Key Points
-
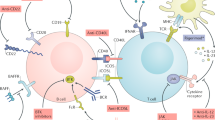
TNF ligand superfamily member 13B (also known as B cell-activating factor of the TNF family [BAFF]) and TNF ligand superfamily member 13 (also known as a proliferation-inducing ligand [APRIL]) are important modulators of autoimmunity
-
Data indicate that alteration of the BAFF/APRIL system affects the capacity of the innate immune system to regulate B-cell activation
-
BAFF and type I interferons function together in systemic lupus erythematosus (SLE) pathogenesis in both a Toll-like receptor-dependent and independent manner
-
Defining the clinical manifestations of disease related to alterations of the BAFF/APRIL system might help to stratify patients with SLE into subgroups that are more likely to benefit from anti-BAFF treatment
-
Differences in the molecular forms of BAFF might affect the efficacy of BAFF-specific therapies
Abstract
Systemic lupus erythematosus (SLE) is characterized by multisystem immune-mediated injury in the setting of autoimmunity to nuclear antigens. The clinical heterogeneity of SLE, the absence of universally agreed clinical trial end points, and the paucity of validated therapeutic targets have, historically, contributed to a lack of novel treatments for SLE. However, in 2011, a therapeutic monoclonal antibody that neutralizes the cytokine TNF ligand superfamily member 13B (also known as B-cell-activating factor of the TNF family [BAFF]), belimumab, became the first targeted therapy for SLE to have efficacy in a randomized clinical trial. Because of its specificity, the efficacy of belimumab provides an opportunity to increase understanding of SLE pathophysiology. Although belimumab depletes B cells, this effect is not as powerful as that of other B-cell-directed therapies that have not been proven efficacious in randomized clinical trials. In this article, therefore, we review results suggesting that neutralizing BAFF can have effects on the immune system other than depletion of B cells. We also identify aspects of the BAFF system for which data in relation to SLE are still missing, and we suggest studies to investigate the pathogenesis of SLE and ways to refine anti-BAFF therapies. The role of a related cytokine, TNF ligand superfamily member 13 (also known as a proliferation-inducing ligand [APRIL]) in SLE is much less well understood, and hence this review focuses on BAFF.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Tsokos, G. C. Systemic lupus erythematosus. N. Eng. J. Med. 365, 2110–2121 (2011).
Mackay, F. et al. Mice transgenic for BAFF develop lymphocytic disorders along with autoimmune manifestations. J. Exp. Med. 190, 1697–1710 (1999).
Schneider, P. et al. BAFF, a novel ligand of the tumor necrosis factor family, stimulates B cell growth. J. Exp. Med. 189, 1747–1756 (1999).
Batten, M. et al. BAFF mediates survival of peripheral immature B lymphocytes. J. Exp. Med. 192, 1453–1466 (2000).
Navarra, S. V. et al. Efficacy and safety of belimumab in patients with active systemic lupus erythematosus: a randomised, placebo-controlled, phase 3 trial. Lancet 377, 721–731 (2011).
Furie, R. et al. A phase 3, randomized, placebo-controlled study of belimumab, a monoclonal antibody that inhibits BLyS, in patients with systemic lupus erythematosus. Arthritis Rheum. 63, 3918–3930 (2011).
Vincent, F. B., Northcott, M., Hoi, A., Mackay, F. & Morand, E. F. Association of serum B cell activating factor from the tumour necrosis factor family (BAFF) and a proliferation-inducing ligand (APRIL) with central nervous system and renal disease in systemic lupus erythematosus. Lupus 22, 873–884 (2013).
Figgett, W. A. et al. The TACI receptor regulates T-cell-independent marginal zone B cell responses through innate activation-induced cell death. Immunity 39, 573–583 (2013).
Zhang, L. et al. Identification of BLyS (B lymphocyte stimulator), a non-myelin-associated protein, as a functional ligand for Nogo-66 receptor. J. Neurosci. 29, 6348–6352 (2009).
Liu, Y. et al. Crystal structure of sTALL-1 reveals a virus-like assembly of TNF family ligands. Cell 108, 383–394 (2002).
Cachero, T. G. et al. Formation of virus-like clusters is an intrinsic property of the tumor necrosis factor family member BAFF (B cell activating factor). Biochemistry 45, 2006–2013 (2006).
Vincent, F. B., Morand, E. F. & Mackay, F. BAFF and innate immunity: new therapeutic targets for systemic lupus erythematosus. Immunol. Cell Biol. 90, 293–303 (2012).
Vincent, F. B., Saulep-Easton, D., Figgett, W. A., Fairfax, K. A. & Mackay, F. The BAFF/APRIL system: Emerging functions beyond B cell biology and autoimmunity. Cytokine Growth Factor Rev. 24, 203–215 (2013).
Gorelik, L. et al. Normal B cell homeostasis requires B cell activation factor production by radiation-resistant cells. J. Exp. Med. 198, 937–945 (2003).
Moore, P. A. et al. BLyS: member of the tumor necrosis factor family and B lymphocyte stimulator. Science 285, 260–263 (1999).
Hahne, M. et al. APRIL, a new ligand of the tumor necrosis factor family, stimulates tumor cell growth. J. Exp. Med. 188, 1185–1190 (1998).
López-Fraga, M., Fernandez, R., Albar, J. P. & Hahne, M. Biologically active APRIL is secreted following intracellular processing in the Golgi apparatus by furin convertase. EMBO Rep. 2, 945–951 (2001).
Maia, S. et al. Aberrant expression of functional BAFF-system receptors by malignant B-cell precursors impacts leukemia cell survival. PLoS ONE 6, e20787 (2011).
Pradet-Balade, B. et al. An endogenous hybrid mRNA encodes TWE-PRIL, a functional cell surface TWEAK-APRIL fusion protein. EMBO J. 21, 5711–5720 (2002).
Bossen, C. et al. TACI, unlike BAFF-R, is solely activated by oligomeric BAFF and APRIL to support survival of activated B cells and plasmablasts. Blood 111, 1004–1012 (2008).
Dillon, S. R. et al. B-lymphocyte stimulator/a proliferation-inducing ligand heterotrimers are elevated in the sera of patients with autoimmune disease and are neutralized by atacicept and B-cell maturation antigen-immunoglobulin. Arthritis Res. Ther. 12, R48 (2010).
Roschke, V. et al. BLyS and APRIL form biologically active heterotrimers that are expressed in patients with systemic immune-based rheumatic diseases. J. Immunol. 169, 4314–4321 (2002).
Bossen, C. et al. Mutation of the BAFF furin cleavage site impairs B-cell homeostasis and antibody responses. E. J. Immunol. 41, 787–797 (2011).
Le Pottier, L. et al. New ELISA for B cell-activating factor. Clin. Chem. 55, 1843–1851 (2009).
Gavin, A. L. et al. ΔBAFF, a splice isoform of BAFF, opposes full-length BAFF activity in vivo in transgenic mouse models. J. Immunol. 175, 319–328 (2005).
Gavin, A. L., Aït-Azzouzene, D., Ware, C. F. & Nemazee, D. ΔBAFF, an alternate splice isoform that regulates receptor binding and biopresentation of the B cell survival cytokine, BAFF. J. Biol. Chem. 278, 38220–38228 (2003).
Krumbholz, M. et al. BAFF is produced by astrocytes and up-regulated in multiple sclerosis lesions and primary central nervous system lymphoma. J. Exp. Med. 201, 195–200 (2005).
Lopez De Padilla, C. M. et al. BAFF expression correlates with idiopathic inflammatory myopathy disease activity measures and autoantibodies. J. Rheumatol. 40, 294–302 (2013).
Gross, J. A. et al. TACI-Ig neutralizes molecules critical for B cell development and autoimmune disease. impaired B cell maturation in mice lacking BLyS. Immunity 15, 289–302 (2001).
Gross, J. A. et al. TACI and BCMA are receptors for a TNF homologue implicated in B-cell autoimmune disease. Nature 404, 995–999 (2000).
Mackay, F. & Schneider, P. Cracking the BAFF code. Nat. Rev. Immunol. 9, 491–502 (2009).
Huard, B., Tran, N. L., Benkhoucha, M., Manzin-Lorenzi, C. & Santiago-Raber, M. L. Selective APRIL blockade delays systemic lupus erythematosus in mouse. PLoS ONE 7, e31837 (2012).
Jacob, C. O. et al. Dispensability of APRIL to the development of systemic lupus erythematosus in NZM 2328 mice. Arthritis Rheum. 64, 1610–1619 (2012).
Ginzler, E. M. et al. Atacicept in combination with MMF and corticosteroids in lupus nephritis: results of a prematurely terminated trial. Arthritis Res. Ther. 14, R33 (2012).
Baker, K. P. et al. Generation and characterization of LymphoStat-B, a human monoclonal antibody that antagonizes the bioactivities of B lymphocyte stimulator. Arthritis Rheum. 48, 3253–3265 (2003).
Bombardier, C., Gladman, D. D., Urowitz, M. B., Caron, D. & Chang, C. H. Derivation of the SLEDAI. A disease activity index for lupus patients. The Committee on Prognosis Studies in SLE. Arthritis Rheum. 35, 630–640 (1992).
Hay, E. M. et al. The BILAG index: a reliable and valid instrument for measuring clinical disease activity in systemic lupus erythematosus. Q. J. Med. 86, 447–458 (1993).
Furie, R. A. et al. Novel evidence-based systemic lupus erythematosus responder index. Arthritis Rheum. 61, 1143–1151 (2009).
Strand, V. et al. Improvements in health-related quality of life with belimumab, a B-lymphocyte stimulator-specific inhibitor, in patients with autoantibody-positive systemic lupus erythematosus from the randomised controlled BLISS trials. Ann. Rheum. Dis. 10.1136/annrheumdis-2012-202865 (2013).
Wallace, D. J. et al. Safety profile of belimumab: pooled data from placebo-controlled phase 2 and 3 studies in patients with systemic lupus erythematosus. Lupus 22, 144–154 (2013).
Stohl, W. et al. Belimumab reduces autoantibodies, normalizes low complement levels, and reduces select B cell populations in patients with systemic lupus erythematosus. Arthritis Rheum. 64, 2328–2337 (2012).
van Vollenhoven, R. F. et al. Belimumab in the treatment of systemic lupus erythematosus: high disease activity predictors of response. Ann. Rheum. Dis. 71, 1343–1349 (2012).
Chatham, W. W. et al. Effect of belimumab on vaccine antigen antibodies to influenza, pneumococcal, and tetanus vaccines in patients with systemic lupus erythematosus in the BLISS-76 trial. J. Rheumatol. 39, 1632–1640 (2012).
Petri, M. et al. Association of plasma B lymphocyte stimulator levels and disease activity in systemic lupus erythematosus. Arthritis Rheum. 58, 2453–2459 (2008).
Hegazy, M., Darwish, H., Darweesh, H., El-Shehaby, A. & Emad, Y. Raised serum level of APRIL in patients with systemic lupus erythematosus: correlations with disease activity indices. Clin. Immunol. 135, 118–124 (2010).
Stohl, W. et al. Inverse association between circulating APRIL levels and serological and clinical disease activity in patients with systemic lupus erythematosus. Ann. Rheum. Dis. 63, 1096–1103 (2004).
Morel, J. et al. Serum levels of tumour necrosis factor family members a proliferation-inducing ligand (APRIL) and B lymphocyte stimulator (BLyS) are inversely correlated in systemic lupus erythematosus. Ann. Rheum. Dis. 68, 997–1002 (2009).
Stohl, W. et al. B lymphocyte stimulator overexpression in patients with systemic lupus erythematosus: longitudinal observations. Arthritis Rheum. 48, 3475–3486 (2003).
Zhang, J. et al. Cutting edge: a role for B lymphocyte stimulator in systemic lupus erythematosus. J. Immunol. 166, 6–10 (2001).
Collins, C. E. et al. B lymphocyte stimulator (BLyS) isoforms in systemic lupus erythematosus: disease activity correlates better with blood leukocyte BLyS mRNA levels than with plasma BLyS protein levels. Arthritis Res. Ther. 8, R6 (2006).
Petri, M. A. et al. Baseline predictors of systemic lupus erythematosus flares: Data from the combined placebo groups in the Phase 3 belimumab trials. Arthritis Rheum. 65, 2143–2153 (2013).
Carter, L. M., Isenberg, D. A. & Ehrenstein, M. R. Elevated serum B-cell activating factor (BAFF/BLyS) is associated with rising anti-dsDNA antibody levels and flare following B-cell depletion therapy in systemic lupus erythematosus. Arthritis Rheum. 65, 2672–2679 (2013).
Cheema, G. S., Roschke, V., Hilbert, D. M. & Stohl, W. Elevated serum B lymphocyte stimulator levels in patients with systemic immune-based rheumatic diseases. Arthritis Rheum. 44, 1313–1319 (2001).
Baechler, E. C. et al. Interferon-inducible gene expression signature in peripheral blood cells of patients with severe lupus. Proc. Natl Acad. Sci. USA 100, 2610–2615 (2003).
Vincent, F. B., Northcott, M., Hoi, A., Mackay, F. & Morand, E. F. Clinical associations of serum interleukin-17 in systemic lupus erythematosus. Arthritis Res. Ther. 15, R97 (2013).
Kirou, K. A. et al. Activation of the interferon-α pathway identifies a subgroup of systemic lupus erythematosus patients with distinct serologic features and active disease. Arthritis Rheum. 52, 1491–1503 (2005).
Feng, X. et al. Association of increased interferon-inducible gene expression with disease activity and lupus nephritis in patients with systemic lupus erythematosus. Arthritis Rheum. 54, 2951–2962 (2006).
Landolt-Marticorena, C. et al. Lack of association between the interferon-alpha signature and longitudinal changes in disease activity in systemic lupus erythematosus. Ann. Rheum. Dis. 68, 1440–1446 (2009).
Bertsias, G. K., Salmon, J. E. & Boumpas, D. T. Therapeutic opportunities in systemic lupus erythematosus: state of the art and prospects for the new decade. Ann. Rheum. Dis. 69, 1603–1611 (2010).
Rana, A. et al. Gene expression of cytokines (TNF-α, IFN-γ), serum profiles of IL-17 and IL-23 in paediatric systemic lupus erythematosus. Lupus 21, 1105–1112 (2012).
Vuyyuru, R., Mohan, C., Manser, T. & Rahman, Z. S. The lupus susceptibility locus Sle1 breaches peripheral B cell tolerance at the antibody-forming cell and germinal center checkpoints. J. Immunol. 183, 5716–5727 (2009).
Liu, K. et al. Sle3 and Sle5 can independently couple with Sle1 to mediate severe lupus nephritis. Genes Immun. 8, 634–645 (2007).
Dooley, M. et al. Effect of belimumab treatment on renal outcomes: results from the phase 3 belimumab clinical trials in patients with SLE. Lupus 22, 63–72 (2013).
Manzi, S. et al. Effects of belimumab, a B lymphocyte stimulator-specific inhibitor, on disease activity across multiple organ domains in patients with systemic lupus erythematosus: combined results from two phase III trials. Ann. Rheum. Dis. 71, 1833–1838 (2012).
Vincent, F. B., Bourke, P., Morand, E. F., Mackay, F. & Bossingham, D. Focus on systemic lupus erythematosus in Indigenous Australians: towards a better understanding of autoimmune diseases. Intern. Med. J. 43, 227–234 (2013).
Borchers, A. T., Naguwa, S. M., Shoenfeld, Y. & Gershwin, M. E. The geoepidemiology of systemic lupus erythematosus. Autoimmun. Rev. 9, A277–A287 (2010).
Thumboo, J. et al. A comparative study of the clinical manifestations of systemic lupus erythematosus in Caucasians in Rochester, Minnesota, and Chinese in Singapore, from 1980 to 1992. Arthritis Rheum. 45, 494–500 (2001).
Golder, V., Connelly, K., Staples, M., Morand, E. & Hoi, A. Association of Asian ethnicity with disease activity in SLE: an observational study from the Monash Lupus Clinic. Lupus 22, 1425–1430 (2013).
Ritterhouse, L. L. et al. B lymphocyte stimulator levels in systemic lupus erythematosus: higher circulating levels in African American patients and increased production after influenza vaccination in patients with low baseline levels. Arthritis Rheum. 63, 3931–3941 (2011).
Kawasaki, A., Tsuchiya, N., Fukazawa, T., Hashimoto, H. & Tokunaga, K. Analysis on the association of human BLYS (BAFF, TNFSF13B) polymorphisms with systemic lupus erythematosus and rheumatoid arthritis. Genes Immun. 3, 424–429 (2002).
Eilertsen, G. O., Van Ghelue, M., Strand, H. & Nossent, J. C. Increased levels of BAFF in patients with systemic lupus erythematosus are associated with acute-phase reactants, independent of BAFF genetics: a case–control study. Rheumatology 50, 2197–2205 (2011).
Zayed, R. A. et al. B-cell activating factor promoter polymorphisms in Egyptian patients with systemic lupus erythematosus. Ann. Clin. Lab. Sci. 43, 289–294 (2013).
Koyama, T. et al. A novel polymorphism of the human APRIL gene is associated with systemic lupus erythematosus. Rheumatology (Oxford) 42, 980–985 (2003).
Lee, Y. H., Ota, F., Kim-Howard, X., Kaufman, K. M. & Nath, S. K. APRIL polymorphism and systemic lupus erythematosus (SLE) susceptibility. Rheumatology (Oxford) 46, 1274–1276 (2007).
Kawasaki, A. et al. Role of APRIL (TNFSF13) polymorphisms in the susceptibility to systemic lupus erythematosus in Japanese. Rheumatology (Oxford) 46, 776–782 (2007).
Furuya, T., Koga, M., Hikami, K., Kawasaki, A. & Tsuchiya, N. Effects of APRIL (TNFSF13) polymorphisms and splicing isoforms on the secretion of soluble APRIL. Mod. Rheumatol. 22, 541–549 (2012).
Jiang, C., Loo, W. M., Greenley, E. J., Tung, K. S. & Erickson, L. D. B cell maturation antigen deficiency exacerbates lymphoproliferation and autoimmunity in murine lupus. J. Immunol. 186, 6136–6147 (2011).
Jacob, C. O. et al. Development of systemic lupus erythematosus in NZM 2328 mice in the absence of any single BAFF receptor. Arthritis Rheum. 65, 1043–1054 (2013).
Groom, J. R. et al. BAFF and MyD88 signals promote a lupus-like disease independent of T cells. J. Exp. Med. 204, 1959–1971 (2007).
Thangarajh, M., Masterman, T., Hillert, J., Moerk, S. & Jonsson, R. A proliferation-inducing ligand (APRIL) is expressed by astrocytes and is increased in multiple sclerosis. Scand. J. Immunol. 65, 92–98 (2007).
George-Chandy, A., Trysberg, E. & Eriksson, K. Raised intrathecal levels of APRIL and BAFF in patients with systemic lupus erythematosus: relationship to neuropsychiatric symptoms. Arthritis Res. Ther. 10, R97 (2008).
Bennett, L. et al. Interferon and granulopoiesis signatures in systemic lupus erythematosus blood. J. Exp. Med. 197, 711–723 (2003).
Peterson, K. S. et al. Characterization of heterogeneity in the molecular pathogenesis of lupus nephritis from transcriptional profiles of laser-captured glomeruli. J. Clin. Invest. 113, 1722–1733 (2004).
Ytterberg, S. R. & Schnitzer, T. J. Serum interferon levels in patients with systemic lupus erythematosus. Arthritis Rheum. 25, 401–406 (1982).
Kim, T. et al. Serum levels of interferons in patients with systemic lupus erythematosus. Clin. Exp. Immunol. 70, 562–569 (1987).
Rönnblom, L. Potential role of IFNalpha in adult lupus. Arthritis research & therapy 12 (Suppl. 1), S3 (2010).
Lovgren, T., Eloranta, M. L., Bave, U., Alm, G. V. & Ronnblom, L. Induction of interferon-α production in plasmacytoid dendritic cells by immune complexes containing nucleic acid released by necrotic or late apoptotic cells and lupus IgG. Arthritis Rheum. 50, 1861–1872 (2004).
Vallin, H., Blomberg, S., Alm, G. V., Cederblad, B. & Rönnblom, L. Patients with systemic lupus erythematosus (SLE) have a circulating inducer of interferon-α (IFN-α) production acting on leucocytes resembling immature dendritic cells. Clin. Exp. Immunol. 115, 196–202 (1999).
Panchanathan, R. & Choubey, D. Murine BAFF expression is up-regulated by estrogen and interferons: implications for sex bias in the development of autoimmunity. Mol. Immunol. 53, 15–23 (2013).
Litinskiy, M. B. et al. DCs induce CD40-independent immunoglobulin class switching through BLyS and APRIL. Nat. Immunol. 3, 822–829 (2002).
Harigai, M. et al. Excessive production of IFN-gamma in patients with systemic lupus erythematosus and its contribution to induction of B lymphocyte stimulator/B cell-activating factor/TNF ligand superfamily-13B. J. Immunol. 181, 2211–2219 (2008).
Yao, Y. et al. Neutralization of interferon-alpha/beta-inducible genes and downstream effect in a phase I trial of an anti-interferon-alpha monoclonal antibody in systemic lupus erythematosus. Arthritis Rheum. 60, 1785–1796 (2009).
Jacob, N. et al. B Cell and BAFF dependence of IFN-α-exaggerated disease in systemic lupus erythematosus-prone NZM 2328 mice. J. Immunol. 186, 4984–4993 (2011).
Blanco, P., Palucka, A. K., Gill, M., Pascual, V. & Banchereau, J. Induction of dendritic cell differentiation by IFN-alpha in systemic lupus erythematosus. Science 294, 1540–1543 (2001).
Joo, H. et al. Serum from patients with SLE instructs monocytes to promote IgG and IgA plasmablast differentiation. J. Exp. Med. 209, 1335–1348 (2012).
Kikly, K., Manetta, J., Smith, H. & Wierda, D. Characterization of LY2127399, A neutralizing antibody for BAFF [abstract]. Arthritis Rheum. 60 (Suppl. 10), 693 (2009).
Hsu, H. et al. A novel modality of BAFF-specific inhibitor AMG623 peptibody reduces B-cell number and improves outcomes in murine models of autoimmune disease. Clin. Exp. Rheumatol. 30, 197–201 (2012).
Wallweber, H. J., Compaan, D. M., Starovasnik, M. A. & Hymowitz, S. G. The crystal structure of a proliferation-inducing ligand, APRIL. J. Mol. Biol. 343, 283–290 (2004).
Puga, I. et al. B cell-helper neutrophils stimulate the diversification and production of immunoglobulin in the marginal zone of the spleen. Nat. Immunol. 13, 170–180 (2012).
Holden, N. J. et al. ANCA-stimulated neutrophils release BLyS and promote B cell survival: a clinically relevant cellular process. Ann. Rheum. Dis. 70, 2229–2233 (2011).
Assi, L. K. et al. Tumor necrosis factor-α activates release of B lymphocyte stimulator by neutrophils infiltrating the rheumatoid joint. Arthritis Rheum. 56, 1776–1786 (2007).
Suzuki, K. et al. Effect of interleukin-2 on synthesis of B cell activating factor belonging to the tumor necrosis factor family (BAFF) in human peripheral blood mononuclear cells. Cytokine 44, 44–48 (2008).
Zhang, W. et al. hsBAFF enhances activity of NK cells by regulation of CD4(+) T lymphocyte function. Immunol. Lett. 120, 96–102 (2008).
Author information
Authors and Affiliations
Contributions
All authors researched the data for the article. F.B.V. wrote the manuscript and all authors reviewed/edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
F.M. and E.F.M declare that they have acted as consultants for Eli Lilly and GSK. P.S. declares that he has a research agreement with Merck-Serono. F.B.V. declares no competing interests.
Rights and permissions
About this article
Cite this article
Vincent, F., Morand, E., Schneider, P. et al. The BAFF/APRIL system in SLE pathogenesis. Nat Rev Rheumatol 10, 365–373 (2014). https://doi.org/10.1038/nrrheum.2014.33
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2014.33
This article is cited by
-
B cell phenotype and serum levels of interferons, BAFF, and APRIL in multisystem inflammatory syndrome in children associated with COVID-19 (MIS-C)
Molecular and Cellular Pediatrics (2023)
-
Role of telitacicept in the treatment of IgA nephropathy
European Journal of Medical Research (2023)
-
Isoform-specific knockdown of long and intermediate prolactin receptors interferes with evolution of B-cell neoplasms
Communications Biology (2023)
-
Development and Characterization of a Novel Single-Chain Antibody Against B-Cell Activating Factor
Molecular Biotechnology (2023)
-
An early-onset SLE patient with a novel paternal inherited BACH2 mutation
Journal of Clinical Immunology (2023)