Key Points
-
More than 100 genetic loci have been associated with ankylosing spondylitis (AS), but together they explain less than 30% of AS heritability
-
Multiple genes involved in antigen processing and presentation are associated with AS
-
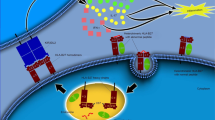
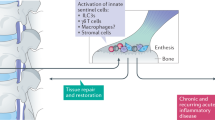
Investigation into the cellular sources and regulation of IL-17 production is crucial to understanding the pathogenesis of AS
-
Cells involved in type 3 immunity produce IL-17 and include innate lymphoid cells and γδ T cells
-
Given the efficacy of the anti-IL-17 monoclonal antibody secukinumab in the treatment of AS, other therapies targeting type 3 immunity could also be effective
Abstract
Over the past 5 years, advances in high-throughput techniques and studies involving large cohorts of patients have led to considerable advances in the identification of novel genetic associations and immune pathways involved in ankylosing spondylitis (AS). These discoveries include genes encoding cytokine receptors, transcription factors, signalling molecules and transport proteins. Although progress has been made in understanding the functions and potential pathogenic roles of some of these molecules, much work remains to be done to comprehend their complex interactions and therapeutic potential in AS. In this Review, we outline the current knowledge of AS pathogenesis, including genetic risk associations, HLA-B27-mediated pathology, perturbations in antigen-presentation pathways and the contribution of the type 3 immune response.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Van Praet, L., Jacques, P., Van Den Bosch, F., & Elewaut, D. The transition of acute to chronic bowel inflammation in spondyloarthritis. Nat. Rev. Rheumatol. 8, 288–295 (2012).
Marroquin Belaunzaran, O. et al. HLA-B27-homodimer-specific antibody modulates the expansion of pro-inflammatory T-cells in HLA-B27 transgenic rats. PLoS ONE 10, e0130811 (2015).
Baeten, D. et al. Anti-interleukin-17A monoclonal antibody secukinumab in treatment of ankylosing spondylitis: a randomised, double-blind, placebo-controlled trial. Lancet 382, 1705–1713 (2013).
Brewerton, D. A. et al. Ankylosing spondylitis and HL-A 27. Lancet 1, 904–907 (1973).
The Australo-Anglo-American Spondyloarthritis Consortium et al. Interaction between ERAP1 and HLA-B27 in ankylosing spondylitis implicates peptide handling in the mechanism for HLA-B27 in disease susceptibility. Nat. Genet. 43, 761–767 (2011).
Wellcome Trust Case Control Consortium et al. Association scan of 14,500 nonsynonymous SNPs in four diseases identifies autoimmunity variants. Nat. Genet. 39, 1329–1337 (2007).
International Genetics of Ankylosing Spondylitis Consortium et al. Identification of multiple risk variants for ankylosing spondylitis through high-density genotyping of immune-related loci. Nat. Genet. 45, 730–738 (2013).
Australo-Anglo-American Spondyloarthritis Consortium et al. Genome-wide association study of ankylosing spondylitis identifies non-MHC susceptibility loci. Nat. Genet. 42, 123–127 (2010).
Ellinghaus, D. et al. Analysis of five chronic inflammatory diseases identifies 27 new associations and highlights disease-specific patterns at shared loci. Nat. Genet. 48, 510–518 (2016).
Brown, M. A., Kenna, T. & Wordsworth, B. P. Genetics of ankylosing spondylitis — insights into pathogenesis. Nat. Rev. Rheumatol. 12, 81–91 (2016).
Cortes, A. et al. Major histocompatibility complex associations of ankylosing spondylitis are complex and involve further epistasis with ERAP1. Nat. Commun. 6, 7146 (2015).
Robinson, W. P. et al. HLA-Bw60 increases susceptibility to ankylosing spondylitis in HLA-B27+ patients. Arthritis Rheum. 32, 1135–1141 (1989).
Brown, M. A. et al. HLA class I associations of ankylosing spondylitis in the white population in the United Kingdom. Ann. Rheum. Dis. 55, 268–270 (1996).
Kim, K. et al. An HLA-C amino-acid variant in addition to HLA-B*27 confers risk for ankylosing spondylitis in the Korean population. Arthritis Res. Ther. 17, 342 (2015).
Genetic Analysis of Psoriasis Consortium & the Wellcome Trust Case Control Consortium 2 et al. A genome-wide association study identifies new psoriasis susceptibility loci and an interaction between HLA-C and ERAP1. Nat. Genet. 42, 985–990 (2010).
Kirino, Y. et al. Genome-wide association analysis identifies new susceptibility loci for Behcet's disease and epistasis between HLA-B*51 and ERAP1. Nat. Genet. 45, 202–207 (2013).
Kuiper, J. J. et al. A genome-wide association study identifies a functional ERAP2 haplotype associated with birdshot chorioretinopathy. Hum. Mol. Genet. 23, 6081–6087 (2014).
Alvarez-Navarro, C., Martin-Esteban, A., Barnea, E., Admon, A. & Lopez de Castro, J. A. Endoplasmic reticulum aminopeptidase 1 (ERAP1) polymorphism relevant to inflammatory disease shapes the peptidome of the birdshot chorioretinopathy-associated HLA-A*29:02 antigen. Mol. Cell. Proteomics 14, 1770–1780 (2015).
Rausch, P. et al. Colonic mucosa-associated microbiota is influenced by an interaction of crohn disease and FUT2 (Secretor) genotype. Proc. Natl Acad. Sci. USA 108, 19030–19035 (2011).
Costello, M. E. et al. Intestinal dysbiosis in ankylosing spondylitis. Arthritis Rheumatol. 67, 686–691 (2014).
O'Rielly, D. D. et al. Private rare deletions in SEC16A and MAMDC4 may represent novel pathogenic variants in familial axial spondyloarthritis. Ann. Rheum. Dis. 75, 772–779 (2015).
Bowness, P. Hla-B27. Annu. Rev. Immunol. 33, 29–48 (2015).
Khan, M. A. Polymorphism of HLA-B27: 105 subtypes currently known. Curr. Rheumatol. Rep. 15, 362 (2013).
Garcia-Medel, N. et al. Peptide handling by HLA-B27 subtypes influences their biological behavior, association with ankylosing spondylitis and susceptibility to endoplasmic reticulum aminopeptidase 1 (ERAP1). Mol. Cell. Proteomics 13, 3367–3380 (2014).
Schittenhelm, R. B., Sian, T. C., Wilmann, P. G., Dudek, N. L. & Purcell, A. W. Revisiting the arthritogenic peptide theory: quantitative not qualitative changes in the peptide repertoire of HLA-B27 allotypes. Arthritis Rheumatol. 67, 702–713 (2015).
Schittenhelm, R. B., Sivaneswaran, S., Lim Kam Sian, T. C., Croft, N. P. & Purcell, A. W. Human leukocyte antigen (HLA) B27 allotype-specific binding and candidate arthritogenic peptides revealed through heuristic clustering of data-independent acquisition mass spectrometry (DIA-MS) data. Mol. Cell. Proteomics 15, 1867–1876 (2016).
Hulsmeyer, M. et al. Dual, HLA-B27 subtype-dependent conformation of a self-peptide. J. Exp. Med. 199, 271–281 (2004).
Rysnik, O. et al. Non-conventional forms of HLA-B27 are expressed in spondyloarthritis joints and gut tissue. J. Autoimmun. 70, 12–21 (2016).
Kenna, T. J., Robinson, P. C. & Haroon, N. Endoplasmic reticulum aminopeptidases in the pathogenesis of ankylosing spondylitis. Rheumatology (Oxford) 54, 1549–1556 (2015).
Allen, R. L., O'Callaghan, C. A., McMichael, A. J. & Bowness, P. Cutting edge: HLA-B27 can form a novel β2-microglobulin-free heavy chain homodimer structure. J. Immunol. 162, 5045–5048 (1999).
Colbert, R. A., DeLay, M. L., Klenk, E. I. & Layh-Schmitt, G. From HLA-B27 to spondyloarthritis: a journey through the ER. Immunol. Rev. 233, 181–202 (2010).
Ciccia, F. et al. Evidence that autophagy, but not the unfolded protein response, regulates the expression of IL-23 in the gut of patients with ankylosing spondylitis and subclinical gut inflammation. Ann. Rheum. Dis. 73, 1566–1574 (2013).
Neerinckx, B., Carter, S. & Lories, R. J. No evidence for a critical role of the unfolded protein response in synovium and blood of patients with ankylosing spondylitis. Ann. Rheum. Dis. 73, 629–630 (2014).
Ciccia, F. & Haroon, N. Autophagy in the pathogenesis of ankylosing spondylitis. Clin. Rheumatol. 35, 1433–1436 (2016).
Neerinckx, B., Carter, S. & Lories, R. IL-23 expression and activation of autophagy in synovium and PBMCs of HLA-B27 positive patients with ankylosing spondylitis. Response to: 'Evidence that autophagy, but not the unfolded protein response, regulates the expression of IL-23 in the gut of patients with ankylosing spondylitis and subclinical gut inflammation' by Ciccia et al. Ann. Rheum. Dis. 73, e68 (2014).
Guiliano, D. B. et al. Endoplasmic reticulum degradation-enhancing α-mannosidase-like protein 1 targets misfolded HLA-B27 dimers for endoplasmic reticulum-associated degradation. Arthritis Rheumatol. 66, 2976–2988 (2014).
Bowness, P. et al. Th17 cells expressing KIR3DL2+ and responsive to HLA-B27 homodimers are increased in ankylosing spondylitis. J. Immunol. 186, 2672–2680 (2011).
Abdullah, H., Zhang, Z., Yee, K. & Haroon, N. KIR3DL1 interaction with HLA-B27 is altered by ankylosing spondylitis associated ERAP1 and enhanced by MHC class I cross-linking. Discov. Med. 20, 79–89 (2015).
Ridley, A. et al. Activation-induced killer cell immunoglobulin-like receptor 3DL2 binding to HLA-B27 licenses pathogenic T cell differentiation in spondyloarthritis. Arthritis Rheumatol. 68, 901–914 (2016).
Saveanu, L. et al. Concerted peptide trimming by human ERAP1 and ERAP2 aminopeptidase complexes in the endoplasmic reticulum. Nat. Immunol. 6, 689–697 (2005).
Chang, S. C., Momburg, F., Bhutani, N. & Goldberg, A. L. The ER aminopeptidase, ERAP1, trims precursors to lengths of MHC class I peptides by a “molecular ruler” mechanism. Proc. Natl Acad. Sci. USA 102, 17107–17112 (2005).
Saric, T. et al. An IFN-γ-induced aminopeptidase in the ER, ERAP1, trims precursors to MHC class I-presented peptides. Nat. Immunol. 3, 1169–1176 (2002).
Kochan, G. et al. Crystal structures of the endoplasmic reticulum aminopeptidase-1 (ERAP1) reveal the molecular basis for N-terminal peptide trimming. Proc. Natl Acad. Sci. USA 108, 7745–7750 (2011).
Chen, L. et al. Silencing or inhibition of endoplasmic reticulum aminopeptidase 1 (ERAP1) suppresses free heavy chain expression and Th17 responses in ankylosing spondylitis. Ann. Rheum. Dis. 75, 916–923 (2016).
Evnouchidou, I. et al. Coding single nucleotide polymorphisms of endoplasmic reticulum aminopeptidase 1 can affect antigenic peptide generation in vitro by influencing basic enzymatic properties of the enzyme. J. Immunol. 186, 1909–1913 (2011).
Reeves, E., Colebatch-Bourn, A., Elliott, T., Edwards, C. J. & James, E. Functionally distinct ERAP1 allotype combinations distinguish individuals with ankylosing spondylitis. Proc. Natl Acad. Sci. USA 111, 17594–17599 (2014).
Haroon, N., Tsui, F. W., Uchanska-Ziegler, B., Ziegler, A. & Inman, R. D. Endoplasmic reticulum aminopeptidase 1 (ERAP1) exhibits functionally significant interaction with HLA-B27 and relates to subtype specificity in ankylosing spondylitis. Ann. Rheum. Dis. 71, 589–595 (2012).
Garcia-Medel, N. et al. Functional interaction of the ankylosing spondylitis-associated endoplasmic reticulum aminopeptidase 1 polymorphism and HLA-B27 in vivo. Mol. Cell. Proteomics 11, 1416–1429 (2012).
Akram, A., Lin, A., Gracey, E., Streutker, C. J. & Inman, R. D. HLA-B27, but not HLA-B7, immunodominance to influenza is ERAP dependent. J. Immunol. 192, 5520–5528 (2014).
Tran, T. M., Hong, S., Edwan, J. H. & Colbert, R. A. ERAP1 reduces accumulation of aberrant and disulfide-linked forms of HLA-B27 on the cell surface. Mol. Immunol. 74, 10–17 (2016).
Kenna, T. J. et al. Disease-associated polymorphisms in ERAP1 do not alter endoplasmic reticulum stress in patients with ankylosing spondylitis. Genes Immun. 16, 35–42 (2014).
Tsui, F. W. et al. Association of an ERAP1 ERAP2 haplotype with familial ankylosing spondylitis. Ann. Rheum. Dis. 69, 733–736 (2010).
Evnouchidou, I., Weimershaus, M., Saveanu, L. & van Endert, P. ERAP1–ERAP2 dimerization increases peptide-trimming efficiency. J. Immunol. 193, 901–908 (2014).
Robinson, P. C. et al. ERAP2 functional knockout in humans does not alter surface heavy chains or HLA-B27, inflammatory cytokines or endoplasmic reticulum stress markers. Ann. Rheum. Dis. 74, 2092–2095 (2015).
Martin-Esteban, A., Guasp, P., Barnea, E., Admon, A. & Lopez de Castro, J. A. Functional interaction of the ankylosing spondylitis associated endoplasmic reticulum aminopeptidase 2 with the HLA-B*27 peptidome in human cells. Arthritis Rheumatol. 68, 2466–2475 (2016).
Wendling, D., Cedoz, J. P., Racadot, E. & Dumoulin, G. Serum IL-17, BMP-7, and bone turnover markers in patients with ankylosing spondylitis. Joint Bone Spine 74, 304–305 (2007).
Mei, Y. et al. Increased serum IL-17 and IL-23 in the patient with ankylosing spondylitis. Clin. Rheumatol. 30, 269–273 (2011).
Shen, H., Goodall, J. C. & Hill Gaston, J. S. Frequency and phenotype of peripheral blood Th17 cells in ankylosing spondylitis and rheumatoid arthritis. Arthritis Rheum. 60, 1647–1656 (2009).
Glatigny, S. et al. Proinflammatory Th17 cells are expanded and induced by dendritic cells in spondylarthritis-prone HLA-B27-transgenic rats. Arthritis Rheum. 64, 110–120 (2012).
Benham, H. et al. Interleukin-23 mediates the intestinal response to microbial β-1,3-glucan and the development of spondyloarthritis pathology in SKG mice. Arthritis Rheumatol. 66, 1755–1767 (2014).
Gracey, E. et al. Sexual dimorphism in the Th17 signature of ankylosing spondylitis. Arthritis Rheumatol. 68, 679–689 (2016).
Smith, J. A. & Colbert, R. A. Review: the interleukin-23/interleukin-17 axis in spondyloarthritis pathogenesis: Th17 and beyond. Arthritis Rheumatol. 66, 231–241 (2014).
Lau, M. C. et al. Genetic association of ankylosing spondylitis with TBX21 influences T-bet and pro-inflammatory cytokine expression in humans and SKG mice as a model of spondyloarthritis. Ann. Rheum. Dis. 76, 261–269 (2016).
Krausgruber, T. et al. T-bet is a key modulator of IL-23-driven pathogenic CD4+ T cell responses in the intestine. Nat. Commun. 7, 11627 (2016).
Sujino, T. et al. Tissue adaptation of regulatory and intraepithelial CD4+ T cells controls gut inflammation. Science 352, 1581–1586 (2016).
Gracey, E. et al. IL-7 primes IL-17 in mucosal-associated invariant T (MAIT) cells, which contribute to the Th17-axis in ankylosing spondylitis. Ann. Rheum. Dis. 75, 2124–2132 (2016).
Hayashi, E. et al. Involvement of mucosal-associated invariant T cells in ankylosing spondylitis. J. Rheumatol. 43, 1695–1703 (2016).
Ciccia, F. et al. Type 3 innate lymphoid cells producing IL-17 and IL-22 are expanded in the gut, in the peripheral blood, synovial fluid and bone marrow of patients with ankylosing spondylitis. Ann. Rheum. Dis. 74, 1739–1747 (2015).
Rihl, M. et al. Identification of interleukin-7 as a candidate disease mediator in spondylarthritis. Arthritis Rheum. 58, 3430–3435 (2008).
Kenna, T. J. et al. Enrichment of circulating interleukin-17-secreting interleukin-23 receptor-positive γ/δ T cells in patients with active ankylosing spondylitis. Arthritis Rheum. 64, 1420–1429 (2012).
Sherlock, J. P. et al. IL-23 induces spondyloarthropathy by acting on ROR-γt+ CD3+CD4−CD8− entheseal resident T cells. Nat. Med. 18, 1069–1076 (2012).
Reinhardt, A. et al. IL-23-dependent γ/δ T cells produce IL-17 and accumulate in enthesis, aortic valve, and ciliary body. Arthritis Rheumatol. 68, 2476–2486 (2016).
Ono, T. et al. IL-17-producing γδ T cells enhance bone regeneration. Nat. Commun. 7, 10928 (2016).
Benjamin, M. & McGonagle, D. The enthesis organ concept and its relevance to the spondyloarthropathies. Adv. Exp. Med. Biol. 649, 57–70 (2009).
McGonagle, D., Lories, R. J., Tan, A. L. & Benjamin, M. The concept of a “synovio–entheseal complex” and its implications for understanding joint inflammation and damage in psoriatic arthritis and beyond. Arthritis Rheum. 56, 2482–2491 (2007).
Jacques, P. et al. Proof of concept: enthesitis and new bone formation in spondyloarthritis are driven by mechanical strain and stromal cells. Ann. Rheum. Dis. 73, 437–445 (2014).
Ciccia, F. et al. Interleukin-22 and interleukin-22-producing NKp44+ natural killer cells in subclinical gut inflammation in ankylosing spondylitis. Arthritis Rheum. 64, 1869–1878 (2012).
Hepworth, M. R. et al. Immune tolerance. Group 3 innate lymphoid cells mediate intestinal selection of commensal bacteria-specific CD4+ T cells. Science 348, 1031–1035 (2015).
Lee, J. S. et al. Interleukin-23-independent IL-17 production regulates intestinal epithelial permeability. Immunity 43, 727–738 (2015).
Lee, Y. et al. Induction and molecular signature of pathogenic TH17 cells. Nat. Immunol. 13, 991–999 (2012).
Zuniga, L. A., Jain, R., Haines, C. & Cua, D. J. Th17 cell development: from the cradle to the grave. Immunol. Rev. 252, 78–88 (2013).
Bettelli, E. et al. Reciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory T cells. Nature 441, 235–238 (2006).
Mangan, P. R. et al. Transforming growth factor-β induces development of the TH17 lineage. Nature 441, 231–234 (2006).
Ghoreschi, K. et al. Generation of pathogenic TH17 cells in the absence of TGF-β signalling. Nature 467, 967–971 (2010).
Gagliani, N. et al. TH17 cells transdifferentiate into regulatory T cells during resolution of inflammation. Nature 523, 221–225 (2015).
Sefik, E. et al. Mucosal immunology. Individual intestinal symbionts induce a distinct population of RORγ+ regulatory T cells. Science 349, 993–997 (2015).
Ohnmacht, C. et al. Mucosal immunology. The microbiota regulates type 2 immunity through RORγ+ T cells. Science 349, 989–993 (2015).
Maxwell, J. R. et al. Differential roles for interleukin-23 and interleukin-17 in intestinal immunoregulation. Immunity 43, 739–750 (2015).
Jain, R. et al. Interleukin-23-induced transcription factor Blimp-1 promotes pathogenicity of T helper 17 cells. Immunity 44, 131–142 (2016).
Ellinghaus, D. et al. Association between variants of PRDM1 and NDP52 and Crohn's disease, based on exome sequencing and functional studies. Gastroenterology 145, 339–347 (2013).
Baeten, D. et al. Immunomodulatory effects of anti-tumor necrosis factor alpha therapy on synovium in spondylarthropathy: histologic findings in eight patients from an open-label pilot study. Arthritis Rheum. 44, 186–195 (2001).
Haroon, N. et al. From gene expression to serum proteins: biomarker discovery in ankylosing spondylitis. Ann. Rheum. Dis. 69, 297–300 (2008).
Milanez, F. M. et al. IL-23/Th17 axis is not influenced by TNF-blocking agents in ankylosing spondylitis patients. Arthritis Res. Ther. 18, 52 (2016).
Evans, H. G. et al. TNF-alpha blockade induces IL-10 expression in human CD4+ T cells. Nat. Commun. 5, 3199 (2014).
Sieper, J., Porter-Brown, B., Thompson, L., Harari, O. & Dougados, M. Assessment of short-term symptomatic efficacy of tocilizumab in ankylosing spondylitis: results of randomised, placebo-controlled trials. Ann. Rheum. Dis. 73, 95–100 (2014).
Sieper, J. et al. Sarilumab for the treatment of ankylosing spondylitis: results of a phase II, randomized, double-blind, placebo-controlled, international study (ALIGN). Ann. Rheum. Dis. 74, 1051–1057 (2015).
Wilson, N. J. et al. Development, cytokine profile and function of human interleukin 17-producing helper T cells. Nat. Immunol. 8, 950–957 (2007).
Poddubnyy, D., Hermann, K. G., Callhoff, J., Listing, J. & Sieper, J. Ustekinumab for the treatment of patients with active ankylosing spondylitis: results of a 28-week, prospective, open-label, proof-of-concept study (TOPAS). Ann. Rheum. Dis. 73, 817–823 (2014).
Yao, C. et al. Prostaglandin E2–EP4 signaling promotes immune inflammation through TH1 cell differentiation and TH17 cell expansion. Nat. Med. 15, 633–640 (2009).
Withers, D. R. et al. Transient inhibition of ROR-γt therapeutically limits intestinal inflammation by reducing TH17 cells and preserving group 3 innate lymphoid cells. Nat. Med. 22, 319–323 (2016).
de Wit, J. et al. RORγt inhibitors suppress TH17 responses in inflammatory arthritis and inflammatory bowel disease. J. Allergy Clin. Immunol. 137, 960–963 (2016).
Hueber, W. et al. Secukinumab, a human anti-IL-17A monoclonal antibody, for moderate to severe Crohn's disease: unexpected results of a randomised, double-blind placebo-controlled trial. Gut 61, 1693–1700 (2012).
Schreiber, S. et al. OP0113 No increased incidence of inflammatory bowel disease among secukinumab-treated patients with moderate to severe psoriasis, psoriatic arthritis, or ankylosing spondylitis: data from 14 phase 2 and phase 3 clinical studies [abstract]. Ann. Rheum. Dis. 75 (Suppl.), 97–98 (2016).
Asquith, M. & Rosenbaum, J. T. The interaction between host genetics and the microbiome in the pathogenesis of spondyloarthropathies. Curr. Opin. Rheumatol. 28, 405–412 (2016).
Van de Wiele, T., Van Praet, J. T., Marzorati, M., Drennan, M. B. & Elewaut, D. How the microbiota shapes rheumatic diseases. Nat. Rev. Rheumatol. 12, 398–411 (2016).
Tito, R. Y. et al. Dialister as microbial marker of disease activity in spondyloarthritis. Arthritis Rheumatol. 69, 114–121 (2016).
Lin, P. et al. HLA-B27 and human β2-microglobulin affect the gut microbiota of transgenic rats. PLoS ONE 9, e105684 (2014).
Rehaume, L. M. et al. ZAP-70 genotype disrupts the relationship between microbiota and host, leading to spondyloarthritis and ileitis in SKG mice. Arthritis Rheumatol. 66, 2780–2792 (2014).
Taurog, J. D. et al. The germfree state prevents development of gut and joint inflammatory disease in HLA-B27 transgenic rats. J. Exp. Med. 180, 2359–2364 (1994).
Baillet, A. C. et al. High chlamydia burden promotes tumor necrosis factor-dependent reactive arthritis in SKG mice. Arthritis Rheumatol. 67, 1535–1547 (2015).
Ruutu, M. et al. β-glucan triggers spondylarthritis and Crohn's disease-like ileitis in SKG mice. Arthritis Rheum. 64, 2211–2222 (2012).
Author information
Authors and Affiliations
Contributions
All the authors researched the data for the article, made substantial contributions to discussion of content, wrote the article, and edited/reviewed the manuscript before submission. V.R. and E.G. contributed equally to this work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- Genome-wide significance
-
In most genome-wide association studies, the threshold that an association must reach to be considered statistically significant is a high P value (≥5 × 10−8) owing to the multiple tests conducted in such studies.
- Seronegative diseases
-
Diseases not associated with serum autoantibodies.
- M1 family of zinc metallopeptidases
-
Aminopeptidases that cleave polypeptides from the N-terminus and are dependent on a single zinc ion for activity.
- Immunochip platform
-
A microarray chip containing probes that recognize approximately195,000 single nucleotide polymorphisms and 700 small insertion and/or deletions; the main aim of this platform is to fine-map |genetic associations identified in 11 autoimmune and inflammatory diseases.
- Data-independent acquisition mass spectrometry
-
Mass spectrometry technique in which all ions generated are fragmented and analysed without pre-selection.
- Autophagy
-
A process that involves the orderly degradation of dysfunctional intracellular components through their delivery to lysosomes in structures called autophagosomes.
- Endoplasmic reticulum-associated degradation
-
(ERAD). A process that facilitates the degradation of misfolded proteins in the endoplasmic reticulum by transporting them to the cytoplasm, where ubiquitylation followed by proteasome-mediated degradation occurs.
- Coat protein complex II
-
Vesicle coat protein that aids anterograde transport of proteins from the endoplasmic reticulum to the Golgi apparatus.
- SKG mice
-
Mice carrying a hypomorphic single nucleotide polymorphism in ZAP70 (a T cell receptor signalling molecule), which predisposes T cells to a T helper 17 cell phenotype. Under specific pathogen-free conditions, SKG mice are disease-free; however, a single dose of curdlan, an IL-23-inducing molecule, induces progressive spondyloathropathy characterized by axial and peripheral arthritis, dermatitis and colitis.
- Innate-like lymphocytes
-
Cells of the lymphocyte lineage that express T cell receptors of limited diversity and are restricted by non-classical MHC molecules such as CD1 or MR1. These cells typically recognize non-peptide antigens and are activated faster than regular peptide-restricted adaptive immune cells.
- Mucosal-associated invariant T (MAIT) cells
-
A population of innate-like lymphocytes that recognize bacterially derived vitamin B metabolites presented on the non-classical MHC molecule MR1.
- Synovio-enthesal complex
-
Anatomical unit comprising the fibrous insertion of tendon or ligament enthesis and the adjacent synovial membrane of the bursa.
- Fate-mapping studies
-
Studies that investigate the origin of cell populations through labelling and tracking of cells of interest. Immunological studies typically use membrane-incorporated dyes, or genetic switches that result in constitutive fluorochrome expression if a cell marker is expressed.
Rights and permissions
About this article
Cite this article
Ranganathan, V., Gracey, E., Brown, M. et al. Pathogenesis of ankylosing spondylitis — recent advances and future directions. Nat Rev Rheumatol 13, 359–367 (2017). https://doi.org/10.1038/nrrheum.2017.56
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2017.56
This article is cited by
-
Efficacy and safety of gut microbiota-based therapies in autoimmune and rheumatic diseases: a systematic review and meta-analysis of 80 randomized controlled trials
BMC Medicine (2024)
-
Programmed cell death 10 can be used as a potential biomarker for ankylosing spondylitis diagnosis and treatment
Spinal Cord (2024)
-
The complement factor H-related protein-5 (CFHR5) exacerbates pathological bone formation in ankylosing spondylitis
Journal of Molecular Medicine (2024)
-
Zur Rolle von HLA-B27 in der Pathogenese und Diagnostik der axialen Spondyloarthritis
Zeitschrift für Rheumatologie (2024)
-
“Long-term MRI findings in Ankylosing spondylitis patients treated with TNF inhibitors for a decade”
Rheumatology International (2024)