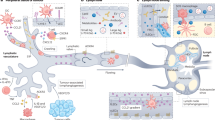
Key Points
-
Dramatic changes in lymphatic vessel contraction and in the lymph nodes that drain inflamed joints are associated with disease progression and response to therapy in murine models of rheumatoid arthritis (RA)
-
During mild to moderate experimental arthritis, lymphatic vessels and nodes that drain the joint undergo an initial 'expansion' phase that facilitates efficient lymphatic clearance and lymphatic vessel contractions
-
In preclinical models, the expansion phase is followed by a 'collapsed' phase, in which B cells in the draining lymph node translocate from the follicles to lymphatic sinuses and the lymph node collapses
-
The collapsed phase is characterized by lymphatic vessel structural damage, loss of contraction, and reduction in lymphatic clearance
-
Pilot clinical studies indicate that alterations in lymph node volume and/or lymphatic flow could serve as biomarkers of treatment response with the potential to predict RA flare
-
Several lymphatic system-modulating therapies show promise in preclinical models of inflammatory arthritis and RA
Abstract
Although clinical outcomes for patients with rheumatoid arthritis (RA) have greatly improved with the use of biologic and conventional DMARDs, approximately 40% of patients do not achieve primary clinical outcomes in randomized trials, and only a small proportion achieve lasting remission. Over the past decade, studies in murine models point to the critical role of the lymphatic system in the pathogenesis and therapy of inflammatory-erosive arthritis, presumably by the removal of catabolic factors, cytokines and inflammatory cells from the inflamed synovium. Murine studies demonstrate that lymphatic drainage increases at the onset of inflammatory-erosive arthritis but, as inflammation progresses to a more chronic phase, lymphatic clearance declines and both structural and cellular changes are observed in the draining lymph node. Specifically, chronic damage to the lymphatic vessel from persistent inflammation results in loss of lymphatic vessel contraction followed by lymph node collapse, reduced lymphatic drainage, and ultimately severe synovitis and joint erosion. Notably, clinical pilot studies in patients with RA report lymph node changes following treatment, and thus draining lymphatic vessels and nodes could represent a potential biomarker of arthritis activity and response to therapy. Most importantly, targeting lymphatics represents an innovative strategy for therapeutic intervention for RA.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Firestein, G. S. Evolving concepts of rheumatoid arthritis. Nature 423, 356–361 (2003).
Rudan, I. et al. Prevalence of rheumatoid arthritis in low- and middle-income countries: a systematic review and analysis. J. Glob. Health 5, 010409 (2015).
Weinblatt, M. E. et al. Adalimumab, a fully human anti-tumor necrosis factor alpha monoclonal antibody, for the treatment of rheumatoid arthritis in patients taking concomitant methotrexate: the ARMADA trial. Arthritis Rheum. 48, 35–45 (2003).
Weinblatt, M. E. et al. A trial of etanercept, a recombinant tumor necrosis factor receptor:Fc fusion protein, in patients with rheumatoid arthritis receiving methotrexate. N. Engl. J. Med. 340, 253–259 (1999).
Firestein, G. S. The disease formerly known as rheumatoid arthritis. Arthritis Res. Ther. 16, 114 (2014).
Pratt, A. G. & Isaacs, J. D. Seronegative rheumatoid arthritis: pathogenetic and therapeutic aspects. Best Pract. Res. Clin. Rheumatol 28, 651–659 (2014).
Zawieja, D. Lymphatic biology and the microcirculation: past, present and future. Microcirculation 12, 141–150 (2005).
Skandalakis, J. E., Skandalakis, L. J. & Skandalakis, P. N. Anatomy of the lymphatics. Surg. Oncol. Clin. N. Am. 16, 1–16 (2007).
Aspelund, A., Robciuc, M. R., Karaman, S., Makinen, T. & Alitalo, K. Lymphatic system in cardiovascular medicine. Circ. Res. 118, 515–530 (2016).
Chakraborty, S., Davis, M. J. & Muthuchamy, M. Emerging trends in the pathophysiology of lymphatic contractile function. Semin. Cell Dev. Biol. 38, 55–66 (2015).
Yao, L. C., Baluk, P., Srinivasan, R. S., Oliver, G. & McDonald, D. M. Plasticity of button-like junctions in the endothelium of airway lymphatics in development and inflammation. Am. J. Pathol. 180, 2561–2575 (2012).
Trzewik, J., Mallipattu, S. K., Artmann, G. M., Delano, F. A. & Schmid-Schonbein, G. W. Evidence for a second valve system in lymphatics: endothelial microvalves. FASEB J. 15, 1711–1717 (2001).
Collin, H. B. The ultrastructure of conjunctival lymphatic anchoring filaments. Exp. Eye Res. 8, 102–105 (1969).
Baluk, P. et al. Functionally specialized junctions between endothelial cells of lymphatic vessels. J. Exp. Med. 204, 2349–2362 (2007).
Breslin, J. W. Mechanical forces and lymphatic transport. Microvasc. Res. 96, 46–54 (2014).
Wiig, H. & Swartz, M. A. Interstitial fluid and lymph formation and transport: physiological regulation and roles in inflammation and cancer. Physiol. Rev. 92, 1005–1060 (2012).
Olszewski, W. L. & Engeset, A. Intrinsic contractility of leg lymphatics in man. Preliminary communication. Lymphology 12, 81–84 (1979).
Olszewski, W. L. & Engeset, A. Intrinsic contractility of prenodal lymph vessels and lymph flow in human leg. Am. J. Physiol. 239, H775–783 (1980).
Armenio, S., Cetta, F., Tanzini, G. & Guercia, C. Spontaneous contractility in the human lymph vessels. Lymphology 14, 173–178 (1981).
Muthuchamy, M., Gashev, A., Boswell, N., Dawson, N. & Zawieja, D. Molecular and functional analyses of the contractile apparatus in lymphatic muscle. FASEB J. 17, 920–922 (2003).
Davis, M. J., Davis, A. M., Ku, C. W. & Gashev, A. A. Myogenic constriction and dilation of isolated lymphatic vessels. Am. J. Physiol. Heart Circ. Physiol. 296, H293–H302 (2009).
Wang, W. et al. Inhibition of myosin light chain phosphorylation decreases rat mesenteric lymphatic contractile activity. Am. J. Physiol. Heart Circ. Physiol. 297, H726–H734 (2009).
Ito, M., Nakano, T., Erdodi, F. & Hartshorne, D. J. Myosin phosphatase: structure, regulation and function. Mol. Cell Biochem. 259, 197–209 (2004).
Lee, S., Roizes, S. & von der Weid, P. Y. Distinct roles of L- and T-type voltage-dependent Ca2+ channels in regulation of lymphatic vessel contractile activity. J. Physiol. 592, 5409–5427 (2014).
Liang, Q. et al. Lymphatic endothelial cells efferent to inflamed joints produce iNOS and inhibit lymphatic vessel contraction and drainage in TNF-induced arthritis in mice. Arthritis Res. Ther. 18, 62 (2016).
Munn, L. L. Mechanobiology of lymphatic contractions. Semin. Cell Dev. Biol. 38, 67–74 (2015).
Koning, J. J. & Mebius, R. E. Interdependence of stromal and immune cells for lymph node function. Trends Immunol. 33, 264–270 (2012).
Ding, Y., Xu, J. & Bromberg, J. S. Regulatory T cell migration during an immune response. Trends Immunol. 33, 174–180 (2012).
Germain, R. N., Robey, E. A. & Cahalan, M. D. A decade of imaging cellular motility and interaction dynamics in the immune system. Science 336, 1676–1681 (2012).
Mueller, S. N. & Germain, R. N. Stromal cell contributions to the homeostasis and functionality of the immune system. Nat. Rev. Immunol. 9, 618–629 (2009).
Alitalo, K. The lymphatic vasculature in disease. Nat. Med. 17, 1371–1380 (2011).
Iwami, D., Brinkman, C. C. & Bromberg, J. S. Vascular endothelial growth factor C/vascular endothelial growth factor receptor 3 signaling regulates chemokine gradients and lymphocyte migration from tissues to lymphatics. Transplantation 99, 668–677 (2015).
Abadie, V. et al. Neutrophils rapidly migrate via lymphatics after Mycobacterium bovis BCG intradermal vaccination and shuttle live bacilli to the draining lymph nodes. Blood 106, 1843–1850 (2005).
Cromer, W. et al. Colonic insult impairs lymph flow, increases cellular content of the lymph, alters local lymphatic microenvironment, and leads to sustained inflammation in the rat ileum. Inflamm. Bowel Dis. 21, 1553–1563 (2015).
Pflicke, H. & Sixt, M. Preformed portals facilitate dendritic cell entry into afferent lymphatic vessels. J. Exp. Med. 206, 2925–2935 (2009).
Randolph, G. J., Angeli, V. & Swartz, M. A. Dendritic-cell trafficking to lymph nodes through lymphatic vessels. Nat. Rev. Immunol. 5, 617–628 (2005).
Thomas, S. N. et al. Impaired humoral immunity and tolerance in K14-VEGFR-3-Ig mice that lack dermal lymphatic drainage. J. Immunol. 189, 2181–2190 (2012).
Bouta, E. M. et al. Brief report: Treatment of tumor necrosis factor-transgenic mice with anti-tumor necrosis factor restores lymphatic contractions, repairs lymphatic vessels, and may increase monocyte/macrophage egress. Arthritis Rheumatol. 69, 1187–1193 (2017).
Zheng, W., Aspelund, A. & Alitalo, K. Lymphangiogenic factors, mechanisms, and applications. J. Clin. Invest. 124, 878–887 (2014).
Kim, H., Kataru, R. P. & Koh, G. Y. Inflammation-associated lymphangiogenesis: a double-edged sword? J. Clin. Invest. 124, 936–942 (2014).
Ji, R. C. Macrophages are important mediators of either tumor- or inflammation-induced lymphangiogenesis. Cell. Mol. Life Sci. 69, 897–914 (2012).
Ogata, F. et al. Excess lymphangiogenesis cooperatively induced by macrophages and CD4+ T cells drives the pathogenesis of lymphedema. J. Invest. Dermatol. 136, 706–714 (2016).
Johnson, L. A. & Jackson, D. G. Inflammation-induced secretion of CCL21 in lymphatic endothelium is a key regulator of integrin-mediated dendritic cell transmigration. Int. Immunol. 22, 839–849 (2010).
Liao, S. et al. Impaired lymphatic contraction associated with immunosuppression. Proc. Natl Acad. Sci. USA 108, 18784–18789 (2011).
Aldrich, M. B. & Sevick-Muraca, E. M. Cytokines are systemic effectors of lymphatic function in acute inflammation. Cytokine 64, 362–369 (2013).
Chen, Y. et al. The pro-inflammatory cytokine TNF-alpha inhibits lymphatic pumping via activation of the NF-κB-iNOS signaling pathway. Microcirculation 24, e12364 (2017).
von der Weid, P. Y. & Muthuchamy, M. Regulatory mechanisms in lymphatic vessel contraction under normal and inflammatory conditions. Pathophysiology 17, 263–276 (2010).
Liao, S. & von der Weid, P. Y. Inflammation-induced lymphangiogenesis and lymphatic dysfunction. Angiogenesis 17, 325–334 (2014).
Becker, F. et al. Lymphatic dysregulation in intestinal inflammation: new insights into inflammatory bowel disease pathomechanisms. Lymphology 47, 3–27 (2014).
Gashev, A. A., Davis, M. J., Delp, M. D. & Zawieja, D. C. Regional variations of contractile activity in isolated rat lymphatics. Microcirculation 11, 477–492 (2004).
Scallan, J. P. et al. Independent and interactive effects of preload and afterload on the pump function of the isolated lymphangion. Am. J. Physiol. Heart Circ. Physiol. 303, H809–H824 (2012).
Koller, A., Mizuno, R. & Kaley, G. Flow reduces the amplitude and increases the frequency of lymphatic vasomotion: role of endothelial prostanoids. Am. J. Physiol. 277, R1683–R1689 (1999).
Davis, M. J., Davis, A. M., Lane, M. M., Ku, C. W. & Gashev, A. A. Rate-sensitive contractile responses of lymphatic vessels to circumferential stretch. J. Physiol. 587, 165–182 (2009).
Davis, M. J. et al. Intrinsic increase in lymphangion muscle contractility in response to elevated afterload. Am. J. Physiol. Heart Circ. Physiol. 303, H795–H808 (2012).
Scallan, J. P., J. H. Wolpers, M. Muthuchamy, D. C. Zawieja, A. A. Gashev, M. J. Davis. Independent and interactive effects of preload and afterload on the pump function of the isolated lymphangion. Am. J. Physiol. Heart Circ. Physiol. 303, H809–H824 (2012).
Lynch, P. M., Delano, F. A. & Schmid-Schonbein, G. W. The primary valves in the initial lymphatics during inflammation. Lymphat. Res. Biol. 5, 3–10 (2007).
Kajiya, K. et al. Promotion of lymphatic integrity by angiopoietin-1/Tie2 signaling during inflammation. Am. J. Pathol. 180, 1273–1282 (2012).
Savetsky, I. L. et al. Lymphatic function regulates contact hypersensitivity dermatitis in obesity. J. Invest. Dermatol. 135, 2742–2752 (2015).
Zhou, Q., Wood, R., Schwarz, E. M., Wang, Y. J. & Xing, L. Near-infrared lymphatic imaging demonstrates the dynamics of lymph flow and lymphangiogenesis during the acute versus chronic phases of arthritis in mice. Arthritis Rheum. 62, 1881–1889 (2010).
Meier, T. O. et al. Increased permeability of cutaneous lymphatic capillaries and enhanced blood flow in psoriatic plaques. Dermatology 227, 118–125 (2013).
Kuan, E. L. et al. Collecting lymphatic vessel permeability facilitates adipose tissue inflammation and distribution of antigen to lymph node-homing adipose tissue DCs. J. Immunol. 194, 5200–5210 (2015).
Ivanov, S. et al. CCR7 and IRF4-dependent dendritic cells regulate lymphatic collecting vessel permeability. J. Clin. Invest. 126, 1581–1591 (2016).
Scallan, J. P., Hill, M. A. & Davis, M. J. Lymphatic vascular integrity is disrupted in type 2 diabetes due to impaired nitric oxide signalling. Cardiovasc. Res. 107, 89–97 (2015).
da Fonseca, D. M. et al. Microbiota-dependent sequelae of acute infection compromise tissue-specific immunity. Cell 163, 354–366 (2015).
Kawai, Y., Kaidoh, M., Yokoyama, Y. & Ohhashi, T. Pivotal roles of lymphatic endothelial cell layers in the permeability to hydrophilic substances through collecting lymph vessel walls: effects of inflammatory cytokines. Lymphat. Res. Biol. 12, 124–135 (2014).
Kakei, Y., Akashi, M., Shigeta, T., Hasegawa, T. & Komori, T. Alteration of cell-cell junctions in cultured human lymphatic endothelial cells with inflammatory cytokine stimulation. Lymphat. Res. Biol. 12, 136–143 (2014).
van den Berg, W. B. Animal models of arthritis. What have we learned? J. Rheumatol. Suppl. 72, 7–9 (2005).
Kouskoff, V. et al. Organ-specific disease provoked by systemic autoimmunity. Cell 87, 811–822 (1996).
Keffer, J. et al. Transgenic mice expressing human tumour necrosis factor: a predictive genetic model of arthritis. EMBO J. 10, 4025–4031 (1991).
Proulx, S. T. et al. Longitudinal assessment of synovial, lymph node, and bone volumes in inflammatory arthritis in mice by in vivo magnetic resonance imaging and microfocal computed tomography. Arthritis Rheum. 56, 4024–4037 (2007).
Bouta, E. M. et al. Validation of power Doppler versus contrast-enhanced magnetic resonance imaging quantification of joint inflammation in murine inflammatory arthritis. J. Bone Miner. Res. 30, 690–694 (2015).
Bouta, E. M. et al. Power Doppler ultrasound phenotyping of expanding versus collapsed popliteal lymph nodes in murine inflammatory arthritis. PLoS ONE 8, e73766 (2013).
Li, J. et al. Efficacy of B cell depletion therapy for murine joint arthritis flare is associated with increased lymphatic flow. Arthritis Rheum. 65, 130–138 (2012).
Li, J. et al. CD23+/CD21hi B cell translocation and ipsilateral lymph node collapse is associated with asymmetric arthritic flare in TNF-Tg mice. Arthritis Res. Ther. 13, R138 (2011).
Proulx, S. T. et al. MRI and quantification of draining lymph node function in inflammatory arthritis. Ann. NY Acad. Sci. 1117, 106–123 (2007).
Proulx, S. T. et al. Elucidating bone marrow edema and myelopoiesis in murine arthritis using contrast-enhanced magnetic resonance imaging. Arthritis Rheum. 58, 2019–2029 (2008).
Guo, R. et al. Inhibition of lymphangiogenesis and lymphatic drainage via vascular endothelial growth factor receptor 3 blockade increases the severity of inflammation in a mouse model of chronic inflammatory arthritis. Arthritis Rheum. 60, 2666–2676 (2009).
Zhang, Q. et al. Increased lymphangiogenesis in joints of mice with inflammatory arthritis. Arthritis Res. Ther. 9, R118 (2007).
Li, J. et al. Expanded CD23+/CD21hi B cells in inflamed lymph nodes are associated with the onset of inflammatory-erosive arthritis in TNF-transgenic mice and are targets of anti-CD20 therapy. J. Immunol. 184, 6142–6150 (2010).
Kuzin, II et al. Increased numbers of CD23+CD21hi Bin-like B cells in human reactive and rheumatoid arthritis lymph nodes. Eur. J. Immunol. 46, 1752–1757 (2016).
Bouta, E. M. et al. In vivo quantification of lymph viscosity and pressure in lymphatic vessels and draining lymph nodes of arthritic joints in mice. J. Physiol. 592, 1213–1223 (2014).
Moshkani, S. et al. CD23+CD21highCD1dhigh B cells in inflamed lymph nodes are a locally differentiated population with increased antigen capture and activation potential. J. Immunol. 188, 5944–5953 (2012).
Bouta, E. M. et al. Treatment of TNF-Tg mice with anti-TNF restores lymphatic contraction, repairs lymphatic vessels, and may increase monocyte/macrophage egress. Arthritis Rheumatol. 69, 1187–1193 (2017).
Bouta, E. M. et al. The role of the lymphatic system in inflammatory-erosive arthritis. Semin. Cell Dev. Biol. 38, 90–97 (2015).
Young, A. & Koduri, G. Extra-articular manifestations and complications of rheumatoid arthritis. Best Pract. Res. Clin. Rheumatol. 21, 907–927 (2007).
Robertson, M. D., Hart, F. D., White, W. F., Nuki, G. & Boardman, P. L. Rheumatoid lymphadenopathy. Ann. Rheum. Dis. 27, 253–260 (1968).
Suami, H., Pan, W. R. & Taylor, G. I. Changes in the lymph structure of the upper limb after axillary dissection: radiographic and anatomical study in a human cadaver. Plast. Reconstr. Surg. 120, 982–991 (2007).
Suami, H., Taylor, G. I. & Pan, W. R. The lymphatic territories of the upper limb: anatomical study and clinical implications. Plast. Reconstr. Surg. 119, 1813–1822 (2007).
van de Stadt, L. A. et al. The value of ultrasonography in predicting arthritis in auto-antibody positive arthralgia patients: a prospective cohort study. Arthritis Res. Ther. 12, R98 (2010).
Rahimi, H. et al. Relationship between lymph node volume and pain following certolizumab therapy for rheumatoid arthritis flare: a pilot study. Clin. Med. Insights Arthritis Musculoskelet. Disord. 9, 203–208 (2016).
Foltz, V. et al. Power Doppler ultrasound, but not low-field magnetic resonance imaging, predicts relapse and radiographic disease progression in rheumatoid arthritis patients with low levels of disease activity. Arthritis Rheum. 64, 67–76 (2012).
Kiely, P. D., Bland, J. M., Joseph, A. E., Mortimer, P. S. & Bourke, B. E. Upper limb lymphatic function in inflammatory arthritis. J. Rheumatol 22, 214–217 (1995).
Rahimi, H. et al. Lymphatic imaging to assess rheumatoid flare: mechanistic insights and biomarker potential. Arthritis Res. Ther. 18, 194 (2016).
Benaglio, F. et al. The draining lymph node in rheumatoid arthritis: current concepts and research perspectives. Biomed. Res. Int. 2015, 420251 (2015).
Manzo, A. et al. Power Doppler ultrasonographic assessment of the joint-draining lymph node complex in rheumatoid arthritis: a prospective, proof-of-concept study on treatment with tumor necrosis factor inhibitors. Arthritis Res. Ther. 18, 242 (2016).
Manzo, A. et al. Subclinical remodelling of draining lymph node structure in early and established rheumatoid arthritis assessed by power Doppler ultrasonography. Rheumatology (Oxford) 50, 1395–1400 (2011).
Bingham, C. O. 3rd et al. Developing a standardized definition for disease “flare” in rheumatoid arthritis (OMERACT 9 Special Interest Group). J. Rheumatol 36, 2335–2341 (2009).
Alten, R. et al. Developing a construct to evaluate flares in rheumatoid arthritis: a conceptual report of the OMERACT RA Flare Definition Working Group. J. Rheumatol 38, 1745–1750 (2011).
Hewlett, S. et al. 'I'm hurting, I want to kill myself': rheumatoid arthritis flare is more than a high joint count — an international patient perspective on flare where medical help is sought. Rheumatology (Oxford) 51, 69–76 (2012).
Bartlett, S. J. et al. Identifying core domains to assess flare in rheumatoid arthritis: an OMERACT international patient and provider combined Delphi consensus. Ann. Rheum. Dis. 71, 1855–1860 (2012).
Bykerk, V. P. et al. Establishing a core domain set to measure rheumatoid arthritis flares: report of the OMERACT 11 RA Flare Workshop. J. Rheumatol 41, 799–809 (2014).
Bartlett, S. J. et al. Feasibility and domain validation of rheumatoid arthritis (RA) flare core domain set: report of the OMERACT 2014 RA Flare Group Plenary. J. Rheumatol. 42, 2185–2189 (2015).
Bykerk, V. P. et al. Identifying flares in rheumatoid arthritis: reliability and construct validation of the OMERACT RA Flare Core Domain Set. RMD Open 2, e000225 (2016).
Naredo, E. et al. Longitudinal power Doppler ultrasonographic assessment of joint inflammatory activity in early rheumatoid arthritis: predictive value in disease activity and radiologic progression. Arthritis Rheum. 57, 116–124 (2007).
Szkudlarek, M., Wakefield, R. J., Backhaus, M. & Terslev, L. The discriminatory capacity of ultrasound in rheumatoid arthritis: active versus inactive, early versus advanced, and more. Rheumatology (Oxford) 51 (Suppl. 7), vii6–vii9 (2012).
Patil, P. & Dasgupta, B. Role of diagnostic ultrasound in the assessment of musculoskeletal diseases. Ther. Adv. Musculoskelet Dis. 4, 341–355 (2012).
Tan, Y. K., Ostergaard, M. & Conaghan, P. G. Imaging tools in rheumatoid arthritis: ultrasound versus magnetic resonance imaging. Rheumatology (Oxford) 51 (Suppl. 7), vii36–vii42 (2012).
Ostergaard, M., Pedersen, S. J. & Dohn, U. M. Imaging in rheumatoid arthritis — status and recent advances for magnetic resonance imaging, ultrasonography, computed tomography and conventional radiography. Best Pract. Res. Clin. Rheumatol. 22, 1019–1044 (2008).
Saleem, B. et al. Can flare be predicted in DMARD treated RA patients in remission, and is it important? A cohort study. Ann. Rheum. Dis. 71, 1316–1321 (2012).
Bellis, E. et al. Ultrasound-detected tenosynovitis independently associates with patient-reported flare in patients with rheumatoid arthritis in clinical remission: results from the observational study STARTER of the Italian Society for Rheumatology. Rheumatology (Oxford) 55, 1826–1836 (2016).
Berthelot, J. M. et al. A tool to identify recent or present rheumatoid arthritis flare from both patient and physician perspectives: the 'FLARE' instrument. Ann. Rheum. Dis. 71, 1110–1116 (2012).
Fautrel, B. et al. Validation of FLARE-RA, a self-administered tool to detect recent or current rheumatoid arthritis flare. Arthritis Rheumatol. 69, 309–319 (2017).
Konig, H., Sieper, J. & Wolf, K. J. Rheumatoid arthritis: evaluation of hypervascular and fibrous pannus with dynamic MR imaging enhanced with Gd-DTPA. Radiology 176, 473–477 (1990).
Gaffney, K., Cookson, J., Blake, D., Coumbe, A. & Blades, S. Quantification of rheumatoid synovitis by magnetic-resonance-imaging. Arthritis Rheum. 38, 1610–1617 (1995).
Tamai, K., Yamato, M., Yamaguchi, T. & Ohno, W. Dynamic magnetic-resonance-imaging for the evaluation of synovitis in patients with rheumatoid-arthritis. Arthritis Rheum. 37, 1151–1157 (1994).
Bjorkengren, A. G., Geborek, P., Rydholm, U., Holtas, S. & Petterson, H. MR imaging of the knee in acute rheumatoid arthritis: synovial uptake of gadolinium-DOTA. Am. J. Roentgenol. 155, 329–332 (1990).
Nusman, C. M. et al. Dynamic contrast-enhanced magnetic resonance imaging can play a role in predicting flare in juvenile idiopathic arthritis. Eur. J. Radiol. 88, 77–81 (2017).
Bremander, A. B. I., Petersson, I. F. & Roos, E. Validation of the rheumatoid arthritis outcome score (RAOS) — for the lower extremity. Ann. Rheum. Dis. 62, 364–364 (2003).
Rasmussen, J. C., Tan, I. C., Marshall, M. V., Fife, C. E. & Sevick-Muraca, E. M. Lymphatic imaging in humans with near-infrared fluorescence. Curr. Opin. Biotechnol. 20, 74–82 (2009).
Rasmussen, J. C. et al. Near-infrared fluorescence imaging techniques for non-invasive lymphatic architectural and functional analysis. J. Nucl. Med. 52, 671–671 (2011).
Tan, I. C. et al. Assessment of lymphatic contractile function after manual lymphatic drainage using near-infrared fluorescence imaging. Arch. Phys. Med. Rehab. 92, 756–764 (2011).
Rasmussen, J. C. et al. Human lymphatic architecture and dynamic transport imaged using near-infrared fluorescence. Transl. Oncol. 3, 362–372 (2010).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02680067 (2017).
Kameda, H. et al. Etanercept (ETN) with methotrexate (MTX) is better than ETN monotherapy in patients with active rheumatoid arthritis despite MTX therapy: a randomized trial. Mod. Rheumatol. 20, 531–538 (2010).
Benucci, M. et al. Efficacy and safety of leflunomide or methotrexate plus subcutaneous tumour necrosis factor-alpha blocking agents in rheumatoid arthritis. Int. J. Immunopathol. Pharmacol. 24, 269–274 (2011).
Nam, J. L. et al. A randomised controlled trial of etanercept and methotrexate to induce remission in early inflammatory arthritis: the EMPIRE trial. Ann. Rheum. Dis. 73, 1027–1036 (2014).
Cohen, S. B. et al. Rituximab for rheumatoid arthritis refractory to anti-tumor necrosis factor therapy: results of a multicenter, randomized, double-blind, placebo-controlled, phase III trial evaluating primary efficacy and safety at twenty-four weeks. Arthritis Rheum. 54, 2793–2806 (2006).
Rosengren, S. et al. Elevated autoantibody content in rheumatoid arthritis synovia with lymphoid aggregates and the effect of rituximab. Arthritis Res. Ther. 10, R105 (2008).
Smeets, T. J., Kraan, M. C., van Loon, M. E. & Tak, P. P. Tumor necrosis factor α blockade reduces the synovial cell infiltrate early after initiation of treatment, but apparently not by induction of apoptosis in synovial tissue. Arthritis Rheum. 48, 2155–2162 (2003).
Wijbrandts, C. A. et al. Analysis of apoptosis in peripheral blood and synovial tissue very early after initiation of infliximab treatment in rheumatoid arthritis patients. Arthritis Rheum. 58, 3330–3339 (2008).
Herenius, M. M. et al. Monocyte migration to the synovium in rheumatoid arthritis patients treated with adalimumab. Ann. Rheum. Dis. 70, 1160–1162 (2011).
Kontoyiannis, D., Pasparakis, M., Pizarro, T. T., Cominelli, F. & Kollias, G. Impaired on/off regulation of TNF biosynthesis in mice lacking TNF AU-rich elements: implications for joint and gut-associated immunopathologies. Immunity 10, 387–398 (1999).
Angeli, V. et al. B cell-driven lymphangiogenesis in inflamed lymph nodes enhances dendritic cell mobilization. Immunity 24, 203–215 (2006).
Zhou, Q. et al. Vascular endothelial growth factor C attenuates joint damage in chronic inflammatory arthritis by accelerating local lymphatic drainage in mice. Arthritis Rheum. 63, 2318–2328 (2011).
Zhang, Y. et al. Activation of vascular endothelial growth factor receptor-3 in macrophages restrains TLR4-NF-κB signaling and protects against endotoxin shock. Immunity 40, 501–514 (2014).
Evans, C. H. et al. Gene therapy for rheumatoid arthritis. Expert Opin. Biol. Ther. 1, 971–978 (2001).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02994771 (2017).
Liang, Q. Q., Shi, Q., Wood, R. W., Xing, L. P. & Wang, Y. J. Peri-articular lymphatic system and “Bi” theory of Chinese medicine in the pathogenesis and treatment of arthritis. Chin. J. Integr. Med. 21, 648–655 (2015).
Chen, Y. et al. Du-Huo-Ji-Sheng-Tang attenuates inflammation of TNF-Tg mice related to promoting lymphatic drainage function. Evid. Based Complement. Alternat. Med. 2016, 7067691 (2016).
Li, J. et al. Total saponins of panaxnotoginseng promotes lymphangiogenesis by activation VEGF-C expression of lymphatic endothelial cells. J. Ethnopharmacol. 193, 293–302 (2016).
Seymour, M. et al. Ultrasonographic measures of synovitis in an early phase clinical trial: a double-blind, randomised, placebo and comparator controlled phase IIa trial of GW274150 (a selective inducible nitric oxide synthase inhibitor) in rheumatoid arthritis. Clin. Exp. Rheumatol 30, 254–261 (2012).
Vitecek, J., Lojek, A., Valacchi, G. & Kubala, L. Arginine-based inhibitors of nitric oxide synthase: therapeutic potential and challenges. Mediators Inflamm. 2012, 318087 (2012).
Connor, J. R. et al. Suppression of adjuvant-induced arthritis by selective inhibition of inducible nitric oxide synthase. Eur. J. Pharmacol. 273, 15–24 (1995).
Iliff, J. J. et al. A paravascular pathway facilitates CSF flow through the brain parenchyma and the clearance of interstitial solutes, including amyloid β. Sci. Transl Med. 4, 147ra111 (2012).
Aspelund, A. et al. The Schlemm's canal is a VEGF-C/VEGFR-3–responsive lymphatic-like vessel. J. Clin. Invest. 124, 3975–3986 (2014).
Wiig, H. et al. Immune cells control skin lymphatic electrolyte homeostasis and blood pressure. J. Clin. Invest. 123, 2803–2815 (2013).
Klotz, L. et al. Cardiac lymphatics are heterogeneous in origin and respond to injury. Nature 522, 62–67 (2015).
Hoshida, T. et al. Imaging steps of lymphatic metastasis reveals that vascular endothelial growth factor-C increases metastasis by increasing delivery of cancer cells to lymph nodes: therapeutic implications. Cancer Res. 66, 8065–8075 (2006).
Baluk, P. et al. Pathogenesis of persistent lymphatic vessel hyperplasia in chronic airway inflammation. J. Clin. Invest. 115, 247–257 (2005).
Visuri, M. T. et al. VEGF-C and VEGF-C156S in the pro-lymphangiogenic growth factor therapy of lymphedema: a large animal study. Angiogenesis 18, 313–326 (2015).
Tervala, T. V. et al. Growth factor therapy and lymph node graft for lymphedema. J. Surg. Res. 196, 200–207 (2015).
Saaristo, A. et al. Lymphangiogenic gene therapy with minimal blood vascular side effects. J. Exp. Med. 196, 719–730 (2002).
Tammela, T. et al. Therapeutic differentiation and maturation of lymphatic vessels after lymph node dissection and transplantation. Nat. Med. 13, 1458–1466 (2007).
Tian, W. et al. Leukotriene B4 antagonism ameliorates experimental lymphedema. Sci. Transl. Med. 9, eaal3920 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02700529 (2017).
Cursiefen, C. et al. Lymphatic vessels in vascularized human corneas: immunohistochemical investigation using LYVE-1 and podoplanin. Invest. Ophthalmol. Vis. Sci. 43, 2127–2135 (2002).
Dietrich, T. et al. Cutting edge: lymphatic vessels, not blood vessels, primarily mediate immune rejections after transplantation. J. Immunol. 184, 535–539 (2010).
Chen, W. S. et al. Pathological lymphangiogenesis is modulated by galectin-8-dependent crosstalk between podoplanin and integrin-associated VEGFR-3. Nat. Commun. 7, 11302 (2016).
Steven, P., Bock, F., Huttmann, G. & Cursiefen, C. Intravital two-photon microscopy of immune cell dynamics in corneal lymphatic vessels. PLoS ONE 6, e26253 (2011).
Blum, K. S. et al. Chronic high-fat diet impairs collecting lymphatic vessel function in mice. PLoS ONE 9, e94713 (2014).
Rasmussen, J. C. et al. An abnormal lymphatic phenotype is associated with subcutaneous adipose tissue deposits in Dercum's disease. Obesity 22, 2186–2192 (2014).
Rasmussen, J. C. et al. Lymphatic transport in patients with chronic venous insufficiency and venous leg ulcers following sequential pneumatic compression. Journal of vascular surgery. Ven. Lymphat. Disord. 4, 9–17 (2016).
Aldrich, M. B. et al. Lymphatic abnormalities in the normal contralateral arms of subjects with breast cancer-related lymphedema as assessed by near-infrared fluorescent imaging. Biomed. Opt. Express 3, 1256–1265 (2012).
Mathias, R. & von der Weid, P. Y. Involvement of the NO-cGMP-K(ATP) channel pathway in the mesenteric lymphatic pump dysfunction observed in the guinea pig model of TNBS-induced ileitis. Am. J. Physiol. Gastrointest. Liver Physiol. 304, G623–G634 (2013).
Tavian, D. et al. FOXC2 disease-mutations identified in lymphedema-distichiasis patients cause both loss and gain of protein function. Oncotarget 7, 54228–54239 (2016).
Butler, C., Osterberg, C., Horvai, A. & Breyer, B. Milroy's disease and scrotal lymphoedema: pathological insight. BMJ Case Rep. http://dx.doi.org/10.1136/bcr-2016-215396 (2016).
Harvey, N. L. et al. Lymphatic vascular defects promoted by Prox1 haploinsufficiency cause adult-onset obesity. Nat. Genet. 37, 1072–1081 (2005).
Karkkainen, M. J. et al. A model for gene therapy of human hereditary lymphedema. Proc. Natl Acad. Sci. USA 98, 12677–12682 (2001).
Suami, H. Lymphosome concept: Anatomical study of the lymphatic system. J. Surg. Oncol. 115, 13–17 (2017).
Acknowledgements
E.M.B. and R.D.B. are supported by a training grant from the NIH (T32 AR053459). H.R. is supported by an NIH grant (K08 AR067885). L.X. is supported by NIH grants (AR069789 and AR063650), a University of Rochester CTSA award (UL1 TR000042), a grant from the National Natural Science Foundation of China (grant 81220108027), and a grant from the Lymphatic Malformation Institute. R.W.W. is supported by an NIH grant (AR061307). C.T.R. is supported by NIH grants (R01 AR056702 and R01 AR069000). E.M.S. is supported by NIH grants (P30 AR069655 and R01 AR056702).
Author information
Authors and Affiliations
Contributions
E.M.B., R.D.B., H.R., L.X., R.W.W., C.O.B. and E.M.S. researched data for the article, made substantial contributions to discussion of its content, wrote the article and reviewed and/or edited the manuscript before submission. C.T.R. made a substantial contribution to discussion of the content, wrote the article and reviewed and/or edited the manuscript before submission. E.M.B. and R.D.B. contributed equally to this work.
Corresponding author
Ethics declarations
Competing interests
C.T.R. declares that he has received consulting fees and research support from UCB Pharmaceuticals. E.M.B., R.W.W., L.X. and E.M.S. declare that they have applied for patents related to the content of the manuscript. R.D.B. and H.R. declare no competing interests.
Supplementary information
Supplementary video S1
NIR-ICG imaging of lymphatic functioning of a TNFtg mouse in the 'expansion' phase. At 3 months of age, TNF transgenic (TNFtg) mice have inflammatory arthiritis in their ankle joints, and expanding popliteal lymph nodes as determined by power Doppler ultrasonography. This video demonstrates lymphatic drainage in the lower limb of a 3-month-old TNFtg mouse by near-infrared indocyanine green (NIR-ICG) imaging. This 20-minute video was taken 60 minutes after ICG injection, and is shown at 20× real time. Note the consistent contractions (∼1 per minute) throughout the imaging period. (AVI 2410 kb)
Supplementary video S2
NIR-ICG imaging of lymphatic dysfunctioning in a TNFtg mouse during the 'collapsed' phase. Indocyanine green (ICG) was injected into the footpad of an 8-month-old TNF transgenic (TNFtg) mouse with a collapsed popliteal lymph node, which was phenotyped using power Doppler ultrasonography. Near-infrared (NIR) imaging was performed to demonstrate lymphatic dysfunction in the lower limb, which was affected by advanced inflammatory arthritis. This 20-minute video was taken 60 minutes after ICG injection, and is shown at 20× real time. Note the complete absence of lymphatic vessel contractions, and lack of popliteal lymph node signal enhancement, throughout the imaging period. (AVI 1475 kb)
Supplementary video S3
Injection of ICG into the web spaces of a healthy volunteer, and indentification of lymphatic vessels adjacent to veins in the hands. Indocyanine green (ICG) was injected into the web spaces of a heathly human volunteer. This video (shown at 4× real time) shows the injection procedure and the ICG entering the lymphatic system directly adjacent to the major veins of the dorsal hand. Note the dramatic uptake of ICG in lymphatic vessels efferent to the first and second web spaces, relative to ICG uptake in the third and fourth web spaces, potentially owing to differences in interstial pressure from the injection volume and/or proximatity to the intial lymphatic bed. The video also shows that manipulation of the web space during removal of excess iodine at the injection site increases interstial pressure, pushing ICG into the lymphatics as well (fourth web space). (AVI 14679 kb)
Supplementary video S4
Quantification of cephalic lymphatic vessel contrations at the wrist. Indocyanine green (ICG) was injected into the web spaces of a healthy human volunteer. Approximately 15 minutes later, near-infrared imaging was performed to quantify lymphatic vessel contractions. This 10-minute video (shown at 20× real time) shows lymphatic drainage in the vessels of the dorsal hand. To quantify cephalic lymphatic vessel contractions, a region of interest (ROI) is defined (green box), and the signal intensity within the ROI is quantified in real time (graph of mean signal intensity over time). Note how the dye moves as a bolus to collection points (presumed valves), and then moves proximally after a contraction. (MP4 13587 kb)
Supplementary video S5
Quantification of the cephalic lymphatic vessel at the antecubital fossa. Approximately 40 minutes after after injection of indocyanine green (ICG) into the web spaces of a healthy human volunteer, near-infrared imaging was performed to quantify lymphatic contractions in the forearm. This 10-minute video (shown at 20× real time) shows lymphatic activity in the vessels of the antecubital fossa. Region of interest (ROI; indicated by the green box) quantification of contraction frequency of the cephalic lymphatic vessel was also performed. Note how the dye moves as a bolus as it crosses the antecubital fossa. (MP4 13740 kb)
Supplementary video S6
Sildenafil (a short-acting PDE5 inhibitor) increases ICG uptake into collateral lymphatic vessels in a wild-type mouse. Indocyanine green (ICG) was injected into the footpad of a healthy wild-type mouse, and near-infrared (NIR) imaging was performed to assess lymphatic drainage after intraperitoneal administration of sildenafil (12 mg/kg), a short-acting inhibitor of phosphodiesterase 5 (PDE5). The video (shown at 20× real time) was taken 40 minutes after ICG injection, and 20 minutes after the sildenafil injection. Note the ICG filled collateral lymphatic vessels following sildenafil injection, indicating lymphatic rerouting. (MOV 15521 kb)
Supplementary video S7
NIR-ICG imaging of lymphatic functioning in a healthy mouse. Indocyanine green (ICG) was injected into the footpad of a wild-type mouse, and was imaged by near-infrared imaging to enable visualization of the lymphatic vessels. This 10-minute video (shown at 20× real time) was taken 40 minutes after ICG injection. Note that the ICG travels from the injection site in the footpad (bottom) to the popoliteal lymph node (top) in two lymphatic vessels that contract with a normal frequency (once per minute). (MOV 1504 kb)
Rights and permissions
About this article
Cite this article
Bouta, E., Bell, R., Rahimi, H. et al. Targeting lymphatic function as a novel therapeutic intervention for rheumatoid arthritis. Nat Rev Rheumatol 14, 94–106 (2018). https://doi.org/10.1038/nrrheum.2017.205
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2017.205
This article is cited by
-
Hepatorenal pathologies in TNF-transgenic mouse model of rheumatoid arthritis are alleviated by anti-TNF treatment
Arthritis Research & Therapy (2023)
-
A spike in circulating cytokines TNF-α and TGF-β alters barrier function between vascular and musculoskeletal tissues
Scientific Reports (2023)
-
Single-cell transcriptomics of popliteal lymphatic vessels and peripheral veins reveals altered lymphatic muscle and immune cell populations in the TNF-Tg arthritis model
Arthritis Research & Therapy (2022)
-
Persistent popliteal lymphatic muscle cell coverage defects despite amelioration of arthritis and recovery of popliteal lymphatic vessel function in TNF-Tg mice following anti-TNF therapy
Scientific Reports (2022)
-
Current understanding of osteoarthritis pathogenesis and relevant new approaches
Bone Research (2022)