Key Points
-
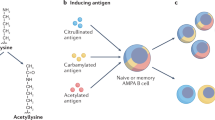
The anti-citrullinated protein antibody (ACPA) response dynamically evolves before the onset of rheumatoid arthritis (RA)
-
HLA-shared epitope alleles and T cells are suggested to be involved in driving the expansion of the ACPA response in the pre-arthritis phase of RA
-
ACPA-IgG molecules are extensively glycosylated in the variable domain because of N-glycosylation sites generated by somatic hypermutation
-
The extensive presence of ACPA-IgG variable domain glycans points to mechanisms by which ACPA-expressing B cells could escape tolerance checkpoints
-
ACPA Fc-glycans potentially function as modulators of inflammation and, via IL-23 and T helper 17 cells, could be involved in the transition from asymptomatic autoimmunity to inflammatory arthritis
-
The frequency and phenotype of ACPA-expressing B cells in the circulation suggest a continuously active B cell immune response that is insufficiently targeted by current therapeutic interventions
Abstract
The immune response to citrullinated antigens is found almost exclusively in patients with rheumatoid arthritis (RA). It is a dynamic response that expands before the onset of disease and generates antibodies (anti-citrullinated protein antibodies (ACPAs)) that are extensively glycosylated in the variable domain. This feature of ACPAs is remarkable and warrants detailed investigation, as it can offer insights into the earliest immunologic mechanisms that lead up to the development of RA. The acquisition of variable domain glycans, in fact, could enable ACPA-expressing B cells to breach tolerance. Although the underlying mechanisms are still elusive, data to support this concept are emerging, owing to the reliable identification and isolation of citrullinated antigen-directed B cells from patients with RA. This technological proficiency also allows for the generation of an increasing number of well-defined monoclonal ACPAs, and provides the opportunity to test and define the mechanisms by which the citrullinated antigen-directed B cell response contributes to the onset and persistence of inflammation. Together with a revised perception of the HLA-risk effect and novel insights into how T cells can govern antibody effector functions, these developments shape an increasingly clear picture of the B cell response to citrullinated antigens in the development of RA.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Pruijn, G. J., Wiik, A. & van Venrooij, W. J. The use of citrullinated peptides and proteins for the diagnosis of rheumatoid arthritis. Arthritis Res. Ther. 12, 203 (2010).
van Gaalen, F. A. et al. Autoantibodies to cyclic citrullinated peptides predict progression to rheumatoid arthritis in patients with undifferentiated arthritis: a prospective cohort study. Arthritis Rheum. 50, 709–715 (2004).
Meyer, O. et al. Anticitrullinated protein/peptide antibody assays in early rheumatoid arthritis for predicting five year radiographic damage. Ann. Rheum. Dis. 62, 120–126 (2003).
van der Linden, M. P. et al. Value of anti-modified citrullinated vimentin and third-generation anti-cyclic citrullinated peptide compared with second-generation anti-cyclic citrullinated peptide and rheumatoid factor in predicting disease outcome in undifferentiated arthritis and rheumatoid arthritis. Arthritis Rheum. 60, 2232–2241 (2009).
Jorgensen, K.T. et al. Cytokines, autoantibodies and viral antibodies in premorbid and postdiagnostic sera from patients with rheumatoid arthritis: case-control study nested in a cohort of Norwegian blood donors. Ann. Rheum. Dis. 67, 860–866 (2009).
Demoruelle, M. K. et al. Performance of anti-cyclic citrullinated peptide assays differs in subjects at increased risk of rheumatoid arthritis and subjects with established disease. Arthritis Rheum. 65, 2243–2252 (2013).
Goeldner, I. et al. Anti-cyclic citrullinated peptide antibodies and rheumatoid factor in rheumatoid arthritis patients and relatives from Brazil. Rheumatol. (Oxford) 49, 1590–1593 (2010).
van Zanten, A. et al. Presence of anticitrullinated protein antibodies in a large population-based cohort from the Netherlands. Ann. Rheum. Dis. 76, 1184–1190 (2017).
Terao, C. et al. Effects of smoking and shared epitope on the production of anti-citrullinated peptide antibody in a Japanese adult population. Arthritis Care Res. 66, 1818–1827 (2014).
Tasliyurt, T. et al. The frequency of antibodies against cyclic citrullinated peptides and rheumatoid factor in healthy population: a field study of rheumatoid arthritis from northern Turkey. Rheumatol. Int. 33, 939–942 (2013).
Hensvold, A. H. et al. Environmental and genetic factors in the development of anticitrullinated protein antibodies (ACPAs) and ACPA-positive rheumatoid arthritis: an epidemiological investigation in twins. Ann. Rheum. Dis. 74, 375–380 (2015).
Nielen, M. M. et al. Specific autoantibodies precede the symptoms of rheumatoid arthritis: a study of serial measurements in blood donors. Arthritis Rheum. 50, 380–386 (2004).
Rantapaa-Dahlqvist, S. et al. Antibodies against cyclic citrullinated peptide and IgA rheumatoid factor predict the development of rheumatoid arthritis. Arthritis Rheum. 48, 2741–2749 (2003).
Majka, D. S. et al. Duration of preclinical rheumatoid arthritis-related autoantibody positivity increases in subjects with older age at time of disease diagnosis. Ann. Rheum. Dis. 67, 801–807 (2008).
van Steenbergen, H. W., Mangnus, L., Reijnierse, M., Huizinga, T. W. & van der Helm-van Mil, A. H. Clinical factors, anticitrullinated peptide antibodies and MRI-detected subclinical inflammation in relation to progression from clinically suspect arthralgia to arthritis. Ann. Rheum. Dis. 75, 1824–1830 (2016).
Nam, J. L., Hunt, L., Hensor, E. M. & Emery, P. Enriching case selection for imminent RA: the use of anti-CCP antibodies in individuals with new non-specific musculoskeletal symptoms - a cohort study. Ann. Rheum. Dis. 75, 1452–1456 (2016).
Bos, W. H. et al. Arthritis development in patients with arthralgia is strongly associated with anti-citrullinated protein antibody status: a prospective cohort study. Ann. Rheum. Dis. 69, 490–494 (2010).
van Steenbergen, H. W. et al. EULAR definition of arthralgia suspicious for progression to rheumatoid arthritis. Ann. Rheum. Dis. 76, 491–496 (2017).
Nienhuis, R. L. & Mandema, E. A. A new serum factor in patients with rheumatoid arthritis; the antiperinuclear factor. Ann. Rheum. Dis. 23, 302–305 (1964).
Young, B. J., Mallya, R. K., Leslie, R. D., Clark, C. J. & Hamblin, T. J. Anti-keratin antibodies in rheumatoid arthritis. Br. Med. J. 2, 97–99 (1979).
Schellekens, G. A., de Jong, B. A., van den Hoogen, F. H., van de Putte, L. B. & van Venrooij, W. J. Citrulline is an essential constituent of antigenic determinants recognized by rheumatoid arthritis-specific autoantibodies. J. Clin. Invest. 101, 273–281 (1998).
Girbal-Neuhauser, E. et al. The epitopes targeted by the rheumatoid arthritis-associated antifilaggrin autoantibodies are posttranslationally generated on various sites of (pro)filaggrin by deimination of arginine residues. J. Immunol. 162, 585–594 (1999).
van der Helm-van Mil, A. H., Verpoort, K. N., Breedveld, F. C., Toes, R. E. & Huizinga, T. W. Antibodies to citrullinated proteins and differences in clinical progression of rheumatoid arthritis. Arthritis Res. Ther. 7, R949–R958 (2005).
Visser, K. et al. A matrix risk model for the prediction of rapid radiographic progression in patients with rheumatoid arthritis receiving different dynamic treatment strategies: post hoc analyses from the BeSt study. Ann. Rheum. Dis. 69, 1333–1337 (2010).
Kroot, E. J. et al. The prognostic value of anti-cyclic citrullinated peptide antibody in patients with recent-onset rheumatoid arthritis. Arthritis Rheum. 43, 1831–1835 (2000).
Harre, U. et al. Induction of osteoclastogenesis and bone loss by human autoantibodies against citrullinated vimentin. J. Clin. Invest. 122, 1791–1802 (2012).
Khandpur, R. et al. NETs are a source of citrullinated autoantigens and stimulate inflammatory responses in rheumatoid arthritis. Sci. Transl Med. 5, 178ra140 (2013).
Trouw, L. A. et al. Anti-cyclic citrullinated peptide antibodies from rheumatoid arthritis patients activate complement via both the classical and alternative pathways. Arthritis Rheum. 60, 1923–1931 (2009).
Sokolove, J., Zhao, X., Chandra, P. E. & Robinson, W. H. Immune complexes containing citrullinated fibrinogen costimulate macrophages via Toll-like receptor 4 and Fcγ receptor. Arthritis Rheum. 63, 53–62 (2011).
Suurmond, J. et al. Toll-like receptor triggering augments activation of human mast cells by anti-citrullinated protein antibodies. Ann. Rheum. Dis. 74, 1915–1923 (2014).
Laurent, L. et al. Fcgamma receptor profile of monocytes and macrophages from rheumatoid arthritis patients and their response to immune complexes formed with autoantibodies to citrullinated proteins. Ann. Rheum. Dis. 70, 1052–1059 (2011).
Wigerblad, G. et al. Autoantibodies to citrullinated proteins induce joint pain independent of inflammation via a chemokine-dependent mechanism. Ann. Rheum. Dis. 75, 730–738 (2016).
Krishnamurthy, A. et al. Identification of a novel chemokine-dependent molecular mechanism underlying rheumatoid arthritis-associated autoantibody-mediated bone loss. Ann. Rheum. Dis. 75, 721–729 (2016).
Lee, Y. H. & Bae, S. C. Association between susceptibility to rheumatoid arthritis and PADI4 polymorphisms: a meta-analysis. Clin. Rheumatol. 35, 961–971 (2016).
Chang, X. et al. PADI2 is significantly associated with rheumatoid arthritis. PLOS ONE 8, e81259 (2013).
Lee, Y. H., Rho, Y. H., Choi, S. J., Ji, J. D. & Song, G. G. PADI4 polymorphisms and rheumatoid arthritis susceptibility: a meta-analysis. Rheumatol. Int. 27, 827–833 (2007).
Vossenaar, E. R. et al. The presence of citrullinated proteins is not specific for rheumatoid synovial tissue. Arthritis Rheum. 50, 3485–3494 (2004).
Masson-Bessiere, C. et al. The major synovial targets of the rheumatoid arthritis-specific antifilaggrin autoantibodies are deiminated forms of the α- and β-chains of fibrin. J. Immunol. 166, 4177–4184 (2001).
Romero, V. et al. Immune-mediated pore-forming pathways induce cellular hypercitrullination and generate citrullinated autoantigens in rheumatoid arthritis. Sci. Transl Med. 5, 209ra150 (2013).
Kerkman, P. F. et al. Synovial fluid mononuclear cells provide an environment for long-term survival of antibody-secreting cells and promote the spontaneous production of anti-citrullinated protein antibodies. Ann. Rheum. Dis. 75, 2201–2207 (2016).
Masson-Bessiere, C. et al. In the rheumatoid pannus, anti-filaggrin autoantibodies are produced by local plasma cells and constitute a higher proportion of IgG than in synovial fluid and serum. Clin. Exp. Immunol. 119, 544–552 (2000).
Rodriguez-Bayona, B., Perez-Venegas, J. J., Rodriguez, C. & Brieva, J. A. CD95-Mediated control of anti-citrullinated protein/peptides antibodies (ACPA)-producing plasma cells occurring in rheumatoid arthritis inflamed joints. Rheumatol. (Oxford) 46, 612–616 (2007).
Humby, F. et al. Ectopic lymphoid structures support ongoing production of class-switched autoantibodies in rheumatoid synovium. PLOS Med. 6, e1 (2009).
Willemze, A. et al. The concentration of anticitrullinated protein antibodies in serum and synovial fluid in relation to total immunoglobulin concentrations. Ann. Rheum. Dis. 72, 1059–1063 (2013).
Ioan-Facsinay, A. et al. Marked differences in fine specificity and isotype usage of the anti-citrullinated protein antibody in health and disease. Arthritis Rheum. 58, 3000–3008 (2008).
van der Woude, D. et al. Epitope spreading of the anti-citrullinated protein antibody response occurs before disease onset and is associated with the disease course of early arthritis. Ann. Rheum. Dis. 69, 1554–1561 (2010).
Verpoort, K. N. et al. Isotype distribution of anti-cyclic citrullinated peptide antibodies in undifferentiated arthritis and rheumatoid arthritis reflects an ongoing immune response. Arthritis Rheum. 54, 3799–3808 (2006).
Willemze, A. et al. The ACPA recognition profile and subgrouping of ACPA-positive RA patients. Ann. Rheum. Dis. 71, 268–274 (2012).
van de Stadt, L. A. et al. The extent of the anti-citrullinated protein antibody repertoire is associated with arthritis development in patients with seropositive arthralgia. Ann. Rheum. Dis. 70, 128–133 (2011).
van de Stadt, L. A. et al. Development of the anti-citrullinated protein antibody repertoire prior to the onset of rheumatoid arthritis. Arthritis Rheum. 63, 3226–3233 (2011).
Sokolove, J. et al. Autoantibody epitope spreading in the pre-clinical phase predicts progression to rheumatoid arthritis. PLOS ONE 7, e35296 (2012).
Suwannalai, P. et al. Anti-citrullinated protein antibodies have a low avidity compared with antibodies against recall antigens. Ann. Rheum. Dis. 70, 373–379 (2011).
Suwannalai, P. et al. Avidity maturation of anti-citrullinated protein antibodies in rheumatoid arthritis. Arthritis Rheum. 64, 1323–1328 (2012).
Li, Z., Woo, C. J., Iglesias-Ussel, M. D., Ronai, D. & Scharff, M. D. The generation of antibody diversity through somatic hypermutation and class switch recombination. Genes Dev. 18, 1–11 (2004).
Goodnow, C. C., Vinuesa, C. G., Randall, K. L., Mackay, F. & Brink, R. Control systems and decision making for antibody production. Nat. Immunol. 11, 681–688 (2010).
van de Stadt, L. A. et al. Monoclonal anti-citrullinated protein antibodies selected on citrullinated fibrinogen have distinct targets with different cross-reactivity patterns. Rheumatol. (Oxford) 52, 631–635 (2013).
Amara, K. et al. Monoclonal IgG antibodies generated from joint-derived B cells of RA patients have a strong bias toward citrullinated autoantigen recognition. J. Exp. Med. 210, 445–455 (2013).
Suwannalai, P. et al. The fine specificity of IgM anti-citrullinated protein antibodies (ACPA) is different from that of IgG ACPA. Arthritis Res. Ther. 13, R195 (2011).
Fan, L. Y. et al. Citrullinated vimentin stimulates proliferation, pro-inflammatory cytokine secretion, and PADI4 and RANKL expression of fibroblast-like synoviocytes in rheumatoid arthritis. Scand. J. Rheumatol. 41, 354–358 (2012).
Sun, B. et al. Citrullination of NF-κB p65 promotes its nuclear localization and TLR-induced expression of IL-1β and TNFα. Sci. Immunol. 2, eaal3062 (2017).
Huizinga, T. W. et al. Refining the complex rheumatoid arthritis phenotype based on specificity of the HLA-DRB1 shared epitope for antibodies to citrullinated proteins. Arthritis Rheum. 52, 3433–3438 (2005).
van Heemst, J. et al. Protective effect of HLA-DRB1*13 alleles during specific phases in the development of ACPA-positive RA. Ann. Rheum. Dis. 75, 1891–1898 (2016).
van der Woude, D. et al. Protection against anti-citrullinated protein antibody-positive rheumatoid arthritis is predominantly associated with HLA-DRB1*1301: a meta-analysis of HLA-DRB1 associations with anti-citrullinated protein antibody-positive and anti-citrullinated protein antibody-negative rheumatoid arthritis in four European populations. Arthritis Rheum. 62, 1236–1245 (2010).
van Wesemael, T. J. et al. Smoking is associated with the concurrent presence of multiple autoantibodies in rheumatoid arthritis rather than with anti-citrullinated protein antibodies per se: a multicenter cohort study. Arthritis Res. Ther. 18, 285 (2016).
Murphy, D., Mattey, D. & Hutchinson, D. Anti-citrullinated protein antibody positive rheumatoid arthritis is primarily determined by rheumatoid factor titre and the shared epitope rather than smoking per se. PLOS ONE 12, e0180655 (2017).
Dekkers, J. S. et al. Breach of autoreactive B cell tolerance by post-translationally modified proteins. Ann. Rheum. Dis. 76, 1449–1457 (2017).
van Heemst, J. et al. Crossreactivity to vinculin and microbes provides a molecular basis for HLA-based protection against rheumatoid arthritis. Nat. Commun. 6, 6681 (2015).
Hand, T. W. et al. Acute gastrointestinal infection induces long-lived microbiota-specific T cell responses. Science 337, 1553–1556 (2012).
Tsuda, R. et al. Monoclonal antibody against citrullinated peptides obtained from rheumatoid arthritis patients reacts with numerous citrullinated microbial and food proteins. Arthritis Rheumatol. 67, 2020–2031 (2015).
Pianta, A. et al. Two rheumatoid arthritis-specific autoantigens correlate microbial immunity with autoimmune responses in joints. J. Clin. Invest. 127, 2946–2956 (2017).
James, E. A. et al. Citrulline-specific Th1 cells are increased in rheumatoid arthritis and their frequency is influenced by disease duration and therapy. Arthritis Rheumatol. 66, 1712–1722 (2014).
Chemin, K. et al. A novel HLA-DRB1*10:01-restricted T cell epitope from citrullinated type II collagen relevant to rheumatoid arthritis. Arthritis Rheumatol. 68, 1124–1135 (2016).
Cornillet, M., Verrouil, E., Cantagrel, A., Serre, G. & Nogueira, L. In ACPA-positive RA patients, antibodies to EBNA35-58Cit, a citrullinated peptide from the Epstein–Barr nuclear antigen-1, strongly cross-react with the peptide β60-74Cit which bears the immunodominant epitope of citrullinated fibrin. Immunol. Res. 61, 117–125 (2015).
Lundberg, K. et al. Antibodies to citrullinated alpha-enolase peptide 1 are specific for rheumatoid arthritis and cross-react with bacterial enolase. Arthritis Rheum. 58, 3009–3019 (2008).
Li, S. et al. Autoantibodies from single circulating plasmablasts react with citrullinated antigens and Porphyromonas gingivalis in rheumatoid arthritis. Arthritis Rheumatol. 68, 614–626 (2016).
Trouw, L. A., Rispens, T. & Toes, R. E. M. Beyond citrullination: other post-translational protein modifications in rheumatoid arthritis. Nat. Rev. Rheum. 13, 331–339 (2017).
Scherer, H. U. et al. Immunoglobulin 1 (IgG1) Fc-glycosylation profiling of anti-citrullinated peptide antibodies from human serum. Proteomics Clin. Appl. 3, 106–115 (2009).
Parekh, R. B. et al. Association of rheumatoid arthritis and primary osteoarthritis with changes in the glycosylation pattern of total serum IgG. Nature 316, 452–457 (1985).
Arnold, J. N., Wormald, M. R., Sim, R. B., Rudd, P. M. & Dwek, R. A. The impact of glycosylation on the biological function and structure of human immunoglobulins. Annu. Rev. Immunol. 25, 21–50 (2007).
Rombouts, Y. et al. Extensive glycosylation of ACPA-IgG variable domains modulates binding to citrullinated antigens in rheumatoid arthritis. Ann. Rheum. Dis. 75, 578–585 (2016).
Hafkenscheid, L. et al. Structural analysis of variable domain glycosylation of anti-citrullinated protein antibodies in rheumatoid arthritis reveals the presence of highly sialylated glycans. Mol. Cell Proteomics 16, 278–287 (2016).
Shakin-Eshleman, S. H., Spitalnik, S. L. & Kasturi, L. The amino acid at the X position of an Asn-X-Ser sequon is an important determinant of N-linked core-glycosylation efficiency. J. Biol. Chem. 271, 6363–6366 (1996).
Vergroesen, R. D. et al. B-cell receptor sequencing of anti-citrullinated protein antibody (ACPA) IgG-expressing B cells indicates a selective advantage for the introduction of N-glycosylation sites during somatic hypermutation. Ann. Rheum. Dis. https://doi.org/10.1136/annrheumdis-2017-212052 (2017).
van de Bovenkamp, F. S., Hafkenscheid, L., Rispens, T. & Rombouts, Y. The emerging importance of IgG Fab glycosylation in immunity. J. Immunol. 196, 1435–1441 (2016).
Kempers, A. C. et al. The extensive glycosylation of the ACPA variable domain observed for ACPA-IgG is absent from ACPA-IgM. Ann. Rheum. Dis. https://doi.org/10.1136/annrheumdis-2017-211533 (2017).
Raju, T. S. Terminal sugars of Fc glycans influence antibody effector functions of IgGs. Curr. Opin. Immunol. 20, 471–478 (2008).
Mimura, Y. et al. Role of oligosaccharide residues of IgG1-Fc in FcγRIIb binding. J. Biol. Chem. 276, 45539–45547 (2001).
Sondermann, P., Pincetic, A., Maamary, J., Lammens, K. & Ravetch, J. V. General mechanism for modulating immunoglobulin effector function. Proc. Natl Acad. Sci. USA 110, 9868–9872 (2013).
Malhotra, R. et al. Glycosylation changes of IgG associated with rheumatoid arthritis can activate complement via the mannose-binding protein. Nat. Med. 1, 237–243 (1995).
Kaneko, Y., Nimmerjahn, F. & Ravetch, J. V. Anti-inflammatory activity of immunoglobulin G resulting from Fc sialylation. Science 313, 670–673 (2006).
Nimmerjahn, F., Anthony, R. M. & Ravetch, J. V. Agalactosylated IgG antibodies depend on cellular Fc receptors for in vivo activity. Proc. Natl Acad. Sci. USA 104, 8433–8437 (2007).
Rademacher, T. W., Williams, P. & Dwek, R. A. Agalactosyl glycoforms of IgG autoantibodies are pathogenic. Proc. Natl Acad. Sci. USA 91, 6123–6127 (1994).
Dekkers, G. et al. Decoding the human immunoglobulin G-glycan repertoire reveals a spectrum of Fc-receptor- and complement-mediated-effector activities. Front. Immunol. 8, 877 (2017).
Youinou, P. et al. Galactose terminating oligosaccharides of IgG in patients with primary Sjögren's syndrome. J. Autoimmun. 5, 393–400 (1992).
Tomana, M., Schrohenloher, R. E., Koopman, W. J., Alarcon, G. S. & Paul, W. A. Abnormal glycosylation of serum IgG from patients with chronic inflammatory diseases. Arthritis Rheum. 31, 333–338 (1988).
Scherer, H. U. et al. Glycan profiling of anti-citrullinated protein antibodies isolated from human serum and synovial fluid. Arthritis Rheum. 62, 1620–1629 (2010).
van Zeben, D. et al. Early agalactosylation of IgG is associated with a more progressive disease course in patients with rheumatoid arthritis: results of a follow-up study. Br. J. Rheumatol. 33, 36–43 (1994).
Croce, A. et al. Effect of infliximab on the glycosylation of IgG of patients with rheumatoid arthritis. J. Clin. Lab. Anal. 21, 303–314 (2007).
Ercan, A. et al. Hypogalactosylation of serum N-glycans fails to predict clinical response to methotrexate and TNF inhibition in rheumatoid arthritis. Arthritis Res. Ther. 14, R43 (2012).
Rook, G. A. et al. Changes in IgG glycoform levels are associated with remission of arthritis during pregnancy. J. Autoimmun. 4, 779–794 (1991).
Alavi, A., Arden, N., Spector, T. D. & Axford, J. S. Immunoglobulin G glycosylation and clinical outcome in rheumatoid arthritis during pregnancy. J. Rheumatol. 27, 1379–1385 (2000).
Bondt, A. et al. Association between galactosylation of immunoglobulin G and improvement of rheumatoid arthritis during pregnancy is independent of sialylation. J. Proteome Res. 12, 4522–4531 (2013).
Rombouts, Y. et al. Anti-citrullinated protein antibodies acquire a pro-inflammatory Fc glycosylation phenotype prior to the onset of rheumatoid arthritis. Ann. Rheum. Dis. 74, 234–241 (2015).
Ercan, A. et al. Aberrant IgG galactosylation precedes disease onset, correlates with disease activity, and is prevalent in autoantibodies in rheumatoid arthritis. Arthritis Rheum. 62, 2239–2248 (2010).
Pfeifle, R. et al. Regulation of autoantibody activity by the IL-23-TH17 axis determines the onset of auto-immune disease. Nat. Immunol. 18, 104–113 (2017).
Ohmi, Y. et al. Sialylation converts arthritogenic IgG into inhibitors of collagen-induced arthritis. Nat. Commun. 7, 11205 (2016).
van Hamburg, J. P. et al. TH17 cells, but not TH1 cells, from patients with early rheumatoid arthritis are potent inducers of matrix metalloproteinases and proinflammatory cytokines upon synovial fibroblast interaction, including autocrine interleukin-17A production. Arthritis Rheum. 63, 73–83 (2011).
Murphy, C. A. et al. Divergent pro- and antiinflammatory roles for IL-23 and IL-12 in joint autoimmune inflammation. J. Exp. Med. 198, 1951–1957 (2003).
Arbuckle, M. R. et al. Development of autoantibodies before the clinical onset of systemic lupus erythema-tosus. N. Engl. J. Med. 349, 1526–1533 (2003).
van Tuyl, L. H. et al. IgM-rheumatoid factor and anti-cyclic citrullinated peptide decrease by 50% during intensive treatment in early rheumatoid arthritis. Ann. Rheum. Dis. 68, 1652–1653 (2009).
ter Borg, E. J., Horst, G., Hummel, E. J., Limburg, P. C. & Kallenberg, C. G. Measurement of increases in anti-double-stranded DNA antibody levels as a predictor of disease exacerbation in systemic lupus erythematosus. A long-term, prospective study. Arthritis Rheum. 33, 634–643 (1990).
Naseer, S. Y., Seiffert-Sinha, K. & Sinha, A. A. Detailed profiling of anti-desmoglein autoantibodies identifies anti-Dsg1 reactivity as a key driver of disease activity and clinical expression in pemphigus vulgaris. Autoimmunity 48, 231–241 (2015).
Ursum, J., Bos, W. H., van Dillen, N., Dijkmans, B. A. & van Schaardenburg, D. Levels of anti-citrullinated protein antibodies and IgM rheumatoid factor are not associated with outcome in early arthritis patients: a cohort study. Arthritis Res. Ther. 12, R8 (2010).
De Rycke, L. et al. Rheumatoid factor, but not anti-cyclic citrullinated peptide antibodies, is modulated by infliximab treatment in rheumatoid arthritis. Ann. Rheum. Dis. 64, 299–302 (2005).
Shiozawa, K. et al. Anticitrullinated protein antibody, but not its titer, is a predictor of radiographic progression and disease activity in rheumatoid arthritis. J. Rheumatol. 39, 694–700 (2012).
Landmann, T., Kehl, G. & Bergner, R. The continuous measurement of anti-CCP-antibodies does not help to evaluate the disease activity in anti-CCP-antibody-positive patients with rheumatoid arthritis. Clin. Rheumatol. 29, 1449–1453 (2010).
Mikuls, T. R. et al. Association of rheumatoid arthritis treatment response and disease duration with declines in serum levels of IgM rheumatoid factor and anti-cyclic citrullinated peptide antibody. Arthritis Rheum. 50, 3776–3782 (2004).
Teng, Y. O. et al. Induction of long-term B-cell depletion in refractory rheumatoid arthritis patients preferentially affects autoreactive more than protective humoral immunity. Arthritis Res. Ther. 14, R57 (2012).
Cambridge, G. et al. Serologic changes following B lymphocyte depletion therapy for rheumatoid arthritis. Arthritis Rheum. 48, 2146–2154 (2003).
Wunderlich, C., Oliviera, I., Figueiredo, C. P., Rech, J. & Schett, G. Effects of DMARDs on citrullinated peptide autoantibody levels in RA patients—a longitudinal analysis. Semin. Arthritis Rheum. 46, 709–714 (2017).
Daridon, C., Burmester, G. R. & Dorner, T. Anticytokine therapy impacting on B cells in autoimmune diseases. Curr. Opin. Rheumatol. 21, 205–210 (2009).
van Vollenhoven, R. F., Wax, S., Li, Y. & Tak, P. P. Safety and efficacy of atacicept in combination with rituximab for reducing the signs and symptoms of rheumatoid arthritis: a phase II, randomized, double-blind, placebo-controlled pilot trial. Arthritis Rheumatol. 67, 2828–2836 (2015).
van Vollenhoven, R. F., Kinnman, N., Vincent, E., Wax, S. & Bathon, J. Atacicept in patients with rheumatoid arthritis and an inadequate response to methotrexate: results of a phase II, randomized, placebo-controlled trial. Arthritis Rheum. 63, 1782–1792 (2011).
Genovese, M. C., Kinnman, N., de La Bourdonnaye, G., Pena Rossi, C. & Tak, P. P. Atacicept in patients with rheumatoid arthritis and an inadequate response to tumor necrosis factor antagonist therapy: results of a phase II, randomized, placebo-controlled, dose-finding trial. Arthritis Rheum. 63, 1793–1803 (2011).
Kerkman, P. F. et al. Circulating plasmablasts/plasmacells as a source of anticitrullinated protein antibodies in patients with rheumatoid arthritis. Ann. Rheum. Dis. 72, 1259–1263 (2013).
Kerkman, P. F. et al. Identification and characterisation of citrullinated antigen-specific B cells in peripheral blood of patients with rheumatoid arthritis. Ann. Rheum. Dis. 75, 1170–1176 (2016).
Pelzek, A. J. et al. Disease associated anti-citrullinated protein memory B cells in rheumatoid arthritis persist in clinical remission. Arthritis Rheumatol. https://doi.org/10.1002/art.40053 (2017).
Dorner, T. Crossroads of B cell activation in autoimmunity: rationale of targeting B cells. J. Rheumatol. Suppl. 77, 3–11 (2006).
Frolich, D. et al. Secondary immunization generates clonally related antigen-specific plasma cells and memory B cells. J. Immunol. 185, 3103–3110 (2010).
Corsiero, E. et al. Single cell cloning and recombinant monoclonal antibodies generation from RA synovial B cells reveal frequent targeting of citrullinated histones of NETs. Ann. Rheum. Dis. 75, 1866–1875 (2016).
Melchers, F. Checkpoints that control B cell development. J. Clin. Invest. 125, 2203–2210 (2015).
Lang, J. et al. Receptor editing and genetic variability in human autoreactive B cells. J. Exp. Med. 213, 93–108 (2015).
Wardemann, H. et al. Predominant autoantibody production by early human B cell precursors. Science 301, 1374–1377 (2003).
Samuels, J., Ng, Y. S., Coupillaud, C., Paget, D. & Meffre, E. Impaired early B cell tolerance in patients with rheumatoid arthritis. J. Exp. Med. 201, 1659–1667 (2005).
Menard, L. et al. The PTPN22 allele encoding an R620W variant interferes with the removal of developing autoreactive B cells in humans. J. Clin. Invest. 121, 3635–3644 (2011).
Lakos, G. et al. Anti-cyclic citrullinated peptide antibody isotypes in rheumatoid arthritis: association with disease duration, rheumatoid factor production and the presence of shared epitope. Clin. Exp. Rheumatol. 26, 253–260 (2008).
Levy, J., Barnett, E. V., MacDonald, N. S. & Klinenberg, J. R. Altered immunoglobulin metabolism in systemic lupus erythematosus and rheumatoid arthritis. J. Clin. Invest. 49, 708–715 (1970).
Kasturi, L., Eshleman, J. R., Wunner, W. H. & Shakin-Eshleman, S. H. The hydroxy amino acid in an Asn-X-Ser/Thr sequon can influence N-linked core glycosylation efficiency and the level of expression of a cell surface glycoprotein. J. Biol. Chem. 270, 14756–14761 (1995).
McCann, K. J. et al. Remarkable selective glycosylation of the immunoglobulin variable region in follicular lymphoma. Mol. Immunol. 45, 1567–1572 (2008).
Radcliffe, C. M. et al. Human follicular lymphoma cells contain oligomannose glycans in the antigen-binding site of the B-cell receptor. J. Biol. Chem. 282, 7405–7415 (2007).
Sabouri, Z. et al. Redemption of autoantibodies on anergic B cells by variable-region glycosylation and mutation away from self-reactivity. Proc. Natl Acad. Sci. USA 111, E2567–E2575 (2014).
Sieger, N. et al. CD22 ligation inhibits downstream B cell receptor signaling and Ca2+ flux upon activation. Arthritis Rheum. 65, 770–779 (2013).
Crocker, P. R., Paulson, J. C. & Varki, A. Siglecs and their roles in the immune system. Nat. Rev. Immunol. 7, 255–266 (2007).
Petersen, J., Heilmann, C., Bjerrum, O. J., Ingemann-Hansen, T. & Halkjaer-Kristensen, J. IgG rheumatoid factor-secreting lymphocytes in rheumatoid arthritis: evaluation of a haemolytic plaque-forming cell technique. Scand. J. Immunol. 17, 471–478 (1983).
Rudolphi, U. et al. The B cell repertoire of patients with rheumatoid arthritis. II. Increased frequencies of IgG+ and IgA+ B cells specific for mycobacterial heat-shock protein 60 or human type II collagen in synovial fluid and tissue. Arthritis Rheum. 40, 1409–1419 (1997).
Reparon-Schuijt, C. C. et al. Functional analysis of rheumatoid factor-producing B cells from the synovial fluid of rheumatoid arthritis patients. Arthritis Rheum. 41, 2211–2220 (1998).
Reparon-Schuijt, C. C. et al. Secretion of anti–citrulline-containing peptide antibody by B lymphocytes in rheumatoid arthritis. Arthritis Rheum. 44, 7 (2001).
Bellatin, M. F. et al. Production of autoantibodies against citrullinated antigens/peptides by human B cells. J. Immunol. 188, 3542–3550 (2012).
Franz, B., May, K. F. Jr., Dranoff, G. & Wucherpfennig, K. Ex vivo characterization and isolation of rare memory B cells with antigen tetramers. Blood 118, 348–357 (2011).
Acknowledgements
The authors thank Lise Hafkenscheid for help with preparing the molecular model depicted in the figure in Box 2. H.U.S, R.E.M.T. and T.H.U. acknowledge support from the Dutch Arthritis Foundation, the Netherlands Organisation for Scientific Research (NWO), and European Union Innovative Medicines Initiative and 7th Framework Program (FP7) grants. G.S. acknowledges support from the German Research Council (DFG) through projects SPP1468, SPP1937 and CRC1181 as well as the Innovative Medicines Initiative-funded project BTCure.
Author information
Authors and Affiliations
Contributions
All authors wrote the article, provided substantial contributions to discussions of its content, and undertook review and/or editing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- Immunoglobulin isotypes
-
Different immunoglobulin variants, defined by the isotype of the immunoglobulin heavy chains (α, δ, γ, ε, μ).
- Avidity
-
In antibodies, describes the cumulative binding strength of more than one receptor–ligand interaction; the binding strength of multiple, polyclonal antibodies (each of which potentially exhibits a different affinity for a particular antigen) can be described based on their avidity.
- HLA shared epitope
-
(HLA-SE). A group of genes encoding HLA alleles that share a common amino acid sequence (the 'shared epitope') in the antigen-binding region of the HLA molecule; carriers of these alleles are at an increased risk of developing anti-citrullinated protein antibody (ACPA)-positive RA.
- N-Glycosylation
-
The post-translational, covalent linkage of glycan residues (carbohydrates) to an asparagine (N) residue in a given protein.
- Somatic hypermutation
-
A process that introduces point mutations in genes that encode variable regions of antibodies; part of affinity maturation that occurs primarily in germinal centres.
- Affinity maturation
-
The process that generates B cell receptors with increased affinity for their cognate antigen, in which B cells with the highest avidity are positively selected; this process requires somatic hypermutation in B cells and occurs mainly in germinal centres.
Rights and permissions
About this article
Cite this article
Scherer, H., Huizinga, T., Krönke, G. et al. The B cell response to citrullinated antigens in the development of rheumatoid arthritis. Nat Rev Rheumatol 14, 157–169 (2018). https://doi.org/10.1038/nrrheum.2018.10
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2018.10
This article is cited by
-
Pathogenesis of autoimmune disease
Nature Reviews Nephrology (2023)
-
Characterization of chronic relapsing antibody mediated arthritis in mice with a mutation in Ncf1 causing reduced oxidative burst
Molecular Biomedicine (2022)
-
Rheumatoid arthritis sera antibodies to citrullinated collagen type II bind to joint cartilage
Arthritis Research & Therapy (2022)
-
Multicellular immune dynamics implicate PIM1 as a potential therapeutic target for uveitis
Nature Communications (2022)
-
The current status of anti-citrullinated protein antibodies and citrullinated protein-reactive B cells in the pathogenesis of rheumatoid arthritis
Molecular Biology Reports (2022)