Abstract
In 1817, James Parkinson described the symptoms of the shaking palsy, a disease that was subsequently defined in greater detail, and named after Parkinson, by Jean-Martin Charcot. Parkinson expected that the publication of his monograph would lead to a rapid elucidation of the anatomical substrate of the shaking palsy; in the event, this process took almost a century. In 1912, Fritz Heinrich Lewy identified the protein aggregates that define Parkinson disease (PD) in some brain regions outside the substantia nigra. In 1919, Konstantin Nikolaevich Tretiakoff found similar aggregates in the substantia nigra and named them after Lewy. In the 1990s, α-synuclein was identified as the main constituent of the Lewy pathology, and its aggregation was shown to be central to PD, dementia with Lewy bodies, and multiple system atrophy. In 2003, a staging scheme for idiopathic PD was introduced, according to which α-synuclein pathology originates in the dorsal motor nucleus of the vagal nerve and progresses from there to other brain regions, including the substantia nigra. In this article, we review the relevance of Lewy's discovery 100 years ago for the current understanding of PD and related disorders.
Key Points
-
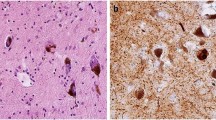
100 years ago, Fritz Heinrich Lewy used light microscopy to describe the nerve cell inclusions that are characteristic of Parkinson disease (PD)
-
The Lewy pathology consists of the protein α-synuclein in an insoluble form
-
Missense and gene dosage mutations in SNCA, the α-synuclein gene, cause inherited cases of PD and dementia with Lewy bodies
-
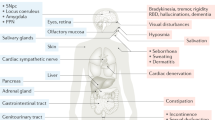
In PD, α-synuclein pathology is widespread in the CNS and PNS
-
α-Synuclein pathology originates in a small number of nerve cells, from which it spreads in a prion-like fashion
-
Clinically, the development of the pathological changes of PD is reflected by the presence of nonmotor and motor symptoms
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Lewy, F. Paralysis agitans. I. Pathologische Anatomie. In Handbuch der Neurologie Vol. 3 (eds Lewandowsky, M. & Abelsdorff, G.) 920–933 (Springer-Verlag, Berlin, 1912).
Tretiakoff, C. Contribution à l'étude de l'anatomie pathologique du locus niger de Soemmering avec quelques déductions relatives à la pathogénie des troubles du tonus musculaire et de la maladie de Parkinson. Thesis, University of Paris (1919).
Braak, H. et al. Amygdala pathology in Parkinson's disease. Acta Neuropathol. 88, 493–500 (1994).
Polymeropoulos, M. H. et al. Mutation in the α-synuclein gene identified in families with Parkinson's disease. Science 276, 2045–2047 (1997).
Spillantini, M. G. et al. α-Synuclein in Lewy bodies. Nature 388, 839–840 (1997).
Satake, W. et al. Genome-wide association study identifies common variants at four loci as genetic risk factors for Parkinson's disease. Nat. Genet. 41, 1303–1307 (2009).
Simón-Sánchez, J. et al. Genome-wide association study reveals genetic risk underlying Parkinson's disease. Nat. Genet. 41, 1308–1311 (2009).
Braak, H. et al. Staging of brain pathology related to sporadic Parkinson's disease. Neurobiol. Aging 24, 197–211 (2003).
Holdorff, B. Friedrich Heinrich Lewy (1885–1950) and his work. J. Hist. Neurosci. 11, 19–28 (2002).
Rodrigues e Silva, A. M. et al. Who was the man who discovered the “Lewy bodies”? Mov. Disord. 25, 1765–1773 (2010).
Sweeney, P. J., Lloyd, M. F. & Daroff, R. B. What's in a name? Dr. Lewey and the Lewy body. Neurology 49, 629–630 (1997).
Lewy, F. H. Zur pathologischen Anatomie der Paralysis agitans [German]. Dtsch. Z. f. Nervenheilk. 50, 50–55 (1913).
Lafora, G. R. & Glueck, B. Beitrag zur Histopathologie der myoklonischen Epilepsie [German]. Z. ges. Neurol. Psychiat. 6, 1–14 (1911).
Blocq, P. & Marinesco, G. Sur un cas de tremblement parkinsonien hémiplégique symptomatique d'une tumeur du pédoncule cérébral [French]. C. R. Soc. Biol. 5, 105–111 (1893).
Lewy, F. H. Die Lehre vom Tonus und der Bewegung (Springer-Verlag, Berlin, 1923).
Hassler, R. Zur Pathologie der Paralysis agitans und des postenzephalitischen Parkinsonismus [German]. J. Psychol. Neurol. 48, 387–455 (1938).
Lewy, F. H. Die Entstehung der Einschlusskörper und ihre Bedeutung für die systematische Einordnung der sogenannten Viruskrankheiten [German]. Dtsch. Z. f. Nervenheilk. 124, 93–100 (1932).
Goedert M., Clavaguera, F. & Tolnay, M. The propagation of prion-like protein inclusions in neurodegenerative diseases. Trends Neurosci. 33, 317–325 (2010).
Lewy, F. H. Historical introduction: the diseases of the basal ganglia. Res. Publ. Ass. Nerv. Ment. Dis. 21, 1–20 (1942).
de Lau, L. M. & Breteler, M. M. Epidemiology of Parkinson's disease. Lancet Neurol. 5, 525–535 (2006).
Reid, W. G., Hely, M. A., Morris, J. G., Loy, C. & Halliday, G. M. Dementia in Parkinson's disease: a 20-year neuropsychological study (Sydney Multicentre Study). J. Neurol. Neurosurg. Psychiatry 82, 1033–1037 (2011).
Braak, H. & Del Tredici, K. Neuroanatomy and pathology of sporadic Parkinson's disease. Adv. Anat. Embryol. Cell Biol. 201, 1–119 (2009).
Krüger, R. et al. Ala30Pro mutation in the gene encoding α-synuclein in Parkinson's disease. Nat. Genet. 18, 106–108 (1998).
Zarranz, J. J. et al. The new mutation, E46K, of α-synuclein causes Parkinson and Lewy body dementia. Ann. Neurol. 55, 164–173 (2004).
Goedert, M. Alpha-synuclein and neurodegenerative diseases. Nat. Rev. Neurosci. 2, 492–501 (2001).
Singleton, A. B. et al. α-Synuclein locus triplication causes Parkinson's disease. Science 302, 841 (2003).
Chartier-Harlin, M. C. et al. α-Synuclein locus duplication in a case of familial Parkinson's disease. Lancet 364, 1167–1169 (2004).
Ibanez, P. et al. Causal relation between α-synuclein gene duplication and familial Parkinson's disease. Lancet 364, 1169–1171 (2004).
Krüger, R. et al. Increased susceptibility to sporadic Parkinson's disease by a certain combined α-synuclein/apolipoprotein E genotype. Ann. Neurol. 45, 611–617 (1999).
Poulopoulos, M., Levy, O. A. & Alcalay, R. N. The neuropathology of genetic Parkinson's disease. Mov. Disord. 27, 831–842 (2012).
Kanazawa, T. et al. Pale neurites, premature α-synuclein aggregates with centripetal extension from axon collaterals. Brain Pathol. 22, 67–78 (2012).
Cremades, N. et al. Direct observation of the interconversion of normal and toxic forms of α-synuclein. Cell 149, 1048–1059 (2012).
Dickson, D. W. et al. Evidence that incidental Lewy body disease is pre-symptomatic Parkinson's disease. Acta Neuropathol. 115, 437–444 (2008).
Josephs, K. A., Parisi, J. E. & Dickson, D. W. Alpha-synuclein studies are negative in post-encephalitic parkinsonism of von Economo. Neurology 59, 645–646 (2002).
Spillantini, M. G., Crowther, R. A., Jakes, R., Hasegawa, M. & Goedert, M. α-Synuclein in filamentous inclusions of Lewy bodies from Parkinson's disease and dementia with Lewy bodies. Proc. Natl Acad. Sci. USA 95, 6469–6473 (1998).
Serpell, L. C., Berriman, J., Jakes, R., Goedert, M. & Crowther, R. A. Fibre diffraction of synthetic α-synuclein filaments shows amyloid-like cross-β conformation. Proc. Natl Acad. Sci. USA 97, 4897–4902 (2000).
Vilar, M. et al. The fold of α-synuclein fibrils. Proc. Natl Acad. Sci. USA 105, 8637–8642 (2008).
Fujiwara, H. et al. α-Synuclein is phosphorylated in synucleinopathy lesions. Nat. Cell Biol. 4, 160–164 (2002).
Anderson, J. P. et al. Phosphorylation of Ser-129 is the dominant modification of α-synuclein in familial and sporadic Lewy body disease. J. Biol. Chem. 281, 29739–29752 (2006).
Kuusisto, E., Parkkinen, L. & Alazuloff, I. Morphogenesis of Lewy bodies: dissimilar incorporation of α-synuclein, ubiquitin, and p62. J. Neuropathol. Exp. Neurol. 62, 1241–1253 (2003).
McKeith, I. et al. Diagnosis and management of dementia with Lewy bodies: third report of the DLB Consortium. Neurology 65, 1863–1872 (2005).
Kosaka, K. & Manabe, Y. The first autopsied case of diffuse Lewy body disease (DLBD); re-examination by recent immunostaining methods. Neuropathology 30, 458–462 (2010).
Kotzbauer, P. T., Trojanowski, J. Q. & Lee, V. M. Lewy body pathology in Alzheimer's disease. J. Mol. Neurosci. 17, 225–232 (2001).
Lee, H. G., Zhu, X., Takeda, A., Perry, G. & Smith, M. A. Emerging evidence for the neuroprotective role of α-synuclein. Exp. Neurol. 200, 1–7 (2006).
Saha, A. R. et al. Parkinson's disease α-synuclein mutations exhibit defective axonal transport in cultured neurons. J. Cell Sci. 117, 1017–1024 (2004).
Beach, T. G. et al. Reduced striatal tyrosine hydroxylase in incidental Lewy body disease. Acta Neuropathol. 115, 445–451 (2008).
Dugger, B. N. & Dickson, D. W. Cell type-specific sequestration of choline acetyltransferase and tyrosine hydroxylase within Lewy bodies. Acta Neuropathol. 120, 633–639 (2010).
Del Tredici, K. & Braak, H. Spinal cord lesions in sporadic Parkinson's disease. Acta Neuropathol. 124, 643–664 (2012).
Braak, H., Sastre, M., Bohl, J. R., de Vos, R. A. & Del Tredici, K. Parkinson's disease: lesions in dorsal horn layer I, involvement of parasympathetic and sympathetic pre- and postganglionic neurons. Acta Neuropathol. 113, 421–429 (2007).
Wakabayashi, K., Takahashi, H., Ohama, E. & Ikuta, F. Parkinson's disease: an immunohistochemical study of Lewy body-containing neurons in the enteric nervous system. Acta Neuropathol. 79, 581–583 (1990).
Pouclet, H. et al. A comparison between rectal and colonic biopsies to detect Lewy pathology in Parkinson's disease. Neurobiol. Dis. 45, 305–309 (2012).
Wakabayashi, K. & Takahashi, H. Neuropathology of autonomic nervous system in Parkinson's disease. Eur. Neurol. 38 (Suppl. 2), 2–7 (1997).
Fumimura, Y. et al. Analysis of the adrenal gland is useful for evaluating pathology of the peripheral autonomic nervous system in Lewy body disease. J. Neuropathol. Exp. Neurol. 66, 354–362 (2007).
Del Tredici, K., Hawkes, C. H., Ghebremedhin, E. & Braak, H. Lewy pathology in the submandibular gland of individuals with incidental Lewy body disease and sporadic Parkinson's disease. Acta Neuropathol. 119, 703–713 (2010).
Iwanaga, K. et al. Lewy body-type degeneration in cardiac plexus in Parkinson's disease and incidental Lewy body diseases. Neurology 52, 1269–1271 (1999).
Orimo, S. et al. Cardiac sympathetic denervation precedes neuronal loss in the sympathetic ganglia in Lewy body disease. Acta Neuropathol. 109, 583–588 (2005).
Ghebremedhin, E., Del Tredici, K., Langston, J. W. & Braak, H. Diminished tyrosine hydroxylase immunoreactivity in the cardiac conduction system and myocardium in Parkinson's disease: an anatomical study. Acta Neuropathol. 118, 777–784 (2009).
Jellinger, K. A. Pathology of Parkinson's disease. Changes other than in the nigrostriatal pathway. Mol. Chem. Neuropathol. 14, 153–197 (1991).
Lang, A. E. & Obeso, J. A. Challenges in Parkinson's disease: restoration of the nigrostriatal dopamine system is not enough. Lancet Neurol. 3, 309–316 (2004).
Langston, J. W. The Parkinson's complex: parkinsonism is just the tip of the iceberg. Ann. Neurol. 59, 591–596 (2006).
Dickson, D. W. et al. Neuropathology of non-motor features of Parkinson's disease. Parkinsonism Relat. Disord. 15 (Suppl. 3), S1–S5 (2009).
Lim, S. Y., Fox, S. H. & Lang, A. E. Overview of the extranigral aspects of Parkinson disease. Arch. Neurol. 66, 167–172 (2009).
Bloch, A., Probst, A., Bissig, H., Adams, H. & Tolnay, M. α-Synuclein pathology of the spinal and peripheral autonomic nmervous system in neurologically unimpaired elderly subjects. Neuropathol. Appl. Neurobiol. 12, 284–295 (2006).
Dickson, D. W., Uchikado, H., Fujishiro, H. & Tsuboi, Y. Evidence in favour of Braak staging of Parkinson's disease. Mov. Disord. 25 (Suppl. 1), S78–S82 (2010).
Halliday, G., McCann, H. & Shepherd, C. Evaluation of the Braak hypothesis: how far can it explain the pathogenesis of Parkinson's disease? Expert Rev. Neurother. 12, 673–686 (2012).
Braak, H. et al. Pathology associated with sporadic Parkinson's disease—where does it end? J. Neural Transm. 70, 89–97 (2006).
Uchikado, H., Lin, W. L., De Lucia, M. W. & Dickson, D. W. Alzheimer disease with amygdala Lewy bodies: a distinct form of α-synucleinopathy. J. Neuropathol. Exp. Neurol. 65, 685–697 (2006).
Saito, Y. et al. Lewy body-related α-synucleinopathy in aging. J. Neuropathol. Exp. Neurol. 63, 742–749 (2004).
Beach, T. G. et al. Unified staging system for Lewy body disorders: correlation with nigrostriatal degeneration, cognitive impairment and motor dysfunction. Acta Neuropathol. 117, 613–634 (2009).
Dickson, D. W. et al. Neuropathological assessment of Parkinson's disease: refining the diagnostic criteria. Lancet Neurol. 8, 1150–1157 (2009).
Braak, H., de Vos, R. A., Bohl, J. & Del Tredici, K. Gastric α-synuclein immunoreactive inclusions in Meissner's and Auerbach's plexuses in cases staged for Parkinson's disease-related brain pathology. Neurosci. Lett. 396, 67–72 (2006).
Del Tredici, K. & Braak, H. Lewy pathology and neurodegeneration in premotor Parkinson's disease. Mov. Disord. 27, 597–607 (2012).
Annerino, D. M. et al. Parkinson's disease is not associated with gastrointestinal myenteric ganglion neuron loss. Acta Neuropathol. 124, 665–680 (2012).
Shannon, K. M., Keshavarzian, A., Dodiya, H. B., Jakate, S. & Kordower, J. H. Is alpha-synuclein in the colon a biomarker for premotor Parkinson's disease? Evidence from 3 cases. Mov. Disord. 27, 716–719 (2012).
Klos, K. J. et al. α-Synuclein pathology in the spinal cord of neurologically asymptomatic aged individuals. Neurology 66, 1100–1102 (2006).
Schapira, A. H. & Tolosa, E. Molecular and clinical prodrome of Parkinson disease: implications for treatment. Nat. Rev. Neurol. 6, 309–317 (2010).
Li, J. Y. et al. Lewy bodies in grafted neurons in subjects with Parkinson's disease suggest host-to-graft disease propagation. Nat. Med. 14, 501–503 (2008).
Kordower, J. H., Chu, Y., Hauser, R. A., Freeman, T. B. & Olanow, C. W. Lewy body-like pathology in long-term embryonic nigral transplants in Parkinson's disease. Nat. Med. 14, 504–506 (2008).
Ahn, T. B., Langston, W. J., Aachi, V. R. & Dickson, D. W. Relationship of neighbouring tissue and gliosis to α-synuclein pathology in a fetal transplant for Parkinson's disease. Am. J. Neurodegener. Dis. 1, 49–59 (2012).
Li, J. Y. et al. Characterization of Lewy body pathology in 12- and 16-year-old intrastriatal mesencephalic grafts surviving in a patient with Parkinson's disease. Mov. Disord. 25, 1091–1096 (2010).
Greffard, S. et al. A stable proportion of Lewy body-bearing neurons in the substantia nigra suggests a model in which the Lewy body causes neuronal death. Neurobiol. Aging 31, 99–103 (2010).
Prusiner, S. B. A unifying role for prions in neurodegenerative diseases. Science 336, 1511–1513 (2012).
Parkkinen, L. et al. Disentangling the relationship between Lewy bodies and nigral neuronal loss in Parkinson's disease. J. Parkinsons Dis. 1, 277–286 (2011).
Hawkes, C. J., Del Tredici, K. & Braak, H. A timeline for Parkinson's disease. Parkinsonism Relat. Disord. 16, 79–84 (2010).
Neudorfer, O. et al. Occurrence of Parkinson's syndrome in type I Gaucher disease. Q. J. Med. 89, 691–694 (1996).
Sidransky, E. et al. Multicenter analysis of glucocerebrosidase mutations in Parkinson's disease. N. Engl. J. Med. 361, 1651–1661 (2009).
Rosenbloom, B. et al. The incidence of parkinsonism in patients with type 1 Gaucher disease: data from the ICGG Gaucher Registry. Blood Cells Mol. Dis. 46, 95–102 (2011).
Goker-Alpan, O., Stubblefield, B. K., Giasson, B. I. & Sidransky E. Glucocerebrosidase is present in α-synuclein inclusions in Lewy body disorders. Acta Neuropathol. 120, 641–649 (2010).
Westbroek, W., Gustafson, A. M. & Sidransky, E. Exploring the link between glucocerebrosidase mutations and parkinsonism. Trends Mol. Med. 17, 485–493 (2011).
Paisán-Ruíz, C. et al. Cloning of the gene containing mutations that cause PARK8-linked Parkinson's disease. Neuron 44, 595–600 (2004).
Zimprich, A. et al. Mutations in LRRK2 cause autosomal-dominant parkinsonism with pleomorphic pathology. Neuron 44, 601–607 (2004).
Ross, O. A. et al. Lrrk2 and Lewy body disease. Ann. Neurol. 59, 388–393 (2006).
Kitada, T. et al. Mutations in the Parkin gene cause autosomal recessive juvenile parkinsonism. Nature 392, 605–608 (1998).
Valente, E. M. et al. Hereditary early-onset Parkinson's disease caused by mutations in PINK1. Science 304, 1158–1160 (2004).
Park, J. et al. Mitochondrial dysfunction in Drosophila PINK1 mutants is complemented by parkin. Nature 441, 1157–1161 (2006).
Clark, I. E. et al. Drosophila pink1 is required for mitochondrial function and interacts genetically with parkin. Nature 441, 1162–1166 (2006).
Kondapalli, C. et al. PINK1 is activated by mitochondrial membrane depolarization and stimulates Parkin E3 ligase activity by phosphorylating serine 65. Open Biol. 2, 120080 (2012).
Youle, R. J. & Narendra, D. P. Mechanisms of mitophagy. Nat. Rev. Mol. Cell. Biol. 12, 9–14 (2011).
Bonifati, V. et al. Mutations in the DJ-1 gene associated with autosomal recessive early-onset parkinsonism. Science 299, 256–259 (2003).
Canet-Avilés, R. M. et al. The Parkinson's disease protein DJ-1 is neuroprotective due to cysteine-sulfinic acid-driven mitochondrial localization. Proc. Natl Acad. Sci. USA 101, 9103–9108 (2004).
Zarow, C., Lyness, S. A., Mortimer, J. A. & Chui, H. C. Neuronal loss is greater in the locus coeruleus than nucleus basalis and substantia nigra in Alzheimer and Parkinson diseases. Arch. Neurol. 60, 337–341 (2003).
Samaranch, L. et al. PINK1-linked parkinsonism is associated with Lewy body pathology. Brain 133, 1128–1142 (2010).
Papp, M. I., Kahn, J. E. & Lantos, P. L. Glial cytoplasmic inclusions in the CNS of patients with multiple system atrophy. J. Neurol. Sci. 94, 79–100 (1989).
Wakabayashi, K., Yoshimoto, M., Tsuji, S. & Takahashi, H. α-Synuclein immunoreactivity in glial cytoplasmic inclusions in multiple system atrophy. Neurosci. Lett. 249, 180–182 (1998).
Spillantini, M. G. et al. Filamentous α-synuclein inclusions link multiple system atrophy with Parkinson's disease and dementia with Lewy bodies. Neurosci. Lett. 251, 205–208 (1998).
Tu, P. H. et al. Glial cytoplasmic inclusions in white matter oligodendrocytes of multiple system atrophy brains contain insoluble α-synuclein. Ann. Neurol. 44, 415–422 (1998).
Scholz, S. W. et al. SNCA variants are associated with increased risk for multiple system atrophy. Ann. Neurol. 65, 610–614 (2009).
Al-Chalabi, A. et al. Genetic variants of the α-synuclein gene SNCA are associated with multiple system atrophy. PLoS ONE 22, e7114 (2009).
Abeliovich A. et al. Mice lacking α-synuclein display functional deficits in the nigrostriatal dopamine system. Neuron 25, 239–252 (2000).
Specht, C. G. & Schoepfer, R. Deletion of the α-synuclein locus in a subpopulation of C57BL/6J inbred mice. BMC Neurosci. 2, 11 (2001).
Greten-Harrison, B. et al. αβγ-Synuclein triple knockout mice reveal age-dependent neuronal dysfunction. Proc. Natl Acad. Sci. USA 107, 19573–19578 (2010).
McCormack, A. L. et al. α-Synuclein suppression by targeted small interfering RNA in the primate substantia nigra. PLoS ONE 5, e12122 (2010).
Lim, V. et al. α-Syn suppression reverses synaptic and memory defects in a mouse model of dementia with Lewy bodies. J. Neurosci. 31, 10076–10087 (2011).
Masliah, E. et al. Dopaminergic loss and inclusion body formation in α-synuclein mice: implications for neurodegenerative disorders. Science 287, 1265–1269 (2000).
Giasson, B. I. et al. Neuronal α-synucleinopathy with severe movement disorder in mice expressing A53T human α-synuclein. Neuron 34, 521–533 (2002).
Emmer, K. L., Waxman, E. A., Covey, J. P. & Giasson, B. I. E46K human α-synuclein transgenic mice develop Lewy-like and tau pathology associated with age-dependent, detrimental motor impairment. J. Biol. Chem. 286, 35104–35118 (2011).
Lee, K. W. et al. Enhanced phosphatase activity attenuates α-synucleinopathy in a mouse model. J. Neurosci. 31, 6963–6971 (2012).
Tofaris, G. K. et al. Pathological changes in dopaminergic nerve cells of the substantia nigra and olfactory bulb in mice transgenic for truncated human α-synuclein(1–120): implications for Lewy body disorders. J. Neurosci. 26, 3942–3950 (2006).
Garcia-Reitböck, P. et al. SNARE protein redistribution and synaptic failure in a transgenic mouse model of Parkinson's disease. Brain 133, 2032–2044 (2010).
Betarbet, R. et al. Chronic systemic pesticide exposure reproduces features of Parkinson's disease. Nat. Neurosci. 3, 1301–1306 (2000).
Pan-Montojo, F. et al. Progression of Parkinson's disease pathology is reproduced by intragastric administration of rotenone in mice. PLoS ONE 5, e8762 (2010).
Kirik, D. et al. Parkinson-like neurodegeneration induced by targeted overexpression of α-synuclein in the nigrostriatal system. J. Neurosci. 22, 2780–2791 (2002).
Lo Bianco, C., Ridet, J. L., Schneider, B. L., Deglon, N. & Aebischer, P. α-Synucleinopathy and selective dopaminergic neuron loss in a rat lentiviral-based model of Parkinson's disease. Proc. Natl Acad. Sci. USA 99, 10813–10818 (2002).
Taschenberger, G. et al. Aggregation of α-synuclein promotes progressive in vivo neurotoxicity in adult rat dopaminergic neurons. Acta Neuropathol. 123, 671–683 (2012).
Burré, J., Sharma, M. & Südhof, T. C. Systematic mutagenesis of α-synuclein reveals distinct sequence requirements for physiological and pathological activities. J. Neurosci. 32, 15227–15242 (2012).
Feany, M. B. & Bender, W. W. A Drosophila model of Parkinson's disease. Nature 404, 394–398 (2000).
Auluck, P. K., Chan, H. Y., Trojanowski, J. Q., Lee, V. M. & Bonini, N. M. Chaperone suppression of α-synuclein toxicity in a Drosophila model for Parkinson's disease. Science 295, 865–868 (2002).
Periquet, M., Fulga, T., Myllykangas, L., Schlossmacher, M. G. & Feany, M. B. Aggregated α-synuclein mediates dopaminergic neurotoxicity in vivo. J. Neurosci. 27, 3338–3346 (2007).
Lakso, M. et al. Dopaminergic neuronal loss and motor deficits in Caenorhabditis elegans overexpressing human α-synuclein. J. Neurochem. 86, 165–172 (2003).
Kuwahara, T. et al. A systematic RNAi screen reveals involvement of endocytic pathway in neuronal dysfunction in α-synuclein transgenic C. elegans. Hum. Mol. Genet. 17, 2997–3009 (2007).
Masuda, M. et al. Small molecule inhibitors of α-synuclein filament assembly. Biochemistry 45, 6085–6094 (2006).
Volpicelli-Daley, L. A. et al. Exogenous α-synuclein fibrils induce Lewy body pathology leading to synaptic dysfunction and neuron death. Neuron 72, 57–71 (2011).
Desplats, P. et al. Inclusion formation and neuronal cell death through neuron-to-neuron transmission of α-synuclein. Proc. Natl Acad. Sci. USA 106, 13010–13015 (2009).
Hansen, C. et al. α-Synuclein propagates from mouse brain to grafted dopaminergic neurons and seeds aggregation in cultured human cells. J. Clin. Invest. 121, 715–725 (2011).
Kordower, J. H. et al. Transfer of host-derived α-synuclein to grafted dopaminergic neurons in rat. Neurobiol. Dis. 43, 552–557 (2011).
Mougenot, A. L. et al. Prion-like acceleration of a synucleinopathy in a transgenic mouse model. Neurobiol. Aging 33, 2225–2228 (2012).
Luk, K. C. et al. Intracerebral inoculation of pathological α-synuclein initiates a rapidly progressive neurodegenerative α-synucleinopathy in mice. J. Exp. Med. 209, 975–986 (2012).
Masliah, E. et al. Effects of α-synuclein immunization in a mouse model of Parkinson's disease. Neuron 46, 857–868 (2005).
Masliah, E. et al. Passive immunization reduces behavioural and neuropathological deficits in an alpha-synuclein transgenic model of Lewy body disease. PLoS ONE 6, e19338 (2011).
Devine, M. J. et al. Parkinson's disease induced pluripotent stem cells with triplication of the α-synuclein locus. Nat. Commun. 2, 440 (2011).
Soldner, F. et al. Generation of isogenic pluripotent stem cells differing exclusively at two early onset Parkinson point mutations. Cell 146, 318–331 (2011).
Duffy, P. E. & Tennyson, V. M. Phase and electron microscopic observations of Lewy bodies and melanin granules in the substantia nigra and locus coeruleus in Parkinson's disease. J. Neuropathol. Exp. Neurol. 24, 398–414 (1965).
Parkinson, J. An Essay on the Shaking Palsy (Sherwood, Nealy and Jones, London, 1817).
Charcot, J. M. Leçons sur les Maladies du Système Nerveux Vol. 1 (Delahaye et Cie, Paris, 1875).
Von Economo, C. Die Encephalitis lethargica [German]. Wien. klin. Wochenschr. 30, 581–585 (1917).
Vogt, C. & Vogt, O. Zur Lehre der Erkrankung des striären Systems [German]. J. Psychol. Neurol. 26, 43–57 (1920).
Bradbury, S. & Eggleston, C. Postural hypotension: a report of three cases. Am. Heart J. 1, 75–86 (1925).
Ehringer, H. & Hornykiewicz, O. Verteilung von Noradrenalin und Dopamin (3-Hydroxytyramin) im Gehirn des Menschen und ihr Verhalten bei Erkrankungen des extrapyramidalen Systems [German]. Klin. Wochenschr. 38, 1236–1239 (1960).
Birkmayer, W. & Hornykiewicz, O. Der L-Dioxyphenylalanineffekt bei der Parkinson-Akinese [German]. Wien. klin. Wochenschr. 73, 787–788 (1961).
Cotzias, G. C., Van Woert, M. H. & Schiffer, L. M. Aromatic amino acids and modification of parkinsonism. N. Engl. J. Med. 276, 374–379 (1967).
Hoehn, M. M. & Yahr, M. D. Parkinsonism: onset, progression and mortality. Neurology 17, 427–442 (1967).
Calne, D. B., Stern, G. M., Laurence, D. R., Sharkey, J. & Armitage, P. L-DOPA in postencephalitic parkinsonism. Lancet 1, 744–747 (1969).
Sacks, O. Awakenings (Duckworth, London, 1973).
Ansari, K. A. & Johnson, A. J. Olfactory function in Parkinson's disease. J. Chronic Dis. 28, 493–497 (1975).
Kosaka, K., Oyanagi, S., Matsushita, M. & Hori, A. Presenile dementia with Alzheimer-, Pick- and Lewy-body changes. Acta Neuropathol. 36, 221–233 (1976).
Langston, J. W., Ballard, P., Tetrud, J. & Irwin, I. Chronic parkinsonism in humans due to a product of meperidine-analog synthesis. Science 219, 979–980 (1983).
Qualman, S., Haupt, H. M., Yang, P. & Hamilton, S. D. Esophageal Lewy bodies associated with ganglion cell loss in achalasia: similarity to Parkinson's disease. Gastroenterology 87, 848–856 (1984).
Schenck, C. H., Bundle, S. R., Ettinger. M. G. & Mahowald, M. W. Chronic behavioural disorders of human REM sleep: a new category of parasomnia. Sleep 9, 293–308 (1986).
Benabid, A. L., Pollak, P., Louveau, A., Henry, S. & de Rougemont, J. Combined (thalamotomy and stimulation) stereotactic surgery of the VIM thalamic nucleus for bilateral Parkinson disease. Appl. Neurophysiol. 50, 344–346 (1987).
McKeith, I. G. et al. Consensus guidelines for the clinical and pathological diagnosis of dementia with Lewy bodies (DLB). Neurology 47, 1113–1124 (1996).
Saiki, M. et al. Association of the human leucocyte antigen region with susceptibility to Parkinson's disease. J. Neurol. Neurosurg. Psychiatry 81, 890–891 (2010).
Braak, H. et al. Stanley Fahn Lecture 2005: The staging procedure for the inclusion body pathology associated with sporadic Parkinson's disease reconsidered. Mov. Disord. 21, 2042–2051 (2006).
Acknowledgements
We thank Mrs Nathalie Cornée for tracking down references from times past. This article was supported in part by the UK Medical Research Council (U105184291), Parkinson's UK and the Deutsche Forschungsgemeinschaft (grant TR 1000/1-1).
Author information
Authors and Affiliations
Contributions
All authors contributed to researching data for the article, discussions of the content, writing the article, and review and/or editing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
M. Goedert has received research support from Eli Lilly, and has acted as a consultant for GlaxoSmithKline and Hoffmann-La Roche. The other authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Goedert, M., Spillantini, M., Del Tredici, K. et al. 100 years of Lewy pathology. Nat Rev Neurol 9, 13–24 (2013). https://doi.org/10.1038/nrneurol.2012.242
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneurol.2012.242
This article is cited by
-
The multifaceted functions of β-arrestins and their therapeutic potential in neurodegenerative diseases
Experimental & Molecular Medicine (2024)
-
Cell-to-cell transmitted alpha-synuclein recapitulates experimental Parkinson’s disease
npj Parkinson's Disease (2024)
-
Neuropathogenesis-on-chips for neurodegenerative diseases
Nature Communications (2024)
-
Autophagy and UPS pathway contribute to nicotine-induced protection effect in Parkinson’s disease
Experimental Brain Research (2024)
-
Neuronal SNCA transcription during Lewy body formation
Acta Neuropathologica Communications (2023)