Key Points
-
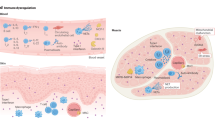
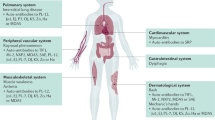
The primary assessment of myositis includes core set measures of disease activity and disease damage and measures of patient-reported outcomes.
-
A new composite myositis response criterion, which combines and differentially weights core set activity measures to determine minimal, moderate and major clinical response, has been developed and validated.
-
Measures of muscle strength and function are being refined for myositis subgroups and are being used as primary or secondary outcome measures in studies and clinical trials in patients with myositis.
-
Disease-specific patient-reported outcomes, including health-related quality of life measures that reflect patient perspectives, are being developed.
-
Imaging and immunological biomarkers provide objective measures that can discriminate disease activity from disease damage, but they still require validation in clinical trials.
Abstract
The adult and juvenile myositis syndromes, commonly referred to collectively as idiopathic inflammatory myopathies (IIMs), are systemic autoimmune diseases with the hallmarks of muscle weakness and inflammation. Validated, well-standardized measures to assess disease activity, known as core set measures, were developed by international networks of myositis researchers for use in clinical trials. Composite response criteria using weighted changes in the core set measures of disease activity were developed and validated for adult and juvenile patients with dermatomyositis and adult patients with polymyositis, with different thresholds for minimal, moderate and major improvement in adults and juveniles. Additional measures of muscle strength and function are being validated to improve content validity and sensitivity to change. A health-related quality of life measure, which incorporates patient input, is being developed for adult patients with IIM. Disease state criteria, including criteria for inactive disease and remission, are being used as secondary end points in clinical trials. MRI of muscle and immunological biomarkers are promising approaches to discriminate between disease activity and damage and might provide much-needed objective outcome measures. These advances in the assessment of outcomes for myositis treatment, along with collaborations between international networks, should facilitate further development of new therapies for patients with IIM.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Miller, F. W. et al. Proposed preliminary core set measures for disease outcome assessment in adult and juvenile idiopathic inflammatory myopathies. Rheumatology 40, 1262–1273 (2001).
Ruperto, N. & Martini, A. Networking in paediatrics: the example of the Paediatric Rheumatology International Trials Organisation (PRINTO). Arch. Dis. Child. 96, 596–601 (2011).
Rider, L. G. et al. Measures of adult and juvenile dermatomyositis, polymyositis, and inclusion body myositis. Arthritis Care Res. 63 (Suppl. 11), S118–S157 (2011).
Ruperto, N. et al. The provisional Paediatric Rheumatology International Trials Organisation/American College of Rheumatology/European League Against Rheumatism Disease activity core set for the evaluation of response to therapy in juvenile dermatomyositis: a prospective validation study. Arthritis Rheum. 59, 4–13 (2008).
Rose, M. R. 188th ENMC International Workshop: Inclusion Body Myositis, 2–4 December 2011, Naarden, The Netherlands. Neuromuscul. Disord. 23, 1044–1055 (2013).
Oddis, C. V. et al. Rituximab in the treatment of refractory adult and juvenile dermatomyositis and adult polymyositis: a randomized, placebo-phase trial. Arthritis Rheum. 65, 314–324 (2013).
Ruperto, N. et al. Paediatric Rheumatology International Trials Organisation (PRINTO). Prednisone versus prednisone plus ciclosporin versus prednisone plus methotrexate in new-onset juvenile dermatomyositis: a randomised trial. Lancet 387, 671–678 (2016).
Muscle Study Group. A randomized, pilot trial of etanercept in dermatomyositis. Ann. Neurol. 70, 427–436 (2011).
Zong, M. et al. Anakinra treatment in patients with refractory inflammatory myopathies and possible predictive response biomarkers: a mechanistic study with 12 months follow-up. Ann. Rheum. Dis. 73, 913–920 (2014).
Tjarnlund, A. et al. Abatacept in the treatment of adult dermatomyositis and polymyositis: a randomised, phase IIb treatment delayed-start trial. Ann. Rheum. Dis. 77, 55–62 (2018).
Rider, L. G. et al. 2016 ACR-EULAR adult dermatomyositis and polymyositis and juvenile dermatomyositis response criteria-methodological aspects. Rheumatology 56, 1884–1893 (2017).
Benveniste, O. & Rider, L. G. 213th ENMC International Workshop: Outcome measures and clinical trial readiness in idiopathic inflammatory myopathies, Heemskerk, The Netherlands, 18–20 September 2015. Neuromuscul. Disord. 26, 523–534 (2016).
McCann, L. J. et al. Development of an optimal core dataset in juvenile dermatomyositis for clinical use to inform research. Ann. Rheum. Dis. 77, 241–250 (2018).
Singh, J. A. et al. Development of classification and response criteria for rheumatic diseases. Arthritis Rheum. 55, 348–352 (2006).
Rider, L. G. et al. International consensus on preliminary definitions of improvement in adult and juvenile myositis. Arthritis Rheum. 50, 2281–2290 (2004).
Ruperto, N. et al. The Pediatric Rheumatology International Trials Organization provisional criteria for the evaluation of response to therapy in juvenile dermatomyositis. Arthritis Care Res. 62, 1533–1541 (2010).
Hansen, P. & Ombler, F. A new method for scoring additive multi-attribute value models using pairwise rankings of alternatives. J. Multi. Crit. Decis. Anal. 15, 87–107 (2008).
van den Hoogen, F. et al. 2013 classification criteria for systemic sclerosis: an American College of Rheumatology/European League against Rheumatism collaborative initiative. Arthritis Rheum. 65, 2737–2747 (2013).
Neogi, T. et al. 2015 Gout Classification Criteria: an American College of Rheumatology/European League Against Rheumatism collaborative initiative. Arthritis Rheum. 67, 2557–2568 (2015).
Neogi, T. et al. The 2010 American College of Rheumatology/European League Against Rheumatism classification criteria for rheumatoid arthritis: Phase 2 methodological report. Arthritis Rheum. 62, 2582–2591 (2010).
de Lautour, H. et al. Development of preliminary remission criteria for gout using Delphi and 1000Minds consensus exercises. Arthritis Care Res. 68, 667–672 (2016).
Aggarwal, R. et al. 2016 American College of Rheumatology/European League Against Rheumatism criteria for minimal, moderate, and major clinical response in adult dermatomyositis and polymyositis: An International Myositis Assessment and Clinical Studies Group/Paediatric Rheumatology International Trials Organisation Collaborative Initiative. Arthritis Rheumatol. 69, 898–910 (2017).
Rider, L. G. et al. 2016 American College of Rheumatology/European League Against Rheumatism criteria for minimal, moderate, and major clinical response in juvenile dermatomyositis: An International Myositis Assessment and Clinical Studies Group/Paediatric Rheumatology International Trials Organisation collaborative initiative. Arthritis Rheumatol. 69, 911–923 (2017).
Aggarwal, R. et al. 2016 American College of Rheumatology/European League Against Rheumatism criteria for minimal, moderate, and major clinical response in adult dermatomyositis and polymyositis: An International Myositis Assessment and Clinical Studies Group/Paediatric Rheumatology International Trials Organisation Collaborative Initiative. Ann. Rheum. Dis. 76, 792–801 (2017).
Rider, L. G. et al. 2016 American College of Rheumatology/European League Against Rheumatism criteria for minimal, moderate, and major clinical response in juvenile dermatomyositis: An International Myositis Assessment and Clinical Studies Group/Paediatric Rheumatology International Trials Organisation collaborative initiative. Ann. Rheum. Dis. 76, 782–791 (2017).
American College of Rheumatology Committee to Reevaluate Improvement Criteria. A proposed revision to the ACR20: the hybrid measure of American College of Rheumatology response. Arthritis Rheum. 57, 193–202 (2007).
Harris-Love, M. O. et al. Distribution and severity of weakness among patients with polymyositis, dermatomyositis and juvenile dermatomyositis. Rheumatology 48, 134–139 (2009).
Hogrel, J. Y. et al. Four-year longitudinal study of clinical and functional endpoints in sporadic inclusion body myositis: implications for therapeutic trials. Neuromuscul. Disord. 24, 604–610 (2014).
Allenbach, Y. et al. Quadriceps strength is a sensitive marker of disease progression in sporadic inclusion body myositis. Neuromuscul. Disord. 22, 980–986 (2012).
Albayda, J. et al. Antinuclear matrix protein 2 autoantibodies and edema, muscle disease, and malignancy risk in dermatomyositis patients. Arthritis Care Res. 69, 1771–1776 (2017).
Pinal-Fernandez, I. et al. Longitudinal course of disease in a large cohort of myositis patients with autoantibodies recognizing the signal recognition particle. Arthritis Care Res. 69, 263–270 (2017).
Rider, L. G. et al. Defining clinical improvement in adult and juvenile myositis. J. Rheumatol. 30, 603–617 (2003).
Pinto, A. J. et al. Poor agreement of objectively measured and self-reported physical activity in juvenile dermatomyositis and juvenile systemic lupus erythematosus. Clin. Rheumatol. 35, 1507–1514 (2016).
Rider, L. G. et al. Novel assessment tools to evaluate clinical and laboratory responses in a subset of patients enrolled in the Rituximab in Myositis trial. Clin. Exp. Rheumatol. 32, 689–696 (2014).
Cortese, A. et al. Longitudinal observational study of sporadic inclusion body myositis: implications for clinical trials. Neuromuscul. Disord. 23, 404–412 (2013).
Rider, L. G. et al. Validation of manual muscle testing and a subset of eight muscles for adult and juvenile idiopathic inflammatory myopathies. Arthritis Care Res. 62, 465–472 (2010).
Varnier, G. C. et al. Development and testing of a hybrid measure of muscle strength in juvenile dermatomyositis for use in routine care. Arthritis Care Res. https://doi.org/10.1002/acr.23491 (2017).
Alexanderson, H. et al. Functional index-2: Validity and reliability of a disease-specific measure of impairment in patients with polymyositis and dermatomyositis. Arthritis Rheum. 55, 114–122 (2006).
Lowes, L. P. et al. Knee extensor strength exhibits potential to predict function in sporadic inclusion-body myositis. Muscle Nerve 45, 163–168 (2012).
Watson, M. J. Refining the ten-metre walking test for use with neurologically impaired people. Physiotherapy 88, 386–397 (2002).
Podsiadlo, D. & Richardson, S. The timed “Up and Go”: a test of basic functional mobility for frail elderly persons. J. Am. Geriatr. Soc. 39, 142–148 (1991).
Jorgensen, A. N. et al. Physical function and muscle strength in sporadic inclusion body myositis. Muscle Nerve 56, E50–E58 (2017).
McCarthy, E. K., Horvat, M. A., Holtsberg, P. A. & Wisenbaker, J. M. Repeated chair stands as a measure of lower limb strength in sexagenarian women. J. Gerontol. A Biol. Sci. Med. Sci. 59, 1207–1212 (2004).
Agarwal, S. & Kiely, P. D. Two simple, reliable and valid tests of proximal muscle function, and their application to the management of idiopathic inflammatory myositis. Rheumatology 45, 874–879 (2006).
Freire, A. N., Guerra, R. O., Alvarado, B., Guralnik, J. M. & Zunzunegui, M. V. Validity and reliability of the short physical performance battery in two diverse older adult populations in Quebec and Brazil. J. Aging Health 24, 863–878 (2012).
Ostir, G. V., Volpato, S., Fried, L. P., Chaves, P. & Guralnik, J. M. Reliability and sensitivity to change assessed for a summary measure of lower body function: results from the Women's Health and Aging Study. J. Clin. Epidemiol. 55, 916–921 (2002).
Amato, A. A. et al. A randomized, double-blind, placebo-controlled study of bimagrumab in patients with sporadic inclusion body myositis [abstract]. Arthritis Rheumatol. 68 (Suppl 10), 8L (2016).
Alfano, L. N. et al. The 2-min walk test is sufficient for evaluating walking abilities in sporadic inclusion body myositis. Neuromuscul. Disord. 24, 222–226 (2014).
Jimenez-Moreno, A. C. et al. Measuring habitual physical activity in neuromuscular disorders: a systematic review. J. Neuromuscul. Dis. 4, 25–52 (2017).
Bachasson, D., Landon-Cardinal, O., Benveniste, O., Hogrel, J. Y. & Allenbach, Y. Physical activity monitoring: a promising outcome measure in idiopathic inflammatory myopathies. Neurology 89, 101–103 (2017).
Ruperto, N. et al. Cross-cultural adaptation and psychometric evaluation of the Childhood Health Assessment Questionnaire (CHAQ) and the Child Health Questionnaire (CHQ) in 32 countries. Review of the general methodology. Clin. Exp. Rheumatol. 19 (Suppl. 23), S1–S9 (2001).
Leclair, V., Regardt, M., Wojcik, S. & Hudson, M. Health-related quality of life (HRQoL) in idiopathic inflammatory myopathy: A systematic review. PLoS ONE 11, e0160753 (2016).
Apaz, M. T. et al. Health-related quality of life of patients with juvenile dermatomyositis: results from the Pediatric Rheumatology International Trials Organisation multinational quality of life cohort study. Arthritis Rheum. 61, 509–517 (2009).
van Tuyl, L. H. D. & Boers, M. Patient-reported outcomes in core domain sets for rheumatic diseases. Nat. Rev. Rheumatol. 11, 705–712 (2015).
Feldon, M. et al. Predictors of reduced health-related quality of life in adult patients with idiopathic inflammatory myopathies. Arthritis Care Res. 69, 1743–1750 (2017).
Guyatt, G. H., Feeny, D. H. & Patrick, D. L. Measuring health-related quality of life. Ann. Intern. Med. 118, 622–629 (1993).
Poulsen, K. B. et al. Quality of life correlates with muscle strength in patients with dermato- or polymyositis. Clin. Rheumatol. 36, 2289–2295 (2017).
Takken, T. et al. The physiological and physical determinants of functional ability measures in children with juvenile dermatomyositis. Rheumatology 42, 591–595 (2003).
Sadjadi, R. & Rose, M. R. What determines quality of life in inclusion body myositis? J. Neurol. Neurosurg. Psychiatry 81, 1164–1166 (2010).
Alexanderson, H. et al. Patient-reported outcomes and adult patients' disease experience in the idiopathic inflammatory myopathies. report from the OMERACT 11 Myositis Special Interest Group. J. Rheumatol. 41, 581–592 (2014).
Regardt, M. et al. Patients' experience of myositis and further validation of a myositis-specific patient reported outcome measure - establishing core domains and expanding patient input on clinical assessment in myositis. Report from OMERACT 12. J. Rheumatol. 42, 2492–2495 (2015).
Park, J. K. et al. Advancing the development of patient-reported outcomes for adult myositis at OMERACT 2016: an international delphi study. J. Rheumatol. 44, 1683–1687 (2017).
DeMuro, C. et al. Development of the sporadic inclusion body myositis physical functioning assessment. Muscle Nerve 54, 653–657 (2016).
Williams, V. et al. Psychometric validation of a patient-reported measure of physical functioning in sporadic inclusion body myositis. Muscle Nerve 54, 658–665 (2016).
Jackson, C. E., Barohn, R. J., Gronseth, G., Pandya, S. & Herbelin, L. Inclusion body myositis functional rating scale: a reliable and valid measure of disease severity. Muscle Nerve 37, 473–476 (2008).
Ahmed, M. et al. Targeting protein homeostasis in sporadic inclusion body myositis. Sci. Transl Med. 8, 331ra41 (2016).
Saketkoo, L. A. et al. Connective tissue disease related interstitial lung diseases and idiopathic pulmonary fibrosis: provisional core sets of domains and instruments for use in clinical trials. Thorax 69, 428–436 (2014).
Olthoff, A. et al. Evaluation of dysphagia by novel real-time MRI. Neurology 87, 2132–2138 (2016).
Hasija, R. et al. Therapeutic approaches in the treatment of juvenile dermatomyositis in patients with recent-onset disease and in those experiencing disease flare: an international multicenter PRINTO study. Arthritis Rheum. 63, 3142–3152 (2011).
Feldman, B. M., Rider, L. G., Reed, A. M. & Pachman, L. M. Juvenile dermatomyositis and other idiopathic inflammatory myopathies of childhood. Lancet 371, 2201–2212 (2008).
Kim, S. et al. Complete and sustained remission of juvenile dermatomyositis resulting from aggressive treatment. Arthritis Rheum. 60, 1825–1830 (2009).
Lazarevic, D. et al. The PRINTO criteria for clinically inactive disease in juvenile dermatomyositis. Ann. Rheum. Dis. 72, 686–693 (2013).
Almeida, B. et al. Analysis of published criteria for clinically inactive disease in a large juvenile dermatomyositis cohort shows that skin disease is underestimated. Arthritis Rheumatol. 67, 2495–2502 (2015).
Oddis, C. V. et al. International consensus guidelines for trials of therapies in the idiopathic inflammatory myopathies. Arthritis Rheum. 52, 2607–2615 (2005).
Day, J., Patel, S. & Limaye, V. The role of magnetic resonance imaging techniques in evaluation and management of the idiopathic inflammatory myopathies. Semin. Arthritis Rheum. 46, 642–649 (2017).
Filli, L., Winklhofer, S., Andreisek, G. & Del Grande, F. Imaging of myopathies. Radiol. Clin. North Am. 55, 1055–1070 (2017).
Adams, E. M., Chow, C. K., Premkumar, A. & Plotz, P. H. The idiopathic inflammatory myopathies: spectrum of MR imaging findings. Radiographics 15, 563–574 (1995).
Abdul-Aziz, R. et al. Muscle MRI at the time of questionable disease flares in juvenile dermatomyositis (JDM). Pediatr. Rheumatol. Online J. 15, 25 (2017).
Kubinova, K., Mann, H. & Vencovsky, J. MRI scoring methods used in evaluation of muscle involvement in patients with idiopathic inflammatory myopathies. Curr. Opin. Rheumatol. 29, 623–631 (2017).
Andersson, H. et al. Comparative analyses of muscle MRI and muscular function in anti-synthetase syndrome patients and matched controls: a cross-sectional study. Arthritis Res. Ther. 19, 17 (2017).
Barsotti, S. et al. Thigh magnetic resonance imaging for the evaluation of disease activity in patients with idiopathic inflammatory myopathies followed in a single center. Muscle Nerve 54, 666–672 (2016).
Guimaraes, J. B. et al. Sporadic inclusion body myositis: MRI findings and correlation with clinical and functional parameters. AJR Am. J. Roentgenol. 209, 1340–1347 (2017).
Morrow, J. M. et al. MRI biomarker assessment of neuromuscular disease progression: a prospective observational cohort study. Lancet Neurol. 15, 65–77 (2016).
Yao, L. et al. Magnetic resonance measurement of muscle T2, fat-corrected T2 and fat fraction in the assessment of idiopathic inflammatory myopathies. Rheumatology 55, 441–449 (2016).
Malattia, C. et al. MRI versus conventional measures of disease activity and structural damage in evaluating treatment efficacy in juvenile idiopathic arthritis. Ann. Rheum. Dis. 72, 363–368 (2013).
Huang, Z. G. et al. An efficacy analysis of whole-body magnetic resonance imaging in the diagnosis and follow-up of polymyositis and dermatomyositis. PLoS ONE 12, e0181069 (2017).
Manole, E., Bastian, A. E., Butoianu, N. & Goebel, H. H. Myositis non-inflammatory mechanisms: An up-dated review. J. Immunoassay Immunochem. 38, 115–126 (2017).
Olazagasti, J. M., Niewold, T. B. & Reed, A. M. Immunological biomarkers in dermatomyositis. Curr. Rheumatol. Rep. 17, 68 (2015).
Reed, A. M. et al. Changes in novel biomarkers of disease activity in juvenile and adult dermatomyositis are sensitive biomarkers of disease course. Arthritis Rheum. 64, 4078–4086 (2012).
Lopez De Padilla, C. M. et al. Interferon-regulated chemokine score associated with improvement in disease activity in refractory myositis patients treated with rituximab. Clin. Exp. Rheumatol. 33, 655–663 (2015).
Lopez de Padilla, C. M. et al. BAFF expression correlates with idiopathic inflammatory myopathy disease activity measures and autoantibodies. J. Rheumatol. 40, 294–302 (2013).
Gono, T. et al. Cytokine profiles in polymyositis and dermatomyositis complicated by rapidly progressive or chronic interstitial lung disease. Rheumatology 53, 2196–2203 (2014).
Lopez De Padilla, C. M. et al. Gene expression profiling in blood and affected muscle tissues reveals differential activation pathways in patients with new-onset juvenile and adult dermatomyositis. J. Rheumatol. 44, 117–124 (2017).
Kobayashi, N. et al. Increased serum B cell activating factor and a proliferation-inducing ligand are associated with interstitial lung disease in patients with juvenile dermatomyositis. J. Rheumatol. 42, 2412–2418 (2015).
Krystufkova, O. et al. Increased serum levels of B cell activating factor (BAFF) in subsets of patients with idiopathic inflammatory myopathies. Ann. Rheum. Dis. 68, 836–843 (2009).
De Bleecker, J. L., De Paepe, B., Vanwalleghem, I. E. & Schroder, J. M. Differential expression of chemokines in inflammatory myopathies. Neurology 58, 1779–1785 (2002).
O'Connor, K. A., Abbott, K. A., Sabin, B., Kuroda, M. & Pachman, L. M. MxA gene expression in juvenile dermatomyositis peripheral blood mononuclear cells: association with muscle involvement. Clin. Immunol. 120, 319–325 (2006).
Greenberg, S. A. et al. Interferon-alpha/beta-mediated innate immune mechanisms in dermatomyositis. Ann. Neurol. 57, 664–678 (2005).
Reed, A. M. et al. RIM Study Group. Biologic predictors of clinical improvement in rituximab-treated refractory myositis. BMC Musculoskelet. Disord. 16, 257 (2015).
Nara, M. et al. Serum interleukin 6 levels as a useful prognostic predictor of clinically amyopathic dermatomyositis with rapidly progressive interstitial lung disease. Mod. Rheumatol. 24, 633–636 (2014).
Baechler, E. C. et al. An interferon signature in the peripheral blood of dermatomyositis patients is associated with disease activity. Mol. Med. 13, 59–68 (2007).
Huard, C. et al. Correlation of cutaneous disease activity with type 1 interferon gene signature and interferon beta in dermatomyositis. Br. J. Dermatol. 176, 1224–1230 (2017).
Shrestha, S. et al. Lesional and nonlesional skin from patients with untreated juvenile dermatomyositis displays increased numbers of mast cells and mature plasmacytoid dendritic cells. Arthritis Rheum. 62, 2813–2822 (2010).
Walsh, R. J. et al. Type I interferon-inducible gene expression in blood is present and reflects disease activity in dermatomyositis and polymyositis. Arthritis Rheum. 56, 3784–3792 (2007).
Nistala, K. et al. Myeloid related protein induces muscle derived inflammatory mediators in juvenile dermatomyositis. Arthritis Res. Ther. 15, R131 (2013).
Zou, J., Chen, J., Yan, Q., Guo, Q. & Bao, C. Serum IL8 and mRNA level of CD11b in circulating neutrophils are increased in clinically amyopathic dermatomyositis with active interstitial lung disease. Clin. Rheumatol. 35, 117–125 (2016).
Yuan, L. et al. Serum levels of soluble ST2 and interleukin-33 in patients with dermatomyositis and polymyositis. Clin. Exp. Rheumatol. 31, 428–432 (2013).
Bellutti Enders, F. et al. Correlation of CXCL10, tumor necrosis factor receptor type II, and galectin 9 with disease activity in juvenile dermatomyositis. Arthritis Rheumatol. 66, 2281–2289 (2014).
Olazagasti, J. M. et al. Adipokine gene expression in peripheral blood of adult and juvenile dermatomyositis patients and their relation to clinical parameters and disease activity measures. J. Inflamm. 12, 29 (2015).
Bandoh, S. et al. Sequential changes of KL-6 in sera of patients with interstitial pneumonia associated with polymyositis/dermatomyositis. Ann. Rheum. Dis. 59, 257–262 (2000).
Sanner, H. et al. Increased levels of eotaxin and MCP-1 in juvenile dermatomyositis median 16.8 years after disease onset; associations with disease activity, duration and organ damage. PLoS ONE 9, e92171 (2014).
Ishizuka, M. et al. Long-term follow-up of 124 patients with polymyositis and dermatomyositis: statistical analysis of prognostic factors. Mod. Rheumatol. 26, 115–120 (2016).
Preusse, C. et al. Immune-mediated necrotizing myopathy is characterized by a specific Th1-M1 polarized immune profile. Am. J. Pathol. 181, 2161–2171 (2012).
Betteridge, Z. & McHugh, N. Myositis-specific autoantibodies: an important tool to support diagnosis of myositis. J. Intern. Med. 280, 8–23 (2016).
Rider, L. G. & Nistala, K. The juvenile idiopathic inflammatory myopathies: pathogenesis, clinical and autoantibody phenotypes, and outcomes. J. Intern. Med. 280, 24–38 (2016).
Aggarwal, R. et al. Autoantibody levels in myositis patients correlate with clinical response during B cell depletion with rituximab. Rheumatology 55, 991–999 (2016).
McHugh, N.J. & Tansley, S.L. Autoantibodies in myositis Nat. Rev. Rheumatol. https://doi.org/10.1038/nrrheum.2018.56 (2018).
Aggarwal, R. et al. Anti-signal recognition particle autoantibody ELISA validation and clinical associations. Rheumatology 54, 1194–1199 (2015).
Stone, K. B. et al. Anti-Jo-1 antibody levels correlate with disease activity in idiopathic inflammatory myopathy. Arthritis Rheum. 56, 3125–3131 (2007).
Matsushita, T. et al. Antimelanoma differentiation-associated protein 5 antibody level is a novel tool for monitoring disease activity in rapidly progressive interstitial lung disease with dermatomyositis. Br. J. Dermatol. 176, 395–402 (2017).
Aggarwal, R. et al. Autoantibody levels in myositis patients correlate with clinical response during B cell depletion with rituximab. Rheumatology 55, 1710 (2016).
Kobayashi, N. et al. Clinical and laboratory features of fatal rapidly progressive interstitial lung disease associated with juvenile dermatomyositis. Rheumatology 54, 784–791 (2015).
Higgs, B. W. et al. A phase 1b clinical trial evaluating sifalimumab, an anti-IFN-alpha monoclonal antibody, shows target neutralisation of a type I IFN signature in blood of dermatomyositis and polymyositis patients. Ann. Rheum. Dis. 73, 256–262 (2014).
Nagaraju, K. et al. Muscle myeloid type I interferon gene expression may predict therapeutic responses to rituximab in myositis patients. Rheumatology 55, 1673–1680 (2016).
Varsani, H. et al. Validation of a score tool for measurement of histological severity in juvenile dermatomyositis and association with clinical severity of disease. Ann. Rheum. Dis. 74, 204–210 (2015).
Ruperto, N. et al. Preliminary core sets of measures for disease activity and damage assessment in juvenile systemic lupus erythematosus and juvenile dermatomyositis. Rheumatology 42, 1452–1459 (2003).
Rider, L. G. Outcome assessment in the adult and juvenile idiopathic inflammatory myopathies. Rheum. Dis. North. Am. 28, 935–977 (2002).
Beenakker, E. A. C., van der Hoeven, J. H., Fock, J. M. & Maurits, N. M. Reference values of maximum isometric muscle force obtained in 270 children aged 4 - 16 years by hand-held dynamometry. Neuromuscul. Disord. 11, 441–446 (2001).
McKay, M. J. et al. Normative reference values for strength and flexibility of 1,000 children and adults. Neurology 88, 36–43 (2017).
Bohannon, R. W. Literature reporting normative data for muscle strength measured by hand-held dynamometry: a systematic review. Isokinet. Exerc. Sci. 19, 143–147 (2011).
Hogrel, J. Y. et al. Development of a French isometric strength normative database for adults using quantitative muscle testing. Arch. Phys. Med. Rehabil. 88, 1289–1297 (2007).
Harbo, T., Brincks, J. & Andersen, H. Maximal isokinetic and isometric muscle strength of major muscle groups related to age, body mass, height, and sex in 178 healthy subjects. Eur. J. Appl. Physiol. 112, 267–275 (2012).
Danneskiold-Samsoe, B. et al. Isokinetic and isometric muscle strength in a healthy population with special reference to age and gender. Acta Physiol. 197 (Suppl. 673), 1–68 (2009).
Moraux, A. et al. Ankle dorsi- and plantar-flexion torques measured by dynamometry in healthy subjects from 5 to 80 years. BMC Musculoskelet. Disord. 14, 104 (2013).
Stoll, T., Bruhlmann, P., Stucki, G., Seifert, B. & Michel, B. A. Muscle strength assessment in polymyositis and dermatomyositis evaluation of the reliability and clinical use of a new, quantitative, easily applicable method. J. Rheumatol. 22, 473–477 (1995).
Mahler, E. A. et al. Rituximab treatment in patients with refractory inflammatory myopathies. Rheumatology 50, 2206–2213 (2011).
Cox, F. M. et al. Magnetic resonance imaging of skeletal muscles in sporadic inclusion body myositis. Rheumatology 50, 1153–1161 (2011).
Lindberg, C. & Oldfors, A. Prognosis and prognostic factors in sporadic inclusion body myositis. Acta Neurol. Scand. 125, 353–358 (2012).
Alfano, L. N. et al. Modeling functional decline over time in sporadic inclusion body myositis. Muscle Nerve 55, 526–531 (2017).
Neri, R. et al. Functional and isokinetic assessment of muscle strength in patients with idiopathic inflammatory myopathies. Autoimmunity 39, 255–259 (2006).
Benveniste, O. et al. Long-term observational study of sporadic inclusion body myositis. Brain 134, 3176–3184 (2011).
Huber, A. M. et al. Consensus treatments for moderate juvenile dermatomyositis: beyond the first two months. Results of the second Childhood Arthritis and Rheumatology Research Alliance consensus conference. Arthritis Care Res. 64, 546–553 (2012).
Yin, L. et al. The clinical utility of serum IL-35 in patients with polymyositis and dermatomyositis. Clin. Rheumatol. 35, 2715–2721 (2016).
Taylor, K. E. et al. Risk alleles for systemic lupus erythematosus in a large case-control collection and associations with clinical subphenotypes. PLoS Genet. 7, e1001311 (2011).
Tansley, S. L. et al. Autoantibodies in juvenile-onset myositis: Their diagnostic value and associated clinical phenotype in a large UK cohort. J. Autoimmun. 84, 55–64 (2017).
Acknowledgements
The authors thank L. Maroski for assistance with references and tables, M. Ward and P. Grayson for helpful comments on the manuscript and J. Morrow for providing the MRI figure. L.G.R. was supported by the intramural research programme of the US National Institutes of Health, National Institute of Environmental Health Sciences. P.M.M. was supported by the UK National Institute for Health Research (NIHR) University College London Hospitals Biomedical Research Centre. The views expressed are those of the authors and not necessarily those of the UK National Health Service (NHS), the NIHR, the UK Department of Health or the US Department of Health and Human Services.
Author information
Authors and Affiliations
Contributions
All authors researched the data for the article, provided substantial contributions to discussions of its content, wrote the article and reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Table S1
Performance characteristics of existing patient-reported outcomes used in studies of the idiopathic inflammatory myopathies, according to the OMERACT Filter 1.0 (PDF 108 kb)
Glossary
- Patient-reported outcome measures
-
(PROMs). Measurements based on information provided directly by the patient (that is, the study subject) about the status of his or her health condition, without amendment or interpretation of the patient's response by a clinician or anyone else.
- Health-related quality of life
-
(HRQoL). A multidimensional assessment of a subject's health that includes domains related to physical, mental, emotional and social functioning. HRQoL goes beyond direct measures of health, life expectancy and causes of death and focuses on the effect that health status has on quality of life.
- Biomarkers
-
Measurable indicators of normal biological processes, pathogenic processes or responses to an exposure or intervention, including therapeutic interventions.
- Core set measures
-
(CSMs). The minimum set of validated assessments that are recommended to be used in therapeutic trials and natural history studies.
- Response criteria
-
A set of conditions, usually involving combinations of assessment tools, that define clinically important improvement in disease symptoms and signs. These criteria allow investigators, clinicians, regulators and patients to determine the efficacy (or lack thereof) of a therapeutic intervention, and they facilitate communication between these parties about response to treatment by using the same metric.
- Manual muscle testing
-
(MMT). A method for assessing the strength of individual muscle groups on the basis of performing a movement in relation to the forces of gravity and manual resistance by the examiner.
- Dynamometers
-
Devices that measure muscle strength during muscle contraction, such as gripping, pushing and pulling.
- Magnetic resonance imaging
-
(MRI). An imaging technique that uses a magnetic field and radio waves to create detailed images of the organs and tissues within the body.
Rights and permissions
About this article
Cite this article
Rider, L., Aggarwal, R., Machado, P. et al. Update on outcome assessment in myositis. Nat Rev Rheumatol 14, 303–318 (2018). https://doi.org/10.1038/nrrheum.2018.33
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2018.33
This article is cited by
-
Inclusion body myositis—health-related quality of life and care situation during phases of the “patience journey” in Germany: results from a qualitative study
Health and Quality of Life Outcomes (2023)
-
Currently recommended skin scores correlate highly in the assessment of patients with Juvenile Dermatomyositis (JDM)
Pediatric Rheumatology (2023)
-
Treat-to-Target in Pediatric Rheumatic Diseases
Current Rheumatology Reports (2023)
-
Dermatomyositis und juvenile Dermatomyositis
Zeitschrift für Rheumatologie (2023)
-
Treatment-naïve idiopathic inflammatory myopathy: disease evaluation by fluorodeoxyglucose versus pyrophosphate
Egyptian Journal of Radiology and Nuclear Medicine (2022)