Abstract
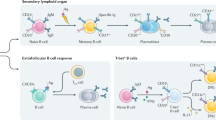
B cells and antibodies account for the most prominent immunodiagnostic feature in patients with multiple sclerosis (MS), namely oligoclonal bands. Furthermore, evidence is accumulating that B cells and antibodies contribute to MS pathogenesis in at least a subset of patients. The CNS provides a B-cell-fostering environment that includes B-cell trophic factors such as BAFF (B-cell-activating factor of the TNF family), APRIL (a proliferation-inducing ligand), and the plasma-cell survival factor CXCL12. Owing to this environment, the CNS of patients with MS is not only the target of the immunopathological process, but also becomes the site of local antibody production. B cells can increase or dampen CNS inflammation, but their proinflammatory effects seem to be more prominent in most patients, as B-cell depletion is a promising therapeutic strategy. Other therapies not primarily designed to target B cells have numerous effects on the B-cell compartment. This Review summarizes key features of B-cell biology, the role of B cells and antibodies in CNS inflammation, and current attempts to identify the targets of pathogenic antibodies in MS. We also review the effects of approved and investigational interventions—including CD20-depleting antibodies, BAFF/APRIL-depleting agents, alemtuzumab, natalizumab, FTY720, IFN-β, glatiramer acetate, steroids and plasma exchange—on B-cell immunology.
Key Points
-
B cells regulate CNS inflammation in various ways
-
The CNS in multiple sclerosis (MS) provides a B-cell-fostering environment
-
Cerebrospinal fluid levels of the B-cell-attracting chemokine CXCL13 are linked to CNS inflammation and local IgG production, and have prognostic value in MS
-
B-cell depletion is a promising MS therapy, largely unrelated to effects on IgG production
-
Many immunomodulatory therapies in MS affect the B-cell compartment
-
Identification and validation of novel autoantibodies in MS is a current research focus; candidate antigens include myelin oligodendrocyte protein, axoglial targets around the node of Ranvier, and the potassium channel KIR4.1
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Change history
22 October 2012
In the version of this article initially published online, in Figure 2 the bar indicating expression of IgM, IgA, IgG or IgE was erroneously extended to include centroblasts, and bars indicating expression of cell-surface markers were incomplete. This has been corrected for the print, HTML and PDF versions of the article.
References
Hiepe, F. et al. Long-lived autoreactive plasma cells drive persistent autoimmune inflammation. Nat. Rev. Rheumatol. 7, 170–178 (2011).
Anthony, R. M. & Nimmerjahn, F. The role of differential IgG glycosylation in the interaction of antibodies with FcγRs in vivo. Curr. Opin. Organ Transplant. http://dx.doi.org/10.1097/MOT.0b013e328342538f.
Barr, T. A. et al. B cell depletion therapy ameliorates autoimmune disease through ablation of IL-6-producing B cells. J. Exp. Med. 209, 1001–1010 (2012).
Fillatreau, S., Sweenie, C. H., McGeachy, M. J., Gray, D. & Anderton, S. M. B cells regulate autoimmunity by provision of IL-10. Nat. Immunol. 3, 944–950 (2002).
Iwata, Y. et al. Characterization of a rare IL-10 competent B-cell subset in humans that parallels mouse regulatory B10 cells. Blood 117, 530–541 (2011).
Weber, M. S. et al. B-cell activation influences T-cell polarization and outcome of anti-CD20 B-cell depletion in central nervous system autoimmunity. Ann. Neurol. 68, 369–383 (2010).
Meinl, E., Krumbholz, M. & Hohlfeld, R. B lineage cells in the inflammatory central nervous system environment: migration, maintenance, local antibody production, and therapeutic modulation. Ann. Neurol. 59, 880–892 (2006).
Pöllinger, B. et al. Spontaneous relapsing–remitting EAE in the SJL/J mouse: MOG-reactive transgenic T cells recruit endogenous MOG-specific B cells. J. Exp. Med. 206, 1303–1316 (2009).
Hauser, S. L. et al. B-cell depletion with rituximab in relapsing–remitting multiple sclerosis. N. Engl. J. Med. 358, 676–688 (2008).
Kappos, L. et al. Ocrelizumab in relapsing–remitting multiple sclerosis: a phase 2, randomised, placebo-controlled, multicentre trial. Lancet 378, 1779–1787 (2011).
Jarius, S. & Wildemann, B. AQP4 antibodies in neuromyelitis optica: diagnostic and pathogenetic relevance. Nat. Rev. Neurol. 6, 383–392 (2010).
Keegan, M. et al. Relation between humoral pathological changes in multiple sclerosis and response to therapeutic plasma exchange. Lancet 366, 579–582 (2005).
Elliott, C. et al. Functional identification of pathogenic autoantibody responses in patients with multiple sclerosis. Brain 135, 1819–1833 (2012).
Lucchinetti, C. et al. Heterogeneity of multiple sclerosis lesions: implications for the pathogenesis of demyelination. Ann. Neurol. 47, 707–717 (2000).
Barnett, M. H., Parratt, J. D., Cho, E. S. & Prineas, J. W. Immunoglobulins and complement in postmortem multiple sclerosis tissue. Ann. Neurol. 65, 32–46 (2009).
Breij, E. C. et al. Homogeneity of active demyelinating lesions in established multiple sclerosis. Ann. Neurol. 63, 16–25 (2008).
Barnett, M. H., Parratt, J. D., Pollard, J. D. & Prineas, J. W. MS: is it one disease? Int. MS J. 16, 57–65 (2009).
Mathey, E. K. et al. Neurofascin as a novel target for autoantibody-mediated axonal injury. J. Exp. Med. 204, 2363–2372 (2007).
Derfuss, T. et al. Contactin-2/TAG-1-directed autoimmunity is identified in multiple sclerosis patients and mediates gray matter pathology in animals. Proc. Natl Acad. Sci. USA 106, 8302–8307 (2009).
Genain, C. P. et al. Antibody facilitation of multiple sclerosis-like lesions in a nonhuman primate. J. Clin. Invest. 96, 2966–2974 (1995).
Mayer, M. C. & Meinl, E. Glycoproteins as targets of autoantibodies in CNS inflammation: MOG and more. Ther. Adv. Neurol. Disord. 5, 147–159 (2012).
O'Connor, K. C. et al. Self-antigen tetramers discriminate between myelin autoantibodies to native or denatured protein. Nat. Med. 13, 211–217 (2007).
McLaughlin, K. A. et al. Age-dependent B cell autoimmunity to a myelin surface antigen in pediatric multiple sclerosis. J. Immunol. 183, 4067–4076 (2009).
Pröbstel, A. K. et al. Antibodies to MOG are transient in childhood acute disseminated encephalomyelitis. Neurology 77, 580–588 (2011).
Mader, S. et al. Complement activating antibodies to myelin oligodendrocyte glycoprotein in neuromyelitis optica and related disorders. J. Neuroinflammation 8, 184 (2011).
Owens, G. P. et al. Antibodies produced by clonally expanded plasma cells in multiple sclerosis cerebrospinal fluid. Ann. Neurol. 65, 639–649 (2009).
von Büdingen, H.-C., Harrer, M. D., Kuenzle, S., Meier, M. & Goebels, N. Clonally expanded plasma cells in the cerebrospinal fluid of MS patients produce myelin-specific antibodies. Eur. J. Immunol. 38, 2014–2023 (2008).
Lambracht-Washington, D. et al. Antigen specificity of clonally expanded and receptor edited cerebrospinal fluid B cells from patients with relapsing remitting MS. J. Neuroimmunol. 186, 164–176 (2007).
Dhaunchak, A. S. et al. Implication of perturbed axoglial apparatus in early pediatric multiple sclerosis. Ann. Neurol. 71, 601–613 (2012).
Kanter, J. L. et al. Lipid microarrays identify key mediators of autoimmune brain inflammation. Nat. Med. 12, 138–143 (2006).
Brennan, K. M. et al. Lipid arrays identify myelin-derived lipids and lipid complexes as prominent targets for oligoclonal band antibodies in multiple sclerosis. J. Neuroimmunol. 238, 87–95 (2011).
Ho, P. P. et al. Identification of naturally occurring fatty acids of the myelin sheath that resolve neuroinflammation. Sci. Transl. Med. 4, 137ra73 (2012).
Quintana, F. J. et al. Antigen microarrays identify unique serum autoantibody signatures in clinical and pathologic subtypes of multiple sclerosis. Proc. Natl Acad. Sci. USA 105, 18889–18894 (2008).
Srivastava, R. et al. Potassium channel KIR4.1 as an immune target in multiple sclerosis. N. Engl. J. Med. 367, 115–123 (2012).
Obermeier, B. et al. Matching of oligoclonal immunoglobulin transcriptomes and proteomes of cerebrospinal fluid in multiple sclerosis. Nat. Med. 14, 688–693 (2008).
Tintore, M. et al. Isolated demyelinating syndromes: comparison of CSF oligoclonal bands and different MR imaging criteria to predict conversion to CDMS. Mult. Scler. 7, 359–363 (2001).
Sharief, M. K. & Thompson, E. J. The predictive value of intrathecal immunoglobulin synthesis and magnetic resonance imaging in acute isolated syndromes for subsequent development of multiple sclerosis. Ann. Neurol. 29, 147–151 (1991).
Villar, L. M. et al. Intrathecal synthesis of oligoclonal IgM against myelin lipids predicts an aggressive disease course in MS. J. Clin. Invest. 115, 187–194 (2005).
Stauch, C. et al. Intrathecal IgM synthesis in pediatric MS is not a negative prognostic marker of disease progression: quantitative versus qualitative IgM analysis. Mult. Scler. 17, 327–334 (2011).
Jarius, S. et al. The intrathecal, polyspecific antiviral immune response: specific for MS or a general marker of CNS autoimmunity? J. Neurol. Sci. 280, 98–100 (2009).
Bednarova, J., Stourac, P. & Adam, P. Relevance of immunological variables in neuroborreliosis and multiple sclerosis. Acta Neurol. Scand. 112, 97–102 (2005).
Cepok, S. et al. Patterns of cerebrospinal fluid pathology correlate with disease progression in multiple sclerosis. Brain 124, 2169–2176 (2001).
Cepok, S. et al. Short-lived plasma blasts are the main B cell effector subset during the course of multiple sclerosis. Brain 128, 1667–1676 (2005).
Corcione, A. et al. Recapitulation of B cell differentiation in the central nervous system of patients with multiple sclerosis. Proc. Natl Acad. Sci. USA 101, 11064–11069 (2004).
Kuenz, B. et al. Cerebrospinal fluid B cells correlate with early brain inflammation in multiple sclerosis. PLoS ONE 3, e2559 (2008).
Cameron, E. M. et al. Potential of a unique antibody gene signature to predict conversion to clinically definite multiple sclerosis. J. Neuroimmunol. 213, 123–130 (2009).
Ligocki, A. J. et al. A unique antibody gene signature is prevalent in the central nervous system of patients with multiple sclerosis. J. Neuroimmunol. 226, 192–193 (2010).
Lovato, L. et al. Related B cell clones populate the meninges and parenchyma of patients with multiple sclerosis. Brain 134, 534–541 (2011).
Obermeier, B. et al. Related B cell clones that populate the CSF and CNS of patients with multiple sclerosis produce CSF immunoglobulin. J. Neuroimmunol. 233, 245–248 (2011).
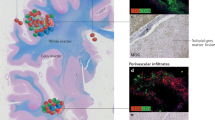
Serafini, B., Rosicarelli, B., Magliozzi, R., Stigliano, E. & Aloisi, F. Detection of ectopic B-cell follicles with germinal centers in the meninges of patients with secondary progressive multiple sclerosis. Brain Pathol. 14, 164–174 (2004).
Lucchinetti, C. F. et al. Inflammatory cortical demyelination in early multiple sclerosis. N. Engl. J. Med. 365, 2188–2197 (2011).
Howell, O. W. et al. Meningeal inflammation is widespread and linked to cortical pathology in multiple sclerosis. Brain 134, 2755–2771 (2011).
Kutzelnigg, A. et al. Cortical demyelination and diffuse white matter injury in multiple sclerosis. Brain 128, 2705–2712 (2005).
Kooi, E.-J., Geurts, J. J., van Horssen, J., Bø, L. & van der Valk, P. Meningeal inflammation is not associated with cortical demyelination in chronic multiple sclerosis. J. Neuropathol. Exp. Neurol. 68, 1021–1028 (2009).
Ascherio, A., Munger, K. L. & Lunemann, J. D. The initiation and prevention of multiple sclerosis. Nat. Rev. Neurol. http://dx.doi.org/10.1038/nrneurol.2012.198.
Charo, I. F. & Ransohoff, R. M. The many roles of chemokines and chemokine receptors in inflammation. N. Engl. J. Med. 354, 610–621 (2006).
Krumbholz, M. et al. CCL19 is constitutively expressed in the CNS, up-regulated in neuroinflammation, active and also inactive multiple sclerosis lesions. J. Neuroimmunol. 190, 72–79 (2007).
Krumbholz, M. et al. Chemokines in multiple sclerosis: CXCL12 and CXCL13 up-regulation is differentially linked to CNS immune cell recruitment. Brain 129, 200–211 (2006).
Pashenkov, M., Soderstrom, M. & Link, H. Secondary lymphoid organ chemokines are elevated in the cerebrospinal fluid during central nervous system inflammation. J. Neuroimmunol. 135, 154–160 (2003).
Rupprecht, T. A. et al. The chemokine CXCL13 (BLC): a putative diagnostic marker for neuroborreliosis. Neurology 65, 448–450 (2005).
Kowarik, M. et al. CXCL13 is the major determinant for B cell recruitment to the CSF during neuroinflammation. J. Neuroinflammation 9, 93 (2012).
Khademi, M. et al. Cerebrospinal fluid CXCL13 in multiple sclerosis: a suggestive prognostic marker for the disease course. Mult. Scler. 17, 335–343 (2011).
Brettschneider, J. et al. The chemokine CXCL13 is a prognostic marker in clinically isolated syndrome (CIS). PLoS ONE 5, e11986 (2010).
MacKay, F. & Schneider, P. Cracking the BAFF code. Nat. Rev. Immunol. 9, 491–502 (2009).
Krumbholz, M. et al. BAFF is produced by astrocytes and up-regulated in multiple sclerosis lesions and primary central nervous system lymphoma. J. Exp. Med. 201, 195–200 (2005).
Martin, F. & Chan, A. C. B cell immunobiology in disease: evolving concepts from the clinic. Annu. Rev. Immunol. 24, 467–496 (2006).
Martin Mdel, P. et al. Depletion of B lymphocytes from cerebral perivascular spaces by rituximab. Arch. Neurol. 66, 1016–1020 (2009).
Pers, J. O. et al. BAFF-modulated repopulation of B lymphocytes in the blood and salivary glands of rituximab-treated patients with Sjögren's syndrome. Arthritis Rheum. 56, 1464–1477 (2007).
Cambridge, G. et al. B cell depletion therapy in systemic lupus erythematosus: effect on autoantibody and antimicrobial antibody profiles. Arthritis Rheum. 54, 3612–3622 (2006).
Cornec, D., Avouac, J., Youinou, P. & Saraux, A. Critical analysis of rituximab-induced serological changes in connective tissue diseases. Autoimmun. Rev. 8, 515–519 (2009).
Pellkofer, H. L. et al. Long-term follow-up of patients with neuromyelitis optica after repeated therapy with rituximab. Neurology 76, 1310–1315 (2011).
Huang, H., Benoist, C. & Mathis, D. Rituximab specifically depletes short-lived autoreactive plasma cells in a mouse model of inflammatory arthritis. Proc. Natl Acad. Sci. USA 107, 4658–4663 (2010).
Lehmann-Horn, K. et al. Anti-CD20 B-cell depletion enhances monocyte reactivity in neuroimmunological disorders. J. Neuroinflammation 8, 146 (2011).
European Medicines Agency. Human Medicines Database: EMEA-000310-PIP03-10. European Medicines Agency [online], (2012).
Tak, P. P. et al. Safety and efficacy of ocrelizumab in patients with rheumatoid arthritis and an inadequate response to at least one tumor necrosis factor inhibitor: results of a forty-eight-week randomized, double-blind, placebo-controlled, parallel-group phase III trial. Arthritis Rheum. 64, 360–370 (2012).
Sorensen, P. S. et al. Magnetic resonance imaging (MRI) efficacy of ofatumumab in relapsing–remitting multiple sclerosis (RRMS)—24-week results of a phase II study. Presented at the 26th Congress of the European Committee for Treatment and Research in Multiple Sclerosis (2010).
CAMMS223 Trial Investigators. Alemtuzumab vs. interferon beta-1a in early multiple sclerosis. New Engl. J. Med. 359, 1786–1801 (2008).
Coles, A. J. et al. Alemtuzumab more effective than interferon β-1a at 5-year follow-up of CAMMS223 Clinical Trial. Neurology 78, 1069–1078 (2012).
Kirk, A. D. et al. Results from a human renal allograft tolerance trial evaluating the humanized CD52-specific monoclonal antibody alemtuzumab (CAMPATH-1H). Transplantation 76, 120–129 (2003).
Sanofi. Alemtuzumab (Lemtrada™*) significantly reduces relapses in multiple sclerosis vs interferon beta-1a in a phase III study. Sanofi [online], (2011).
Jones, J. L. & Coles, A. J. Spotlight on alemtuzumab. Int. MS J. 16, 77–81 (2009).
Cossburn, M. et al. Autoimmune disease after alemtuzumab treatment for multiple sclerosis in a multicenter cohort. Neurology 77, 573–579 (2011).
Thompson, S., Jones, J., Cox, A., Compston, D. & Coles, A. B-cell reconstitution and BAFF after alemtuzumab (Campath-1H) treatment of multiple sclerosis. J. Clin. Immunol. 30, 99–105 (2010).
Darce, J. R., Arendt, B. K., Wu, X. & Jelinek, D. F. Regulated expression of BAFF-binding receptors during human B cell differentiation. J. Immunol. 179, 7276–7286 (2007).
Stohl, W. & Hilbert, D. M. The discovery and development of belimumab: the anti-BLyS-lupus connection. Nat. Biotech. 30, 69–77 (2012).
Hartung, H. P. & Kieseier, B. C. Atacicept: targeting B cells in multiple sclerosis. Ther. Adv. Neurol. Disord. 3, 205–216 (2010).
Tak, P. P. et al. Atacicept in patients with rheumatoid arthritis: results of a multicenter, phase Ib, double-blind, placebo-controlled, dose-escalating, single- and repeated-dose study. Arthritis Rheum. 58, 61–72 (2007).
Genovese, M. C., Kinnman, N., de La Bourdonnaye, G., Pena Rossi, C. & Tak, P. P. Atacicept in patients with rheumatoid arthritis and an inadequate response to tumor necrosis factor antagonist therapy: results of a phase II, randomized, placebo-controlled, dose-finding trial. Arthritis Rheum. 63, 1793–1803 (2011).
Yang, M. et al. Novel function of B cell-activating factor in the induction of IL-10 producing regulatory B cells. J. Immunol. 184, 3321–3325 (2010).
Stohl, W. et al. Inverse association between circulating APRIL levels and serological and clinical disease activity in patients with systemic lupus erythematosus. Ann. Rheum. Dis. 63, 1096–1103 (2004).
Zhang, L. et al. Identification of BLyS (B lymphocyte stimulator), a non-myelin-associated protein, as a functional ligand for Nogo-66 receptor. J. Neurosci. 29, 6348–6352 (2009).
Huntington, N. D. et al. A BAFF antagonist suppresses experimental autoimmune encephalomyelitis by targeting cell-mediated and humoral immune responses. Int. Immunol. 18, 1473–1485 (2006).
Kim, S. S., Richman, D. P., Zamvil, S. S. & Agius, M. A. Accelerated central nervous system autoimmunity in BAFF-receptor-deficient mice. J. Neurol. Sci. 306, 9–15 (2011).
Ransohoff, R. M. & Engelhardt, B. The anatomical and cellular basis of immune surveillance in the central nervous system. Nat. Rev. Immunol. 12, 623–635 (2012).
Krumbholz, M., Meinl, I., Kümpfel, T., Hohlfeld, R. & Meinl, E. Natalizumab disproportionately increases circulating pre-B and B cells in multiple sclerosis. Neurology. 71, 1350–1354 (2008).
Kowarik, M. C. et al. Differential effects of fingolimod (FTY720) on immune cells in the CSF and blood of patients with MS. Neurology 76, 1214–1221 (2011).
Putzki, N., Baranwal, M. K., Tettenborn, B., Limmroth, V. & Kreuzfelder, E. Effects of natalizumab on circulating B cells, T regulatory cells and natural killer cells. Eur. Neurol. 63, 311–317 (2010).
Lesesve, J. F., Debouverie, M., Decarvalho Bittencourt, M. & Bene, M. C. CD49d blockade by natalizumab therapy in patients with multiple sclerosis increases immature B-lymphocytes. Bone Marrow Transplant. 46, 1489–1491 (2011).
Planas, R., Jelcic, I., Schippling, S., Martin, R. & Sospedra, M. Natalizumab treatment perturbs memory- and marginal zone-like B-cell homing in secondary lymphoid organs in multiple sclerosis. Eur. J. Immunol. 42, 790–798 (2012).
Stuve, O. et al. Immune surveillance in multiple sclerosis patients treated with natalizumab. Ann. Neurol. 59, 743–747 (2006).
Villar, L. M. et al. Immunological markers of optimal response to natalizumab in multiple sclerosis. Arch. Neurol. 69, 191–197 (2012).
Schwab, S. R. & Cyster, J. G. Finding a way out: lymphocyte egress from lymphoid organs. Nat. Immunol. 8, 1295–1301 (2007).
Hohlfeld, R., Barkhof, F. & Polman, C. Future clinical challenges in multiple sclerosis: relevance to sphingosine 1-phosphate receptor modulator therapy. Neurology 76, S28–S37 (2011).
Aktas, O., Kury, P., Kieseier, B. & Hartung, H.-P. Fingolimod is a potential novel therapy for multiple sclerosis. Nat. Rev. Neurol. 6, 373–382 (2010).
Cinamon, G. et al. Sphingosine 1-phosphate receptor 1 promotes B cell localization in the splenic marginal zone. Nat. Immunol. 5, 713–720 (2004).
Ando, S. et al. FTY720 exerts a survival advantage through the prevention of end-stage glomerular inflammation in lupus-prone BXSB mice. Biochem. Biophys. Res. Commun. 394, 804–810 (2010).
Han, S. et al. FTY720 suppresses humoral immunity by inhibiting germinal center reaction. Blood 104, 4129–4133 (2004).
Boulton, C., Meiser, K., David, O. J. & Schmouder, R. Pharmacodynamic effects of steady-state fingolimod on antibody response in healthy volunteers: a 4-week, randomized, placebo-controlled, parallel-group, multiple-dose study. J. Clin. Pharmacol. http://dx.doi.org/10.1177/0091270011427908.
Mehling, M. et al. Antigen-specific adaptive immune responses in fingolimod-treated multiple sclerosis patients. Ann. Neurol. 69, 408–413 (2011).
Sinha, R. K., Park, C., Hwang, I. Y., Davis, M. D. & Kehrl, J. H. B lymphocytes exit lymph nodes through cortical lymphatic sinusoids by a mechanism independent of sphingosine-1-phosphate-mediated chemotaxis. Immunity 30, 434–446 (2009).
Matloubian, M. et al. Lymphocyte egress from thymus and peripheral lymphoid organs is dependent on S1P receptor 1. Nature 427, 355–360 (2004).
Kabashima, K. et al. Plasma cell S1P1 expression determines secondary lymphoid organ retention versus bone marrow tropism. J. Exp. Med. 203, 2683–2690 (2006).
Cinamon, G., Zachariah, M. A., Lam, O. M., Foss, F. W. Jr & Cyster, J. G. Follicular shuttling of marginal zone B cells facilitates antigen transport. Nat. Immunol. 9, 54–62 (2008).
Sedlakova, K., Muckersie, E., Robertson, M., Filipec, M. & Forrester, J. V. FTY720 in corneal concordant xenotransplantation. Transplantation 79, 297–303 (2005).
Okazaki, H. et al. Effects of FTY720 in MRL-lpr/lpr mice: therapeutic potential in systemic lupus erythematosus. J. Rheumatol. 29, 707–716 (2002).
Krumbholz, M. et al. Interferon-β increases BAFF levels in multiple sclerosis: implications for B cell autoimmunity. Brain 131, 1415–1463 (2008).
Palace, J., Leite, M. I., Nairne, A. & Vincent, A. Interferon beta treatment in neuromyelitis optica: increase in relapses and aquaporin 4 antibody titers. Arch. Neurol. 67, 1016–1017 (2010).
Warabi, Y., Matsumoto, Y. & Hayashi, H. Interferon beta-1b exacerbates multiple sclerosis with severe optic nerve and spinal cord demyelination. J. Neurol. Sci. 252, 57–61 (2007).
Kantor, A. B. et al. Identification of short-term pharmacodynamic effects of interferon-beta-1a in multiple sclerosis subjects with broad-based phenotypic profiling. J. Neuroimmunol. 188, 103–116 (2007).
Zanotti, C. et al. Opposite effects of interferon-beta on new B and T cell release from production sites in multiple sclerosis patients. J. Neuroimmunol. 240, 147–150 (2011).
Farina, C., Weber, M. S., Meinl, E., Wekerle, H. & Hohlfeld, R. Glatiramer acetate in multiple sclerosis: update on potential mechanisms of action. Lancet Neurol. 4, 567–575 (2005).
Begum-Haque, S. et al. Augmentation of regulatory B cell activity in experimental allergic encephalomyelitis by glatiramer acetate. J. Neuroimmunol. 232, 136–144 (2011).
Kala, M. et al. B cells from glatiramer acetate-treated mice suppress experimental autoimmune encephalomyelitis. Exp. Neurol. 221, 136–145 (2010).
Ireland, S. J. et al. Antibody-independent B cell effector functions in relapsing remitting multiple sclerosis: clues to increased inflammatory and reduced regulatory B cell capacity. Autoimmunity 45, 400–414 (2012).
Andreau, K., Lemaire, C., Souvannavong, V. & Adam, A. Induction of apoptosis by dexamethasone in the B cell lineage. Immunopharmacology 40, 67–76 (1998).
Lill-Elghanian, D., Schwartz, K., King, L. & Fraker, P. Glucocorticoid-induced apoptosis in early B cells from human bone marrow. Exp. Biol. Med. 227, 763–770 (2002).
Motyka, B., Bhogal, H. S. & Reynolds, J. D. Apoptosis of ileal Peyer's patch B cells is increased by glucocorticoids or anti-immunoglobulin antibodies. Eur. J. Immunol. 25, 1865–1871 (1995).
Dau, P. C. Increased antibody production in peripheral blood mononuclear cells after plasma exchange therapy in multiple sclerosis. J. Neuroimmunol. 62, 197–200 (1995).
Dau, P. C. Increased proliferation of blood mononuclear cells after plasmapheresis treatment of patients with demyelinating disease. J. Neuroimmunol. 30, 15–21 (1990).
Paglieroni, T., Caggiano, V. & MacKenzie, M. R. Effects of plasmapheresis on peripheral blood mononuclear cell populations from patients with macroglobulinemia. J. Clin. Apher. 3, 202–208 (1987).
Gold, R., Stangel, M. & Dalakas, M. C. The use of intravenous immunoglobulin in neurology—therapeutic considerations and practical issues. Nat. Clin. Pract. Neurol. 3, 36–44 (2007).
Nimmerjahn, F. & Ravetch, J. V. Anti-inflammatory actions of intravenous immunoglobulin. Annu. Rev. Immunol. 26, 513–533 (2008).
Chen, S. et al. Modulatory effects of 1,25-dihydroxyvitamin D3 on human B cell differentiation. J. Immunol. 179, 1634–1647 (2007).
Kivity, S. et al. Vitamin D and autoimmune thyroid diseases. Cell. Mol. Immunol. 8, 243–247 (2011).
Simon, K. C., Munger, K. L. & Ascherio, A. Vitamin D and multiple sclerosis: epidemiology, immunology, and genetics. Curr. Opin. Neurol. 25, 246–251 (2012).
Knippenberg, S. et al. Effect of vitamin D3 supplementation on peripheral B cell differentiation and isotype switching in patients with multiple sclerosis. Mult. Scler. 17, 1418–1423 (2011).
Hedstrom, A. K., Baarnhielm, M., Olsson, T. & Alfredsson, L. Tobacco smoking, but not Swedish snuff use, increases the risk of multiple sclerosis. Neurology 73, 696–701 (2009).
Zivadinov, R. et al. Smoking is associated with increased lesion volumes and brain atrophy in multiple sclerosis. Neurology 73, 504–510 (2009).
Healy, B. C. et al. Smoking and disease progression in multiple sclerosis. Arch. Neurol. 66, 858–864 (2009).
Handel, A. E., Giovannoni, G., Ebers, G. C. & Ramagopalan, S. V. Environmental factors and their timing in adult-onset multiple sclerosis. Nat. Rev. Neurol. 6, 156–166 (2010).
Handel, A. E. et al. Smoking and multiple sclerosis: an updated meta-analysis. PLoS ONE 6, e16149 (2011).
Fusby, J. S. et al. Cigarette smoke-induced effects on bone marrow B-cell subsets and CD4+:CD8+ T-cell ratios are reversed by smoking cessation: influence of bone mass on immune cell response to and recovery from smoke exposure. Inhal. Toxicol. 22, 785–796 (2010).
Palmer, V. L. et al. N-acetylcysteine increases the frequency of bone marrow pro-B/pre-B cells, but does not reverse cigarette smoking-induced loss of this subset. PLoS ONE 6, e24804 (2011).
Pan, F. et al. Impact of female cigarette smoking on circulating B cells in vivo: the suppressed ICOSLG, TCF3, and VCAM1 gene functional network may inhibit normal cell function. Immunogenetics 62, 237–251 (2010).
Baranzini, S. E. et al. B cell repertoire diversity and clonal expansion in multiple sclerosis brain lesions. J. Immunol. 163, 5133–5144 (1999).
Qin, Y. et al. Clonal expansion and somatic hypermutation of VH genes of B cells from cerebrospinal fluid in multiple sclerosis. J. Clin. Invest. 102, 1045–1050 (1998).
Reijmers, R. M. et al. Disruption of heparan sulfate proteoglycan conformation perturbs B cell maturation and APRIL-mediated plasma cell survival. Blood 117, 6162–6171 (2011).
Hauser, S. et al. A phase II randomized, placebo-controlled, multicenter trial of rituximab in adults with relapsing remitting multiple sclerosis (RRMS). Neurology 68 (Suppl. 1), A99–A100 (2007).
Hawker, K. et al. Rituximab in patients with primary progressive multiple sclerosis: results of a randomized double-blind placebo-controlled multicenter trial. Ann. Neurol. 66, 460–471 (2009).
Kim, S. H., Kim, W., Li, X. F., Jung, I. J. & Kim, H. J. Repeated treatment with rituximab based on the assessment of peripheral circulating memory B cells in patients with relapsing neuromyelitis optica over 2 years. Arch. Neurol. 68, 1412–1420 (2011).
Cross, A. H., Stark, J. L., Lauber, J., Ramsbottom, M. J. & Lyons, J. A. Rituximab reduces B cells and T cells in cerebrospinal fluid of multiple sclerosis patients. J. Neuroimmunol. 180, 63–70 (2006).
Piccio, L. et al. Changes in B- and T-lymphocyte and chemokine levels with rituximab treatment in multiple sclerosis. Arch. Neurol. 67, 707–714 (2010).
Vallerskog, T. et al. Differential effects on BAFF and APRIL levels in rituximab-treated patients with systemic lupus erythematosus and rheumatoid arthritis. Arthritis Res. Ther. 8, R167 (2006).
Kreuzaler, M. et al. Soluble BAFF levels inversely correlate with peripheral B cell numbers and the expression of BAFF receptors. J. Immunol. 188, 497–503 (2012).
Heidt, S., Hester, J., Shankar, S., Friend, P. J. & Wood, K. J. B-cell repopulation after alemtuzumab induction: transient increase in transitional B cells and long-term dominance of naive B cells. Am. J. Transplant. 12, 1784–1792 (2012).
Anderson, A. E. et al. Immunity 12 years after alemtuzumab in RA: CD5+ B-cell depletion, thymus-dependent T-cell reconstitution and normal vaccine responses. Rheumatology 51, 1397–1406 (2012).
Bloom, D. et al. BAFF is increased in renal transplant patients following treatment with alemtuzumab. Am. J. Transplant. 9, 1835–1845 (2009).
LaMattina, J. C. et al. Alemtuzumab as compared to alternative contemporary induction regimens. Transpl. Int. 25, 518–526 (2012).
Kikly, K., Manetta, J., Smith, H., Wierda, D. & Witcher, D. Characterization of LY2127399, a neutralizing antibody for BAFF [abstract]. Arthritis Rheum. 60 (Suppl. 10), 693 (2009).
Gandhi, K. S. et al. BAFF is a biological response marker to IFN-β treatment in multiple sclerosis. J. Interferon Cytokine Res. 28, 529–539 (2008).
Vaknin-Dembinsky, A., Brill, L., Orpaz, N., Abramsky, O. & Karussis, D. Preferential increase of B-cell activating factor in the cerebrospinal fluid of neuromyelitis optica in a white population. Mult. Scler. 16, 1453–1457 (2010).
Hedegaard, C. J. et al. Interferon-beta increases systemic BAFF levels in multiple sclerosis without increasing autoantibody production. Mult. Scler. 17, 567–577 (2011).
Sellebjerg, F. et al. Glatiramer acetate antibodies, gene expression and disease activity in multiple sclerosis. Mult. Scler. 18, 305–313 (2012).
Farina, C. et al. Treatment with glatiramer acetate induces specific IgG4 antibodies in multiple sclerosis patients. J. Neuroimmunol. 123, 188–192 (2002).
Krumbholz, M. et al. BAFF is elevated in serum of patients with Wegener's granulomatosis. J. Autoimmun. 25, 298–302 (2005).
Acknowledgements
The authors' work is supported by the Deutsche Forschungsgemeinschaft (CRC TR 128), the Bundesministerium für Bildung und Forschung (“Krankheitsbezogenes Kompetenznetz Multiple Sklerose”), the Gemeinnützige-Hertie Stiftung, and the Verein zur Therapieforschung für MS Kranke.
Author information
Authors and Affiliations
Contributions
All authors contributed to researching data for the article, discussion of the content, writing, and review and/or editing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
M. Krumbholz has received grant support and speaker's honoraria from Novartis.
T. Derfuss serves on scientific advisory boards for Novartis, Merck Serono, Mitsubishi Pharma, Biogen Idec, Teva, and Bayer Schering Pharma. He has received funding for travel and/or speaker's honoraria from Biogen Idec, Novartis, Merck Serono, and Bayer Schering Pharma. He receives research support from Biogen Idec, Novartis, and Merck Serono.
R. Hohlfeld has received personal compensations and grant support from Teva, Bayer, Merck-Serono, Sanofi-Aventis, Biogen-Idec, and Novartis.
E. Meinl has received honoraria from TEVA and Novartis, and grant support from Novartis.
Rights and permissions
About this article
Cite this article
Krumbholz, M., Derfuss, T., Hohlfeld, R. et al. B cells and antibodies in multiple sclerosis pathogenesis and therapy. Nat Rev Neurol 8, 613–623 (2012). https://doi.org/10.1038/nrneurol.2012.203
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneurol.2012.203
This article is cited by
-
The Node of Ranvier as an Interface for Axo-Glial Interactions: Perturbation of Axo-Glial Interactions in Various Neurological Disorders
Journal of Neuroimmune Pharmacology (2023)
-
Saliva and Serum Acetylcholinesterase Activity in Multiple Sclerosis
Molecular Neurobiology (2023)
-
Persistent virus-specific and clonally expanded antibody-secreting cells respond to induced self-antigen in the CNS
Acta Neuropathologica (2023)
-
High efficacy and safety of CD38 and BCMA bispecific CAR-T in relapsed or refractory multiple myeloma
Journal of Experimental & Clinical Cancer Research (2022)
-
Unmet Needs and Treatment of Relapsing-Remitting Multiple Sclerosis in Saudi Arabia: Focus on the Role of Ofatumumab
Neurology and Therapy (2022)