Key Points
-
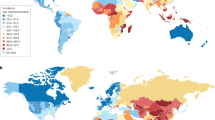
The incidence of intracerebral haemorrhage (ICH) increases steeply with age and has an annual incidence of ∼5 per 100,000 individuals in young adults (<50 years of age).
-
Classic risk factors for ICH are common even in young adults; drug abuse, pregnancy and the postpartum period are additional important young-adult-specific risk factors.
-
Hypertension is the predominant cause of ICH in young adult Indian and Asian populations, whereas hypertension and vascular abnormalities are equally important in white populations.
-
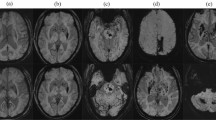
Potential causes of ICH in young people are many and require systematic diagnostic studies with imaging of the intracranial vasculature as the cornerstone of the diagnostic work-up.
-
Observational evidence supports an aggressive approach to the management of ICH in young patients, including lowering of elevated blood pressure, surgical haematoma evacuation (in selected patients) and vigorous rehabilitation.
-
ICH in young adults has a dismal prognosis: 3-month mortality is 17%, 10-year mortality is >25% and long-term disability is common.
Abstract
Nontraumatic intracerebral haemorrhage (ICH) is a common subtype of stroke with a poor prognosis, high mortality and long-term morbidity. The incidence of ICH increases with age. ICH has not been widely investigated in young adults (herein defined as aged ∼18–50 years) despite an annual incidence of ∼5 per 100,000 individuals. Furthermore, ICH characteristics differ between young and elderly patients. Risk factors for ICH are surprisingly common in young adults, in whom ICH is often caused by structural lesions or hypertension, and only rarely by anticoagulation therapy and cerebral amyloid angiopathy (which are common predisposing factors in elderly patients). High short-term mortality (17% at 3 months) and long-term mortality (>25% at 10 years) persist even in contemporary series from high-income countries, and long-term disability is very common. Thus, an aggressive approach to identifying treatable underlying conditions and preventing ICH recurrence is indicated in young patients, although treatment strategies have generally not been investigated specifically in this age group. This narrative Review summarizes existing knowledge on the epidemiology, risk factors, causes, diagnosis, treatment and outcomes of ICH in young adults. We provide comparisons with the population of elderly patients with ICH and discuss challenges for future research.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Qureshi, A. I. et al. Spontaneous intracerebral hemorrhage. N. Engl. J. Med. 344, 1450–1460 (2001).
Qureshi, A. I., Mendelow, A. D. & Hanley, D. F. Intracerebral haemorrhage. Lancet 373, 1632–1644 (2009).
Keep, R. F., Hua, Y. & Xi, G. Intracerebral haemorrhage: mechanisms of injury and therapeutic targets. Lancet Neurol. 11, 720–731 (2012).
Solomon, R. A. & Connolly, E. S. Jr. Arteriovenous malformations of the brain. N. Engl. J. Med. 377, 498 (2017).
Silvis, S. M., de Sousa, D. A., Ferro, J. M. & Coutinho, J. M. Cerebral venous thrombosis. Nat. Rev. Neurol. 13, 555–565 (2017).
Kuroda, S. & Houkin, K. Moyamoya disease: current concepts and future perspectives. Lancet Neurol. 7, 1056–1066 (2008).
Dutra, L. A., de Souza, A. W., Grinberg-Dias, G., Barsottini, O. G. & Appenzeller, S. Central nervous system vasculitis in adults: an update. Autoimmun. Rev. 16, 123–131 (2017).
Kalita, J., Goyal, G., Kumar, P. & Misra, U. K. Intracerebral hemorrhage in young patients from a tertiary neurology center in North India. J. Neurol. Sci. 336, 42–47 (2014).
Koivunen, R. et al. Incidence, risk factors, etiology, severity and short-term outcome of non-traumatic intracerebral hemorrhage in young adults. Eur. J. Neurol. 22, 123–132 (2015).
Lai, S., Chen, S., Lee, T., Ro, L. & Hsu, S. Spontaneous intracerebral hemorrhage in young adults. Eur. J. Neurol. 12, 310–316 (2005).
Ruiz-Sandoval, J. L., Cantu, C. & Barinagarrementeria, F. Intracerebral hemorrhage in young people: analysis of risk factors, location, causes, and prognosis. Stroke 30, 537–541 (1999).
Fuh, J. L., Liu, H. C., Wang, S. J., Lo, Y. K. & Lee, L. S. Nontraumatic hemorrhagic stroke in young adults in Taiwan. J. Stroke Cerebrovasc Dis. 4, 101–105 (1994).
Del Brutto, O. H., Sanchez, J., Campos, X., Santos, J. & Mosquera, A. Non-traumatic intracerebral hemorrhage in young adults living in Guayaquil, Ecuador (South America): analysis of 151 patients. Funct. Neurol. 14, 21–28 (1999).
Kirkman, M. A., Tyrrell, P. J., King, A. T. & Patel, H. C. Imaging in young adults with intracerebral hemorrhage. Clin. Neurol. Neurosurg. 114, 1297–1303 (2012).
Awada, A., Daif, A., Obeid, T. & Al Rajeh, S. Nontraumatic cerebral hemorrhage in the young: a study of 107 cases. J. Stroke Cerebrovasc Dis. 7, 200–204 (1998).
Rutten-Jacobs, L. C. et al. Clinical characteristics and outcome of intracerebral hemorrhage in young adults. J. Neurol. 261, 2143–2149 (2014).
Lin, C. L. & Howng, S. L. Nontraumatic intracerebral hemorrhage in young adult. Kaohsiung J. Med. Sci. 13, 237–242 (1997).
Toffol, G. J., Biller, J. & Adams, H. P. J. Nontraumatic intracerebral hemorrhage in young adults. Arch. Neurol. 44, 483–485 (1987).
Bevan, H., Sharma, K. & Bradley, W. Stroke in young adults. Stroke 21, 382–386 (1990).
Go, G. O. et al. The outcomes of spontaneous intracerebral hemorrhage in young adults — a clinical study. J. Cerebrovasc. Endovasc. Neurosurg. 15, 214–220 (2013).
Feigin, V. L., Lawes, C. M., Bennett, D. A., Barker-Collo, S. L. & Parag, V. Worldwide stroke incidence and early case fatality reported in 56 population-based studies: a systematic review. Lancet Neurol. 8, 355–369 (2009).
Marini, C., Russo, T. & Felzani, G. Incidence of stroke in young adults: a review. Stroke Res. Treat. 2011, 535672 (2010).
Marini, C. et al. Stroke in young adults in the community-based L'Aquila registry: incidence and prognosis. Stroke 32, 52–56 (2001).
Lin, C. H. et al. Cerebrovascular diseases in a fixed population of Hiroshima and Nagasaki, with special reference to relationship between type and risk factors. Stroke 15, 653–660 (1984).
van Asch, C. J. et al. Incidence, case fatality, and functional outcome of intracerebral haemorrhage over time, according to age, sex, and ethnic origin: a systematic review and meta-analysis. Lancet Neurol. 9, 167–176 (2010).
Nencini, P. et al. Incidence of stroke in young adults in Florence. Italy. Stroke 19, 977–981 (1988).
Krishnamurthi, R. V. et al. The global burden of hemorrhagic stroke: a summary of findings from the GBD 2010 study. Glob. Heart 9, 101–106 (2014).
Krishnamurthi, R. V. et al. Stroke prevalence, mortality and disability-adjusted life years in adults aged 20–64 years in 1990–2013: data from the Global Burden of Disease 2013 study. Neuroepidemiology 45, 190–202 (2015).
Jacobs, B. S., Boden-Albala, B., Lin, I. F. & Sacco, R. L. Stroke in the young in the northern Manhattan stroke study. Stroke 33, 2789–2793 (2002).
Kittner, S. J. et al. Black–white differences in stroke risk among young adults. Stroke 24 (Suppl.), I-13–I-15 (1993).
O'Donnell, M. J. et al. Risk factors for ischaemic and intracerebral haemorrhagic stroke in 22 countries (the INTERSTROKE study): a case–control study. Lancet 376, 112–123 (2010).
Monforte, R., Estruch, R., Graus, F., Nicolas, J. M. & Urbano-Marquez, A. High ethanol consumption as risk factor for intracerebral hemorrhage in young and middle-aged people. Stroke 21, 1529–1532 (1990).
Feldmann, E. et al. Major risk factors for intracerebral hemorrhage in the young are modifiable. Stroke 36, 1881–1885 (2005).
Qureshi, A. I. et al. Cocaine use and hypertension are major risk factors for intracerebral hemorrhage in young African Americans. Ethn. Dis. 11, 311–319 (2001).
Kittner, S. J. et al. Pregnancy and the risk of stroke. N. Engl. J. Med. 335, 768–774 (1996).
Sharshar, T., Lamy, C. & Mas, J. L. Incidence and causes of strokes associated with pregnancy and puerperium. A Study Publ. Hospitals Ile France. Stroke Pregnancy Study Group. Stroke 26, 930–936 (1995).
Bateman, B. T. et al. Intracerebral hemorrhage in pregnancy: frequency, risk factors, and outcome. Neurology 67, 424–429 (2006).
Leffert, L. R. et al. Patient characteristics and outcomes after hemorrhagic stroke in pregnancy. Circ. Cardiovasc. Qual. Outcomes 8, S170–S178 (2015).
Yoshida, K. et al. Strokes associated with pregnancy and puerperium: a nationwide study by the Japan Stroke Society. Stroke 48, 276–282 (2017).
Carpenter, A. M., Singh, I. P., Gandhi, C. D. & Prestigiacomo, C. J. Genetic risk factors for spontaneous intracerebral haemorrhage. Nat. Rev. Neurol. 12, 40–49 (2016).
Labauge, P., Denier, C., Bergametti, F. & Tournier-Lasserve, E. Genetics of cavernous angiomas. Lancet Neurol. 6, 237–244 (2007).
Bersano, A. et al. Research progresses in understanding the pathophysiology of moyamoya disease. Cerebrovasc. Dis. 41, 105–118 (2016).
Cotlarciuc, I. et al. Towards the genetic basis of cerebral venous thrombosis — the BEAST Consortium: a study protocol. BMJ Open 6, e012351 (2016).
Miyatake, S. et al. Homozygous c.14576G>A variant of RNF213 predicts early-onset and severe form of moyamoya disease. Neurology 78, 803–810 (2012).
Revencu, N. et al. RASA1 mutations and associated phenotypes in 68 families with capillary malformation–arteriovenous malformation. Hum. Mutat. 34, 1632–1641 (2013).
Amyere, M. et al. Germline loss-of-function mutations in EPHB4 cause a second form of capillary malformation–arteriovenous malformation (CM-AVM2) deregulating RAS–MAPK signaling. Circulation 136, 1037–1048 (2017).
Abdalla, S. A. & Letarte, M. Hereditary haemorrhagic telangiectasia: current views on genetics and mechanisms of disease. J. Med. Genet. 43, 97–110 (2006).
Chong, M. et al. Mendelian genes and risk of intracerebral hemorrhage and small-vessel ischemic stroke in sporadic cases. Stroke 48, 2263–2265 (2017).
Rannikmae, K. et al. COL4A2 is associated with lacunar ischemic stroke and deep ICH: meta-analyses among 21,500 cases and 40,600 controls. Neurology 89, 1829–1839 (2017).
Vahedi, K. et al. COL4A1 mutation in a patient with sporadic, recurrent intracerebral hemorrhage. Stroke 38, 1461–1464 (2007).
Meretoja, A. et al. SMASH-U: a proposal for etiologic classification of intracerebral hemorrhage. Stroke 43, 2592–2597 (2012).
Wu, T. Y. et al. Simultaneous multiple intracerebral hemorrhages (SMICH). Stroke 48, 581–586 (2017).
Ducros, A. Reversible cerebral vasoconstriction syndrome. Lancet Neurol. 11, 906–917 (2012).
McEvoy, A. W., Kitchen, N. D. & Thomas, D. G. Lesson of the week. Intracerebral haemorrhage in young adults: the emerging importance of drug misuse. BMJ 320, 1322–1324 (2000).
Delgado Almandoz, J. E. et al. Practical scoring system for the identification of patients with intracerebral hemorrhage at highest risk of harboring an underlying vascular etiology: the Secondary Intracerebral Hemorrhage Score. AJNR Am. J. Neuroradiol. 31, 1653–1660 (2010).
Olavarria, V. V., Bustamante, G., Lopez, M. J. & Lavados, P. M. Diagnostic accuracy of a simple clinical score to screen for vascular abnormalities in patients with intracerebral hemorrhage. J. Stroke Cerebrovasc. Dis. 23, 2069–2074 (2014).
Kidwell, C. S. et al. Comparison of MRI and CT for detection of acute intracerebral hemorrhage. JAMA 292, 1823–1830 (2004).
Delgado Almandoz, J. E. et al. CT angiography spot sign predicts in-hospital mortality in patients with secondary intracerebral hemorrhage. J. Neurointerv. Surg. 4, 442–447 (2012).
Josephson, C. B., White, P. M., Krishan, A. & Al-Shahi Salman, R. Computed tomography angiography or magnetic resonance angiography for detection of intracranial vascular malformations in patients with intracerebral haemorrhage. Cochrane Database Syst. Rev. 9, 009372 (2014).
Saposnik, G. et al. Diagnosis and management of cerebral venous thrombosis: a statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 42, 1158–1192 (2011).
Ducros, A. et al. The clinical and radiological spectrum of reversible cerebral vasoconstriction syndrome. A prospective series of 67 patients. Brain 130, 3091–3101 (2007).
Yeung, R. et al. Comparison of CTA to DSA in determining the etiology of spontaneous ICH. Can. J. Neurol. Sci. 36, 176–180 (2009).
Hemphill, J. C. 3rd et al. Guidelines for the management of spontaneous intracerebral hemorrhage: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 46, 2032–2060 (2015).
Anderson, C. S. et al. Rapid blood-pressure lowering in patients with acute intracerebral hemorrhage. N. Engl. J. Med. 368, 2355–2365 (2013).
Qureshi, A. I. et al. Intensive blood-pressure lowering in patients with acute cerebral hemorrhage. N. Engl. J. Med. 375, 1033–1043 (2016).
Stroke Unit Trialists' Collaboration. Organised inpatient (stroke unit) care for stroke. Cochrane Database Syst. Rev. 4, CD000197 (2007).
Steiner, T. et al. European Stroke Organisation (ESO) guidelines for the management of spontaneous intracerebral hemorrhage. Int. J. Stroke 9, 840–855 (2014).
Mendelow, A. D. et al. Early surgery versus initial conservative treatment in patients with spontaneous supratentorial intracerebral haematomas in the international Surgical Trial in Intracerebral Haemorrhage (STICH): a randomised trial. Lancet 365, 387–397 (2005).
Mendelow, A. D. et al. Early surgery versus initial conservative treatment in patients with spontaneous supratentorial lobar intracerebral haematomas (STICH II): a randomised trial. Lancet 382, 397–408 (2013).
Koivunen, R. et al. Predictors of early mortality in young adults after intracerebral hemorrhage. Stroke 45, 2454–2456 (2014).
Fung, C. et al. Decompressive hemicraniectomy in patients with supratentorial intracerebral hemorrhage. Stroke 43, 3207–3211 (2012).
SPS3 Study Group et al. Blood-pressure targets in patients with recent lacunar stroke: the SPS3 randomised trial. Lancet 382, 507–515 (2013).
Law, M. R., Morris, J. K. & Wald, N. J. Use of blood pressure lowering drugs in the prevention of cardiovascular disease: meta-analysis of 147 randomised trials in the context of expectations from prospective epidemiological studies. BMJ 338, b1665 (2009).
Mast, H. et al. Risk of spontaneous haemorrhage after diagnosis of cerebral arteriovenous malformation. Lancet 350, 1065–1068 (1997).
Laakso, A. et al. Risk of hemorrhage in patients with untreated Spetzler-Martin grade IV and V arteriovenous malformations: a long-term follow-up study in 63 patients. Neurosurgery 68, 372–377 (2011).
Ogilvy, C. S. et al. AHA Scientific Statement. Recommendations for the management of intracranial arteriovenous malformations: a statement for healthcare professionals from a special writing group of the Stroke Council, AmericanStroke Association. Stroke 32, 1458–1471 (2001).
Kobayashi, E., Saeki, N., Oishi, H., Hirai, S. & Yamaura, A. Long-term natural history of hemorrhagic moyamoya disease in 42 patients. J. Neurosurg. 93, 976–980 (2000).
Miyamoto, S. et al. Effects of extracranial-intracranial bypass for patients with hemorrhagic moyamoya disease: results of the Japan Adult Moyamoya Trial. Stroke 45, 1415–1421 (2014).
Takahashi, J. C. et al. Significance of the hemorrhagic site for recurrent bleeding: prespecified analysis in the Japan Adult Moyamoya Trial. Stroke 47, 37–43 (2016).
Horne, M. A. et al. Clinical course of untreated cerebral cavernous malformations: a meta-analysis of individual patient data. Lancet Neurol. 15, 166–173 (2016).
Mathiesen, T., Edner, G. & Kihlstrom, L. Deep and brainstem cavernomas: a consecutive 8-year series. J. Neurosurg. 99, 31–37 (2003).
Li, C. et al. Clinical and angioarchitectural risk factors associated with intracranial hemorrhage in dural arteriovenous fistulas: a single-center retrospective study. PLOS ONE 10, e0131235 (2015).
Koivunen, R. et al. Medical acute complications of intracerebral hemorrhage in young adults. Stroke Res. Treat. 2015, 357696 (2015).
Chong, J. Y. & Sacco, R. L. Epidemiology of stroke in young adults: race/ethnic differences. J. Thromb. Thrombolysis 20, 77–83 (2005).
Koivunen, R., Tatlisumak, T., Satopaa, J., Niemela, M. & Putaala, J. Intracerebral hemorrhage at young age: long-term prognosis. Eur. J. Neurol. 22, 1029–1037 (2015).
Leno, C. et al. A prospective study of stroke in young adults in Cantabria. Spain. Stroke 24, 792–795 (1993).
Ruiz-Sandoval, J. L. et al. Hypertensive intracerebral hemorrhage in young people: previously unnoticed age-related clinical differences. Stroke 37, 2946–2950 (2006).
Arntz, R. et al. Post-stroke epilepsy in young adults: a long-term follow-up study. PLOS ONE 8, e55498 (2013).
Radholm, K. et al. Older age is a strong predictor for poor outcome in intracerebral haemorrhage: the INTERACT2 study. Age Ageing 44, 422–427 (2015).
Umeano, O. et al. Gender and age interact to affect early outcome after intracerebral hemorrhage. PLOS ONE 8, e81664 (2013).
Synhaeve, N. E. et al. Poor long-term functional outcome after stroke among adults aged 18 to 50 years: follow-up of transient ischemic attack and stroke patients and unelucidated risk factor evaluation (FUTURE) study. Stroke 45, 1157–1160 (2014).
Ellis, C. Stroke in young adults. Disabil. Health. J. 3, 222–224 (2010).
Koivunen, R., Harno, H., Tatlisumak, T. & Putaala, J. Depression, anxiety, and cognitive functioning after intracerebral hemorrhage. Acta Neurol. Scand. 132, 179–184 (2015).
Maaijwee, N. A. et al. Long-term increased risk of unemployment after young stroke: a long-term follow-up study. Neurology 83, 1132–1138 (2014).
Hannerz, H., Holbaek Pedersen, B., Poulsen, O. M., Humle, F. & Andersen, L. L. A nationwide prospective cohort study on return to gainful occupation after stroke in Denmark 1996–2006. BMJ Open 1, e000180–002011-000180 (2011).
Acknowledgements
The authors thank A. Eräkanto for secretarial help.
Author information
Authors and Affiliations
Contributions
J.P. and T.T. conducted the literature search for the article. All authors contributed to the original idea and planning for the article, to drafting the initial version and to critical revision or editing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
T.T. declares that he has received grants for research into intracerebral haemorrhage from Helsinki University Central Hospital, Sahlgrenska University Hospital, University of Gothenburg, and the Sigrid Juselius Foundation. The other authors declare no competing interests.
Related links
RELATED LINKS
Glossary
- Surgical equipoise
-
A principle according to which patients who are eligible for enrolment in randomized clinical trials of medical versus surgical treatments are excluded from randomization if the clinician decides that (despite a lack of scientific evidence) surgical treatment will be superior, or life-saving. Such considerations often result in the exclusion of young patients from clinical trial cohorts.
Rights and permissions
About this article
Cite this article
Tatlisumak, T., Cucchiara, B., Kuroda, S. et al. Nontraumatic intracerebral haemorrhage in young adults. Nat Rev Neurol 14, 237–250 (2018). https://doi.org/10.1038/nrneurol.2018.17
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneurol.2018.17
This article is cited by
-
A 6-month prognostic nomogram incorporating hemoglobin level for intracerebral hemorrhage in younger adults
BMC Neurology (2023)
-
Digital subtraction angiography findings of stroke in young adult population: a multi-center record-based study
The Egyptian Journal of Neurology, Psychiatry and Neurosurgery (2023)
-
Intracerebral haemorrhage
Nature Reviews Disease Primers (2023)
-
Stroke in young adults, stroke types and risk factors: a case control study
BMC Neurology (2022)
-
A Contemporary Review of Epidemiology, Risk Factors, Etiology, and Outcomes of Premature Stroke
Current Atherosclerosis Reports (2022)