Abstract
Although the routine use of serum PSA testing has undoubtedly increased prostate cancer detection, one of its main drawbacks has been its lack of specificity, which results in a high negative biopsy rate. Consequently, a large population of men with chronically elevated serum PSA and one or more negative biopsies has emerged. More accurate tests are needed that can help identify which patients are at high risk of developing prostate cancer, and for whom repeat prostate biopsies are mandatory. To improve the specificity of prostate cancer diagnosis, prostate-cancer-specific markers, such as prostate cancer gene 3 (PCA3), are needed. The strong association between PCA3 mRNA overexpression and malignant transformation of prostate epithelium indicates its potential as a diagnostic biomarker. Quantification of PCA3 mRNA levels in urine was found to help predict the outcome of prostate biopsies. The intensive and time-consuming reverse-transcriptase polymerase chain reaction PCA3 urine test has been translated successfully into the fast and easy transcription-mediated amplification (TMA)-based PCA3 test. This test is the first RNA-based molecular diagnostic assay in body fluids for prostate cancer that is available to urologists. This Review describes the translation of the molecular marker PCA3 from the research laboratory to clinical practice.
Key Points
-
Unlike serum PSA, PCA3 is specific for prostate cancer and is not affected by prostate volume or other non-cancerous prostate conditions
-
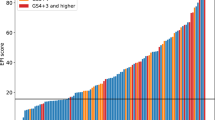
The PCA3 score correlates with the result of a subsequent biopsy; in men with a high PCA3 score, the likelihood of a positive prostate biopsy is increased
-
Preliminary data demonstrate a correlation between PCA3 score and prostate cancer prognostic markers; therefore, PCA3 might identify men who have clinically insignificant prostate cancer and are candidates for active surveillance
-
The PCA3 test is the first fully translated molecular assay in body fluids that is a valuable tool in predicting biopsy outcome
-
Combining PCA3 as a continuous variable with other clinical or pathological factors or biomarkers (for example, TMPRSS2–ERG gene fusions) will provide more-accurate prostate cancer diagnosis and prognosis
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Jemal, A. et al. Cancer statistics, 2008. CA Cancer J. Clin. 58, 71–96 (2008).
Ferlay, J. et al. Estimates of the cancer incidence and mortality in Europe in 2006. Ann. Oncol. 18, 581–592 (2007).
Roddam, A. W. et al. Use of prostate-specific antigen (PSA) isoforms for the detection of prostate cancer in men with a PSA level of 2–10 ng/ml: systematic review and meta-analysis. Eur. Urol. 4, 386–399 (2005).
Hessels, D. et al. Applicability of biomarkers in the early diagnosis of prostate cancer. Expert Rev. Mol. Diagn. 4, 513–526 (2004).
Hessels, D. et al. Molecular Diagnostics in Prostate Cancer. EAU Update Series 3, 200–213 (2005).
Bussemakers, M. J. et al. DD3: a new prostate-specific gene, highly overexpressed in prostate cancer. Cancer Res. 59, 5975–5979 (1999).
de Kok, J. B. et al. DD3 (PCA3), a very sensitive and specific marker to detect prostate tumors. Cancer Res. 62, 2695–2698 (2002).
Hessels, D. et al. DD3 (PCA3)-based molecular urine analysis for the diagnosis of prostate cancer. Eur. Urol. 44, 8–15 (2003).
Schalken, J. A. et al. New targets for therapy in prostate cancer: differential display code 3 (DD3 [PCA3]), a highly prostate cancer-specific gene. Urology 62 (5 Suppl. 1), 34–43 (2003).
Iwakiri, J. et al. An analysis of urinary prostate specific antigen before and after radical prostatectomy: evidence for secretion of prostate specific antigen by the periurethral glands. J. Urol. 149, 783–786 (1993).
Meng, F. J. et al. The expression of a variant prostate-specific antigen in human prostate. Cancer Epidemiol. Biomarkers Prev. 11, 305–309 (2002).
van Gils, M. P. et al. Molecular PCA3 diagnostics on prostatic fluid. Prostate 67, 881–887 (2007).
van Gils, M. P. et al. The time-resolved fluorescence-based PCA3 test on urinary sediments after digital rectal examination; a Dutch multicenter validation of the diagnostic performance. Clin. Cancer Res. 13, 939–943 (2007).
Groskopf, J. et al. APTIMA PCA3 molecular urine test: development of a method to aid in the diagnosis of prostate cancer. Clin. Chem. 52, 1089–1095 (2006).
Sokoll, L. J. et al. A multicenter evaluation of the PCA3 molecular urine test: pre-analytical effects, analytical performance, and diagnostic accuracy. Clin. Chim. Acta 389, 1–6 (2008).
Shappell, S. B. et al. PCA3 urine mRNA testing for prostate carcinoma: patterns of use by community urologists and assay performance in reference laboratory setting. Urology 73, 363–368 (2008).
Marks, L. S. et al. PCA3 molecular urine assay for prostate cancer in men undergoing repeat biopsy. Urology 69, 532–535 (2007).
Deras, I. L. et al. PCA3: a molecular urine assay for predicting prostate biopsy outcome. J. Urol. 179, 1587–1592 (2008).
Haese, A. et al. Clinical utility of the PCA3 urine assay in European men scheduled for repeat biopsy. Eur. Urol. 54, 1081–1088 (2008).
Ankerst, D. P. et al. Predicting prostate cancer risk through incorporation of prostate cancer gene 3. J. Urol. 180, 1303–1308 (2008).
Tomlins, S. A. et al. Recurrent fusion of TMPRSS2 and ETS transcription factor genes in prostate cancer. Science 310, 644–648 (2005).
Tomlins, S. A. et al. TMPRSS2:ETV4 gene fusions define a third molecular subtype of prostate cancer. Cancer Res. 66, 3396–3400 (2006).
Hessels, D. et al. Detection of TMPRSS2–ERG fusion transcripts and prostate cancer antigen 3 in urinary sediments may improve diagnosis of prostate cancer. Clin. Cancer Res. 13, 5103–5108 (2007).
Laxman, B. et al. A first-generation multiplex biomarker analysis of urine for the early detection of prostate cancer. Cancer Res. 68, 645–649 (2008).
van Gils, M. P. et al. Detailed analysis of histopathological parameters in radical prostatectomy specimens and PCA3 urine test results. Prostate 68, 1215–1222 (2008).
Nakanishi, H. et al. PCA3 molecular urine assay correlates with prostate cancer tumor volume: implication in selecting candidates for active surveillance. J. Urol. 179, 1804–1809 (2008).
Whitman, E. J. et al. PCA3 score before radical prostatectomy predicts extracapsular extension and tumor volume. J. Urol. 180, 1975–1978 (2008).
Acknowledgements
The authors would like to thank Dr. Gerald W. Verhaegh (Experimental Urology, Radboud University Nijmegen Medical Centre) and Frank P. Smit (NovioGendix, Nijmegen) for their assistance in critically reading this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
J. A. Schalken has been a speaker for Gen-Probe Inc.
NovioGendix is specialized in molecular diagnostics and offers services (e.g. the PCA3 test) to urologists and clinicians throughout Europe. Daphne Hessels is an employee of NovioGendix.
Rights and permissions
About this article
Cite this article
Hessels, D., Schalken, J. The use of PCA3 in the diagnosis of prostate cancer. Nat Rev Urol 6, 255–261 (2009). https://doi.org/10.1038/nrurol.2009.40
Issue Date:
DOI: https://doi.org/10.1038/nrurol.2009.40
This article is cited by
-
The dynamic role of platelets in cancer progression and their therapeutic implications
Nature Reviews Cancer (2024)
-
Long non-coding RNA ENST00000503625 is a potential prognostic biomarker and metastasis suppressor gene in prostate cancer
Journal of Cancer Research and Clinical Oncology (2023)
-
PCA3 controls chromatin organization and p53 signal activation by regulating LAP2α-lamin A complexes
Cancer Gene Therapy (2022)
-
Intermittent Hormone Therapy Models Analysis and Bayesian Model Comparison for Prostate Cancer
Bulletin of Mathematical Biology (2022)
-
Silencing of TP73-AS1 impairs prostate cancer cell proliferation and induces apoptosis via regulation of TP73
Molecular Biology Reports (2022)