 844–846 (2001).
Johnson, J. I. et al. Relationships between drug activity in NCI preclinical in vitro and in vivo models and early clinical trials. Br. J. Cancer 84, 1424–1431 (2001).
Voskoglou-Nomikos, T., Pater, J. L. & Seymour, L. Clinical predictive value of the in vitro cell line, human xenograft, and mouse allograft preclinical cancer models. Clin. Cancer Res. 9, 227–239 (2003).
Toivanen, R., Taylor, R. A., Pook, D. W., Ellem, S. J. & Risbridger, G. P. Breaking through a roadblock in prostate cancer research: an update on human model systems. J. Steroid Biochem. Mol. Biol. 131, 122–131 (2012).
DeRose, Y. S. et al. Tumour grafts derived from women with breast cancer authentically reflect tumour pathology, growth, metastasis and disease outcomes. Nat. Med. 17, 1514–1520 (2011).
Lopez-Barcons, L. A. Human prostate cancer heterotransplants: a review on this experimental model. Asian J. Androl. 12, 509–518 (2010).
Breslin, S. & O'Driscoll, L. Three-dimensional cell culture: the missing link in drug discovery. Drug Discov. Today 18, 240–249 (2012).
Gerlinger, M. et al. Intratumour heterogeneity and branched evolution revealed by multiregion sequencing. N. Engl. J. Med. 366, 883–892 (2012).
Pretlow, T. G., Yang, B. & Pretlow, T. P. Organ culture of benign, aging, and hyperplastic human prostate. Microsc. Res. Tech. 30, 271–281 (1995).
Varani, J., Dame, M. K., Wojno, K., Schuger, L. & Johnson, K. J. Characteristics of nonmalignant and malignant human prostate in organ culture. Lab. Invest. 79, 723–731 (1999).
Ni, X. et al. Prostate-targeted radiosensitization via aptamer-shRNA chimeras in human tumour xenografts. J. Clin. Invest. 121, 2383–2390 (2011).
McRae, C. U., Ghanadian, R., Fotherby, K. & Chisholm, G. D. The effect of testosterone on the human prostate in organ culture. Br. J. Urol. 45, 156–162 (1973).
Parrish, A. R. et al. Culturing precision-cut human prostate slices as an in vitro model of prostate pathobiology. Cell Biol. Toxicol. 18, 205–219 (2002).
Kiviharju-af Hallstrom, T. M. et al. Human prostate epithelium lacks Wee1A-mediated DNA damage-induced checkpoint enforcement. Proc. Natl Acad. Sci. USA 104, 7211–7216 (2007).
Centenera, M. M. et al. Evidence for efficacy of new Hsp90 inhibitors revealed by ex vivo culture of human prostate tumours. Clin. Cancer Res. 18, 3562–3570 (2012).
Schiewer, M. J. et al. Dual roles of PARP-1 promote cancer growth and progression. Cancer Discov. 2, 1134–1149 (2012).
Bray, K. et al. Bcl-2 modulation to activate apoptosis in prostate cancer. Mol. Cancer Res. 7, 1487–1496 (2009).
Geller, J. et al. Genistein inhibits the growth of human-patient BPH and prostate cancer in histoculture. Prostate 34, 75–79 (1998).
Papini, S. et al. Establishment of an organotypic in vitro culture system and its relevance to the characterization of human prostate epithelial cancer cells and their stromal interactions. Pathol. Res. Pract. 203, 209–216 (2007).
Nevalainen, M. T. et al. Hormone regulation of human prostate in organ culture. Cancer Res. 53, 199–207 (1993).
Geller, J., Sionit, L. R., Connors, K. & Hoffman, R. M. Measurement of androgen sensitivity in the human prostate in in vitro three-dimensional histoculture. Prostate 21, 269–278 (1992).
Jaamaa, S. et al. DNA damage recognition via activated ATM and p53 pathway in nonproliferating human prostate tissue. Cancer Res. 70, 630–641 (2010).
Castells, M., Thibault, B., Delord, J. P. & Couderc, B. Implication of tumour microenvironment in chemoresistance: tumour-associated stromal cells protect tumour cells from cell death. Int. J. Mol. Sci. 13, 545–571 (2012).
Hallden, G. Adenoviral gene expression and replication in human tumour explant models. Methods Mol. Biol. 797, 65–78 (2012).
Scher, H. I. et al. Design and end points of clinical trials for patients with progressive prostate cancer and castrate levels of testosterone: recommendations of the Prostate Cancer Clinical Trials Working Group. J. Clin. Oncol. 26, 1148–1159 (2008).
Collette, L. et al. Is prostate-specific antigen a valid surrogate end point for survival in hormonally treated patients with metastatic prostate cancer? Joint research of the European Organization for Research and Treatment of Cancer, the Limburgs Universitair Centrum, and AstraZeneca Pharmaceuticals. J. Clin. Oncol. 23, 139–148 (2005).
Tan, D. S. et al. Biomarker-driven early clinical trials in oncology: a paradigm shift in drug development. Cancer J. 15, 406–420 (2009).
Khleif, S. N., Doroshow, J. H. & Hait, W. N. AACR-FDA-NCI Cancer Biomarkers Collaborative consensus report: advancing the use of biomarkers in cancer drug development. Clin. Cancer Res. 16, 299–318 (2010).
Stonington, O. G. & Haemmingsen, H. Culture of cells as a monolayer derived from the epithelium of the human prostate: a new cell growth technique. J. Urol. 106, 393–400 (1971).
Kreisberg, J. I., Brattain, M. G. & Pretlow, T. G. Studies on human hyperplastic prostates maintained in organ culture. Invest. Urol. 15, 252–255 (1977).
Sanefuji, H., Heatfield, B. M., Trump, B. F. & Young, J. D. Jr. Studies on carcinogenesis of human prostate. II. Long-term explant culture of normal prostate and benign prostatic hyperplasia: light microscopy. J. Natl Cancer Inst. 69, 751–756 (1982).
Jones, E. G. & Harper, M. E. Studies on the proliferation, secretory activities, and epidermal growth factor receptor expression in benign prostatic hyperplasia explant cultures. Prostate 20, 133–149 (1992).
Nakhla, A. M., Khan, M. S., Romas, N. P. & Rosner, W. Oestradiol causes the rapid accumulation of cAMP in human prostate. Proc. Natl Acad. Sci. USA 91, 402–405 (1994).
Olbina, G., Miljkovic, D., Hoffman, R. M. & Geller, J. New sensitive discovery histoculture model for growth-inhibition studies in prostate cancer and BPH. Prostate 37, 126–129 (1998).
Geller, J. et al. Comparison of androgen-independent growth and androgen-dependent growth in BPH and cancer tissue from the same radical prostatectomies in sponge-gel matrix histoculture. Prostate 31, 250–254 (1997).
Schrodt, G. R. & Foreman, C. D. In vitro maintenance of human hyperplastic prostate tissue. Invest. Urol. 9, 85–94 (1971).
McMahon, M. J., Butler, A. V. & Thomas, G. H. Morphological responses of prostatic carcinoma to testosterone in organ culture. Br. J. Cancer 26, 388–394 (1972).
McMahon, M. J. & Thomas, G. H. Morphological changes of benign prostatic hyperplasia in culture. Br. J. Cancer 27, 323–335 (1973).
Mistry, D., Weaver, J. P. & Riches, A. Organ culture studies of human prostatic adenocarcinomas. Prostate 4, 307–314 (1983).
Eigeliene, N., Harkonen, P. & Erkkola, R. Effects of oestradiol and medroxyprogesterone acetate in morphology, proliferation and apoptosis of human breast tissue in organ cultures. BMC Cancer 6, 246–259 (2006).
Acknowledgements
This work was supported by grants from the National Health and Medical Research Council of Australia (627185 and 1008349), Cancer Australia (627229), the Prostate Cancer Foundation of Australia (NDDA2711), the Royal Adelaide Hospital Research Committee, the Dorothy and James Cleo Thompson Foundation, National Institute of Health (R01 CA116777-05, R01 CA099996-09, and R01 ES016675-11). The Adelaide Prostate Cancer Centre is supported by an establishment grant from the Prostate Cancer Foundation of Australia (2011/0452). M. M. Centenera holds a Young Investigator Award from the Prostate Cancer Foundation of Australia (YI0412).
Author information
Authors and Affiliations
Contributions
M. M. Centenera and L. M. Butler researched, wrote, discussed, and edited the article. G. V. Raj, K. E. Knudsen, and W. D. Tilley contributed towards discussions of contents, edited, and reviewed the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Centenera, M., Raj, G., Knudsen, K. et al. Ex vivo culture of human prostate tissue and drug development. Nat Rev Urol 10, 483–487 (2013). https://doi.org/10.1038/nrurol.2013.126
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrurol.2013.126
This article is cited by
-
Schwann cells regulate tumor cells and cancer-associated fibroblasts in the pancreatic ductal adenocarcinoma microenvironment
Nature Communications (2023)
-
Use of patient-derived explants as a preclinical model for precision medicine in colorectal cancer: A scoping review
Langenbeck's Archives of Surgery (2023)
-
Glutamine addiction promotes glucose oxidation in triple-negative breast cancer
Oncogene (2022)
-
The diversity and breadth of cancer cell fatty acid metabolism
Cancer & Metabolism (2021)
-
Patient derived organoids in prostate cancer: improving therapeutic efficacy in precision medicine
Molecular Cancer (2021)