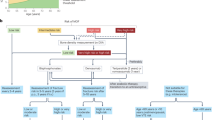
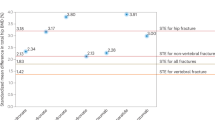
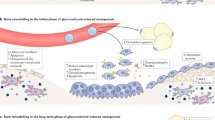
Abstract
Osteoporosis, a skeletal disorder characterized by compromised bone strength and an increased risk of fractures, is an important paediatric disorder that involves almost all paediatric subspecialties. Osteogenesis imperfecta is the most common form of childhood-onset primary osteoporosis, but several other forms are also known. Secondary osteoporosis is caused by an underlying chronic illness or its treatment. The most common causes of secondary osteoporosis include chronic systemic inflammation, glucocorticoid use and neuromuscular disabilities. The skeletal sequelae can present in childhood as low-energy peripheral and vertebral fractures, or become evident in adulthood as low bone mass and an increased propensity to develop osteoporosis. Management should aim at prevention, as interventions to treat symptomatic osteoporosis in the paediatric age group are scarce. Bisphosphonates are the principal pharmacological agents that can be used in this setting, but data on their efficacy and safety in paediatric populations remain inadequate, especially in patients with secondary osteoporosis. Consequently, it is important to understand the potential skeletal effects of paediatric illnesses and their therapies in order to institute effective and timely prevention of skeletal complications.
Key Points
-
Osteoporosis has become an important paediatric illness and can affect patients in any paediatric subspecialty
-
Osteogenesis imperfecta is the most common form of primary osteoporosis in the paediatric age group and includes several forms with variable severity
-
Secondary osteoporosis is common in children with illnesses that involve chronic systemic inflammation, neuromuscular disabilities, or glucocorticoid treatment
-
Osteoporosis presents as low bone mineral density and an increased risk of fractures in the vertebrae and limb bones
-
Preventive measures include optimal control of the underlying illness, vitamin D supplementation and maintenance of weight-bearing activity
-
Bisphosphonates are increasingly being used in children, but data on their efficacy and safety in paediatric patients are inadequate, especially with regard to treatment of secondary osteoporosis
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Karsenty, G. & Ferron, M. The contribution of bone to whole-organism physiology. Nature 481, 314–320 (2012).
Takayanagi, H. Osteoimmunology and the effects of the immune system on bone. Nat. Rev. Rheumatol. 5, 667–676 (2009).
NIH Consensus Development Panel on Osteoporosis Prevention, Diagnosis, and Therapy. Osteoporosis prevention, diagnosis, and therapy. JAMA 285, 785–795 (2001).
Baxter-Jones, A. D., Faulkner, R. A., Forwood, M. R., Mirwald, R. L. & Bailey, D. A. Bone mineral accrual from 8 to 30 years of age: an estimation of peak bone mass. J. Bone Miner. Res. 26, 1729–1739 (2011).
Cummings, S. R. et al. Bone density at various sites for prediction of hip fractures. The Study of Osteoporotic Fractures Research Group. Lancet 341, 72–75 (1993).
Bianchi, M. L. et al. Official positions of the International Society for Clinical Densitometry (ISCD) on DXA evaluation in children and adolescents. Pediatr. Nephrol. 25, 37–47 (2010).
Gafni R. I. & Baron, J. Overdiagnosis of osteoporosis in children due to misinterpretation of dual-energy x-ray absorptiometry (DEXA). J. Pediatr. 144, 253–257 (2004).
Rauch, F. et al. Fracture prediction and the definition of osteoporosis in children and adolescents: the ISCD 2007 Pediatric Official Positions. J. Clin. Densitom. 11, 22–28 (2008).
Mäyränpää, M. K., Viljakainen, H. T., Toiviainen-Salo, S., Kallio, P. E. & Mäkitie, O. Impaired bone health and asymptomatic vertebral compressions in fracture-prone children—a case–control study. J. Bone Miner. Res. 27, 1413–1424 (2012).
Landin, L. A. Epidemiology of children's fractures. J. Pediatr. Orthop. B. 6, 79–83 (1997).
Mäyränpää, M. K., Mäkitie, O. & Kallio, P. E. Decreasing incidence and changing pattern of childhood fractures: a population-based study. J. Bone Miner. Res. 25, 2752–2759 (2010).
Khosla, S. et al. Incidence of childhood distal forearm fractures over 30 years: a population-based study. JAMA 290, 1479–1485 (2003).
Mäyränpää, M. K., Tamminen, I. S., Kröger, H. & Mäkitie, O. Bone biopsy findings and correlation with clinical, radiological, and biochemical parameters in children with fractures. J. Bone Miner. Res. 26, 1748–1758 (2011).
Markula-Patjas, K. P. et al. Prevalence of vertebral compression fractures and associated factors in children and adolescents with severe juvenile idiopathic arthritis. J. Rheumatol. 39, 365–373 (2012).
McCloskey, E. & Kanis, J. A. FRAX updates 2012. Curr. Opin. Rheumatol. 24, 554–560 (2012).
Clark, E. M., Ness, A. R., Bishop, N. J. & Tobias, J. H. Association between bone mass and fractures in children: a prospective cohort study. J. Bone Miner. Res. 21, 1489–1495 (2006).
Manias, K., McCabe, D. & Bishop, N. Fractures and recurrent fractures in children: varying effects of environmental factors as well as bone size and mass. Bone 39, 652–657 (2006).
Buttazzoni, C. et al. Does a childhood fracture predict low bone mass in young adulthood?—A 27-year prospective controlled study. J. Bone Miner. Res. 28, 351–359 (2012).
Ralston, S. H. Genetics of osteoporosis. Ann. NY Acad. Sci. 1192, 181–189 (2010).
Pocock, N. A. et al. Genetic determinants of bone mass in adults. A twin study. J. Clin. Invest. 80, 706–710 (1987).
Lohmueller, K. E., Pearce, C. L., Pike, M., Lander, E. S. & Hirschhorn, J. N. Meta-analysis of genetic association studies supports a contribution of common variants to susceptibility to common disease. Nat. Genet. 33, 177–182 (2003).
Styrkarsdottir, U. et al. Multiple genetic loci for bone mineral density and fractures. N. Engl. J. Med. 358, 2355–2365 (2008).
Estrada, K. et al. Genome-wide meta-analysis identifies 56 bone mineral density loci and reveals 14 loci associated with risk of fracture. Nat. Genet. 44, 491–501 (2012).
Forlino, A., Cabral, W. A., Barnes, A. M. & Marini, J. C. New perspectives on osteogenesis imperfecta. Nat. Rev. Endocrinol. 7, 540–557 (2011).
Bianchi, M. L. & Glorieux, F. H. The spectrum of pediatric osteoporosis. In Pediatric Bone, 2nd edn (eds Glorieux, F. H. et al.) Ch 18 (Elsevier, Amsterdam, 2012).
Cundy, T. Recent advances in osteogenesis imperfecta. Calcif. Tissue Int. 90, 439–449 (2012).
Long, F. Building strong bones: molecular regulation of the osteoblast lineage. Nat. Rev. Mol. Cell. Biol. 13, 27–38 (2011).
Balemans, W. & van Hul, W. The genetics of low-density lipoprotein receptor-related protein 5 in bone: a story of extremes. Endocrinology 148, 2622–2629 (2007).
Gong, Y. et al. LDL receptor-related protein 5 (LRP5) affects bone accrual and eye development. Cell 107, 513–523 (2001).
Dent, C. E. & Friedman, M. Idiopathic juvenile osteoporosis. QJ Med. 34, 177–210 (1965).
Smith, R. Idiopathic juvenile osteoporosis: experience of twenty-one patients. Br. J. Rheumatol. 34, 68–77 (1995).
Rauch, F. et al. Deficient bone formation in idiopathic juvenile osteoporosis: a histomorphometric study of cancellous iliac bone. J. Bone Miner. Res. 15, 957–963 (2000).
Rauch, F. et al. The bone formation defect in idiopathic juvenile osteoporosis is surface-specific. Bone 31, 85–89 (2002).
Lorenc, R. S. Idiopathic juvenile osteoporosis. Calcif. Tissue. Int. 70, 395–397 (2002).
Płudowski, P. et al. Idiopathic juvenile osteoporosis—an analysis of the muscle-bone relationship. Osteoporos. Int. 17, 1681–1690 (2006).
Burnham, J. M. Inflammatory diseases and bone health in children. Curr. Opin. Rheumatol. 24, 548–553 (2012).
Faienza, M. F. et al. Osteoclastogenesis in children with 21-hydroxylase deficiency on long-term glucocorticoid therapy: the role of receptor activator of nuclear factor-κB ligand/osteoprotegerin imbalance. J. Clin. Endocrinol. Metab. 94, 2269–2276 (2009).
Kovacs, C. S. & Kronenberg, H. M. in Primer on the Metabolic Bone Diseases and Disorders of Mineral Metabolism (ed. Favus, M. J.) 63–68 (American Society for Bone and Mineral Research, Washington DC, 2006).
Weiler, H. A., Yuen, C. K. & Seshia, M. M. Growth and bone mineralization of young adults weighing less than 1500 g at birth. Early Hum. Dev. 67, 101–112 (2002).
Fewtrell, M. S. et al. Bone mineralization and turnover in preterm infants at 8–12 years of age: the effect of early diet. J. Bone Miner. Res. 14, 810–820 (1999).
Hovi, P. et al. Decreased bone mineral density in adults born with very low birth weight: a cohort study. PLoS Med. 6, e1000135 (2009).
Fehlings, D. et al. Informing evidence-based clinical practice guidelines for children with cerebral palsy at risk of osteoporosis: a systematic review. Dev. Med. Child. Neurol. 54, 106–116 (2012).
Stevenson, R. D. et al. Fracture rate in children with cerebral palsy. Pediatr. Rehabil. 9, 396–403 (2006).
Henderson, R. C. et al. The relationship between fractures and DXA measures of BMD in the distal femur of children and adolescents with cerebral palsy or muscular dystrophy. J. Bone Miner. Res. 25, 520–526 (2010).
Kilpinen-Loisa, P. et al. Low bone mass in patients with motor disability: prevalence and risk factors in 59 Finnish children. Dev. Med. Child. Neurol. 52, 276–282 (2010).
Boot, A. M. et al. Bone mineral density and body composition in adolescents with childhood—onset growth hormone deficiency. Hormone Res. 71, 364–371 (2009).
Högler, W. & Shaw, N. Childhood growth hormone deficiency, bone density, structures and fractures: scrutinizing the evidence. Clin. Endocrinol. (Oxf.) 72, 281–289 (2010).
Gahlot, M. et al. The effect of growth hormone deficiency on size-corrected bone mineral measures in pre-pubertal children. Osteoporos. Int. 23, 2211–2217 (2012).
Misra, M. Effects of hypogonadism on bone metabolism in female adolescents and young adults. Nat. Rev. Endocrinol. 8, 395–404 (2012).
Rochira, V. & Carani, C. Aromatase deficiency in men: a clinical perspective. Nat. Rev. Endocrinol. 5, 559–568 (2009).
Palmert, M. R. & Dunkel, L. Clinical practice. Delayed puberty. N. Engl. J. Med. 366, 443–453 (2012).
Pitukcheewanont, P. et al. Bone size and density measurements in prepubertal children with Turner syndrome prior to growth hormone therapy. Osteoporos. Int. 22, 1709–1715 (2011).
Hansen, S., Brixen, K. & Gravholt, C. H. Compromised trabecular microarchitecture and lower finite element estimates of radius and tibia bone strength in adults with turner syndrome: a cross-sectional study using high-resolution-pQCT. J. Bone Miner. Res. 27, 1794–803 (2012).
Gafni, R. I. et al. Daily parathyroid hormone 1–34 replacement therapy for hypoparathyroidism induces marked changes in bone turnover and structure. J. Bone Miner. Res. 27, 1811–1820 (2012).
Kraenzlin, M. E. & Meier, C. Parathyroid hormone analogues in the treatment of osteoporosis. Nat. Rev. Endocrinol. 7, 647–656 (2011).
Nicholls, J. J., Brassill, M. J., Williams, G. R. & Bassett, J. H. The skeletal consequences of thyrotoxicosis. J. Endocrinol. 213, 209–221 (2012).
Lodish, M. B. et al. Effects of Cushing disease on bone mineral density in a pediatric population. J. Pediatr. 156, 1001–1005 (2010).
Hamann, C., Kirschner, S., Günther, K. P. & Hofbauer, L. C. Bone, sweet bone—osteoporotic fractures in diabetes mellitus. Nat. Rev. Endocrinol. 8, 297–305 (2012).
Pekkinen, M., Viljakainen, H., Saarnio, E., Lamberg-Allardt, C. & Mäkitie, O. Vitamin D is a major determinant of bone mineral density at school age. PLoS ONE 7, e40090 (2012).
Kilpinen-Loisa, P. et al. Insufficient energy and nutrient intake in children with motor disability. Acta. Paediatr. 98, 1329–1333 (2009).
Mager, D. R., Qiao, J. & Turner, J. Vitamin D and K status influences bone mineral density and bone accrual in children and adolescents with celiac disease. Eur. J. Clin. Nutr. 66, 488–495 (2012).
Jesudason, D. & Clifton, P. The interaction between dietary protein and bone health. J. Bone Miner. Metab. 29, 1–14 (2011).
Jilka, R. L., Noble, B. & Weinstein, R. S. Osteocyte apoptosis. Bone http://dx.doi.org/10.1016/j.bone.2012.11.038.
Kawai, V. K., Stein, C. M., Perrien, D. S. & Griffin, M. R. Effects of anti-tumor necrosis factor α agents on bone. Curr. Opin. Rheumatol. 24, 576–585 (2012).
Bianchi, M. L. et al. Bone mass change during methotrexate treatment in patients with juvenile rheumatoid arthritis. Osteoporos. Int. 10, 20–25 (1999).
Mandel, K., Atkinson, S., Barr, R. D. & Pencharz, P. Skeletal morbidity in childhood acute lymphoblastic leukemia. J. Clin. Oncol. 22, 1215–1221 (2004).
Kalantar-Zadeh, K., Molnar, M. Z., Kovesdy, C. P., Mucsi, I. & Bunnapradist, S. Management of mineral and bone disorder after kidney transplantation. Curr. Opin. Nephrol. Hypertens. 21, 389–403 (2012).
Borusiak, P. et al. Antiepileptic drugs and bone metabolism in children: data from 128 patients. J. Child. Neurol. 28, 176–183 (2013).
Avgeri, M. et al. Assessment of bone mineral density and markers of bone turnover in children under long-term oral anticoagulant therapy. J. Pediatr. Hematol. Oncol. 30, 592–597 (2008).
Yanovski, J. A. et al. Treatment with a luteinizing hormone-releasing hormone agonist in adolescents with short stature. N. Engl. J. Med. 348, 908–917 (2003).
Weinstein, R. S. Clinical practice. Glucocorticoid-induced bone disease. N. Engl. J. Med. 365, 62–70 (2011).
Manzur, A. Y., Kuntzer, T., Pike, M. & Swan, A. Glucocorticoid corticosteroids for Duchenne muscular dystrophy. Cochrane Database of Systematic Reviews, Issue 1, Art. No.: CD003725. http://dx.doi.org/10.1002/14651858.CD003725.pub3.
King, W. M. et al. Orthopedic outcomes of long-term daily corticosteroid treatment in Duchenne muscular dystrophy. Neurology 68, 1607–1613 (2007).
Elmantaser, M. et al. Skeletal morbidity in children receiving chemotherapy for acute lymphoblastic leukaemia. Arch. Dis. Child. 95, 805–809 (2010).
Högler, W. et al. Incidence of skeletal complications during treatment of childhood acute lymphoblastic leukemia: Comparison of fracture risk with the General Practice Research Database. Pediatr. Blood Cancer 48, 21–27 (2007).
Halton, J. et al. Advanced vertebral fracture among newly diagnosed children with acute lymphoblastic leukemia: results of the Canadian Steroid-Associated Osteoporosis in the Pediatric Population (STOPP) research program. J. Bone Miner. Res. 24, 1326–1334 (2009).
Le Meignen, M. et al. Bone mineral density in adult survivors of childhood acute leukemia: impact of hematopoietic stem cell transplantation and other treatment modalities. Blood 118, 1481–1489 (2011).
Alos, N. et al. High incidence of vertebral fractures in children with acute lymphoblastic leukemia 12 months after the initiation of therapy. J. Clin. Oncol. 30, 2760–2767 (2012).
Rayar, M. S., Nayiager, T., Webber, C. E., Barr, R. D. & Athale, U. H. Predictors of bony morbidity in children with acute lymphoblastic leukemia. Pediatr. Blood Cancer 59, 77–82 (2012).
Mostoufi-Moab, S. et al. Bone density and structure in long-term survivors of pediatric allogeneic hematopoietic stem cell transplantation. J. Bone Miner. Res. 27, 760–769 (2012).
Acott, P. D., Crocker, J. F. & Wong, J. A. Decreased bone mineral density in the pediatric renal transplant population. Pediatr. Transplant. 7, 358–363 (2003).
Helenius, I. et al. Incidence and predictors of fractures in children after solid organ transplantation: a 5-year prospective, population-based study. J. Bone Miner. Res. 21, 380–387 (2006).
Valta, H., Jalanko, H., Holmberg, C., Helenius, I. & Mäkitie, O. Impaired bone health in adolescents after liver transplantation. Am. J. Transplant. 8, 150–157 (2008).
Valta, H., Mäkitie, O., Rönnholm, K. & Jalanko, H. Bone health in children and adolescents after renal transplantation. J. Bone Miner. Res. 24, 1699–1708 (2009).
Sachdeva, R. et al. Bone mineral status in pediatric heart transplant recipients: a retrospective observational study of an “at risk” cohort. Pediatr. Transplant. 14, 383–387 (2010).
Huber, A. M. et al. Prevalent vertebral fractures among children initiating glucocorticoid therapy for the treatment of rheumatic disorders. Arthritis Care Res. 62, 516–526 (2010).
Valta, H., Lahdenne, P., Jalanko, H., Aalto, K. & Mäkitie, O. Bone health and growth in glucocorticoid-treated patients with juvenile idiopathic arthritis. J. Rheumatol. 34, 831–836 (2007).
Nakhla, M. et al. Prevalence of vertebral fractures in children with chronic rheumatic diseases at risk for osteopenia. J. Pediatr. 154, 438–443 (2009).
Petty, R. E. et al. International League of Associations for Rheumatology classification of juvenile idiopathic arthritis: second revision, Edmonton, 2001. J. Rheumatol. 31, 390–392 (2004).
Pepmueller, P. H., Cassidy, J. T., Allen, S. H. & Hillman, L. S. Bone mineralization and bone mineral metabolism in children with juvenile rheumatoid arthritis. Arthritis Rheum. 39, 746–757 (1996).
Henderson, C. J. et al. Predictors of total body bone mineral density in noncorticosteroid-treated prepubertal children with juvenile idiopathic arthritis. Arthritis Rheum. 40, 1967–1975 (1997).
Henderson, C. J., Specker, B. L., Sierra, R. I., Campaigne, B. N. & Lovell, D. J. Total-body mineral content in noncorticosteroidtreated postpubertal females with juvenile rheumatoid arthritis. Arthritis Rheum. 43, 531–540 (2000).
Burnham, J. M. et al. Bone density, structure, and strength in juvenile idiopathic arthritis: importance of disease severity and muscle deficits. Arthritis Rheum. 58, 2518–2527 (2008).
Burnham, J. M., Shults, J., Weinstein, R., Lewis, J. D. & Leonard, M. B. Childhood onset arthritis is associated with an increased risk of fracture: a population based study using the General Practice Research Database. Ann. Rheum. Dis. 65, 1074–1079 (2006).
Thornton, J. et al. Bone health in adult men and women with a history of juvenile idiopathic arthritis. J. Rheumatol. 38, 1689–1693 (2011).
Voskaridou, E. & Terpos, E. New insights into the pathophysiology and management of osteoporosis in patients with β thalassaemia. Br. J. Haematol. 127, 127–139 (2004).
Yang, D. C. et al. Hypoxia inhibits osteogenesis in human mesenchymal stem cells through direct regulation of RUNX2 by TWIST. PLoS ONE 6, e23965 (2011).
Wesseling-Perry, K. Bone disease in pediatric chronic kidney disease. Pediatr. Nephrol. http://dx.doi.org/10.1007/s00467-012-2324-4.
Högler, W., Baumann, U. & Kelly, D. Endocrine and bone metabolic complications in chronic liver disease and after liver transplantation in children. J. Pediatr. Gastroenterol. Nutr. 54, 313–321 (2012).
Gunter, K. et al. Impact exercise increases BMC during growth: an 8-year longitudinal study. J. Bone Miner. Res. 23, 986–993 (2008).
Stark, C., Nikopoulou-Smyrni, P., Stabrey, A., Semler, O. & Schoenau, E. Effect of a new physiotherapy concept on bone mineral density, muscle force and gross motor function in children with bilateral cerebral palsy. J. Musculoskelet. Neuronal Interact. 10, 151–158 (2010).
Reyes, M. L., Hernández, M., Holmgren, L. J., Sanhueza, E. & Escobar, R. G. High-frequency, low-intensity vibrations increase bone mass and muscle strength in upper limbs, improving autonomy in disabled children. J. Bone Miner. Res. 26, 1759–1766 (2011).
Semler, O. et al. Results of a prospective pilot trial on mobility after whole body vibration in children and adolescents with osteogenesis imperfecta. Clin. Rehabil. 22, 387–394 (2008).
Misra, M., Pacaud, D., Petryk, A., Collett-Solberg, P. F. & Kappy, M. Drug and Therapeutics Committee of the Lawson Wilkins Pediatric Endocrine Society. Vitamin D deficiency in children and its management: review of current knowledge and recommendations. Pediatrics 122, 398–417 (2008).
Cashman, K. D. et al. Estimation of the dietary requirement for vitamin D in healthy adolescent white girls. Am. J. Clin. Nutr. 93, 549–555 (2011).
Pappa, H. M. et al. Treatment of vitamin D insufficiency in children and adolescents with inflammatory bowel disease: a randomized clinical trial comparing three regimens. J. Clin. Endocrinol. Metab. 97, 2134–2142 (2012).
Ma, N. S. & Gordon, C. M. Pediatric osteoporosis: where are we now? J. Pediatr. 161, 983–990 (2012).
Winzenberg, T. M., Shaw, K., Fryer, J. & Jones, G. Calcium supplementation for improving bone mineral density in children. Cochrane Database of Systematic Reviews, Issue 2, Art No.: CD005119. http://dx.doi.org/10.1002/14651858.CD005119.pub2.
Reid, I. R. & Bolland, M. J. Calcium supplements: bad for the heart? Heart 98, 895–896 (2012).
Phillipi, C. A., Remmington, T. & Steiner, R. D. Bisphosphonate therapy for osteogenesis imperfecta. Cochrane Database of Systematic Reviews, Issue 4, Art. No.: CD005088. http://dx.doi.org/10.1002/14651858.CD005088.pub2.
Castillo, H., Samson-Fang, L. & American Academy for Cerebral Palsy and Developmental Medicine treatment outcomes committee review panel. Effects of bisphosphonates in children with osteogenesis imperfecta: an AACPDM systematic review. Dev. Med. Child Neurol. 51, 17–29 (2009).
Bishop, N. et al. A randomized, controlled dose-ranging study of risedronate in children with moderate and severe osteogenesis imperfecta. J. Bone Miner. Res. 25, 32–40 (2010).
Streeten, E. A. et al. Osteoporosis–pseudoglioma syndrome: description of 9 new cases and beneficial response to bisphosphonates. Bone 43, 584–590 (2008).
Tüysüz, B. et al. Osteoporosis–pseudoglioma syndrome: three novel mutations in the LRP5 gene and response to bisphosphonate treatment. Horm. Res. Paediatr. 77, 115–120 (2012).
Ward, L. et al. Bisphosphonate therapy for children and adolescents with secondary osteoporosis. Cochrane Database of Systematic Reviews, Issue 4, Art. No.: CD005324. http://dx.doi.org/10.1002/14651858.CD005324.pub2.
Thornton, J. et al. Systematic review of effectiveness of bisphosphonates in treatment of low bone mineral density and fragility fractures in juvenile idiopathic arthritis. Arch. Dis. Child. 91, 753–761 (2006).
Cimaz, R. et al. Changes in markers of bone turnover and inflammatory variables during alendronate therapy in pediatric patients with rheumatic diseases. J. Rheumatol. 29, 1786–1792 (2002).
Bianchi, M. L. et al. Efficacy and safety of alendronate for the treatment of osteoporosis in diffuse connective tissue diseases in children: a prospective multicenter study. Arthritis Rheum. 43, 1960–1966 (2000).
Simm, P. J. et al. Zoledronic acid improves bone mineral density, reduces bone turnover and improves skeletal architecture over 2 years of treatment in children with secondary osteoporosis. Bone 49, 939–943 (2011).
Sbrocchi, A. M., Forget, S., Laforte, D., Azouz, E. M. & Rodd, C. Zoledronic acid for the treatment of osteopenia in pediatric Crohn's disease. Pediatr. Int. 52, 754–761 (2010).
Bachrach, L. K. & Ward, L. M. Clinical review 1: bisphosphonate use in childhood osteoporosis. J. Clin. Endocrinol. Metab. 94, 400–409 (2009).
Marini, J. C. Use of bisphosphonates in children—proceed with caution. Nat. Rev. Endocrinol. 5, 241–243 (2009).
Lekawasam, S. et al. A framework for the development of guidelines for the management of glucocorticoid-induced osteoporosis. Osteoporos. Int. 23, 2257–2276 (2012).
Gonzalez, L. & Witchel, S. F. The patient with Turner syndrome: puberty and medical management concerns. Fertil. Steril. 98, 780–6 (2012).
Groth, K. A., Skakkebæk, A., Høst, C., Gravholt, C. H. & Bojesen, A. Clinical review: Klinefelter syndrome-–a clinical update. J. Clin. Endocrinol. Metab. 98, 20–30 (2013).
Simon, D., Prieur, A. M., Quartier, P., Charles Ruiz, J. & Czernichow, P. Early recombinant human growth hormone treatment in glucocorticoid-treated children with juvenile idiopathic arthritis: a 3-year randomized study. J. Clin. Endocrinol. Metab. 92, 2567–2573 (2007).
Winer, K. K. et al. Synthetic human parathyroid hormone 1–34 replacement therapy: a randomized crossover trial comparing pump versus injections in the treatment of chronic hypoparathyroidism. J. Clin. Endocrinol. Metab. 97, 391–9 (2012).
Boyce, A. M. et al. Denosumab treatment for fibrous dysplasia. J. Bone Miner. Res. 27, 1462–70 (2012).
Pappa H. M. et al. Efficacy and harms of nasal calcitonin in improving bone density in young patients with inflammatory bowel disease: a randomized, placebo-controlled, double-blind trial. Am. J. Gastroenterol. 106, 1527–1543 (2011).
Acknowledgements
O. Mäkitie's research work is supported by grants from The Finnish Foundation for Paediatric Research, the Academy of Finland, the Sigrid Jusélius Foundation, Folkhälsan Research Foundation, Helsinki University Central Hospital Research Funds, and by the Sabbatical Leave Programme of the European Society for Paediatric Endocrinology, which is supported by an educational grant from Eli Lilly.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The author declares no competing financial interests.
Rights and permissions
About this article
Cite this article
Mäkitie, O. Causes, mechanisms and management of paediatric osteoporosis. Nat Rev Rheumatol 9, 465–475 (2013). https://doi.org/10.1038/nrrheum.2013.45
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2013.45