Key Points
-
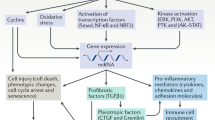
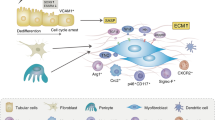
TGF-β acts on multiple cell types to drive fibrosis in progressive kidney disease
-
TGF-β signals through both canonical and non-canonical pathways; TGF-β canonical signalling via Smads has a central role in the development of renal fibrosis
-
The profibrotic actions of TGF-β are positively and negatively regulated by interactions with other signalling pathways and by noncoding RNA and epigenetic mechanisms
-
Direct targeting of TGF-β is unlikely to be therapeutically feasible due to the involvement of TGF-β in other systems, including the immune system
-
Greater understanding of the fibrotic pathways regulated by TGF-β has identified alternative therapeutic targets; re-establishing the balance between profibrotic Smad3 activation and antifibrotic Smad7 action is once such approach
Abstract
Transforming growth factor-β (TGF-β) is the primary factor that drives fibrosis in most, if not all, forms of chronic kidney disease (CKD). Inhibition of the TGF-β isoform, TGF-β1, or its downstream signalling pathways substantially limits renal fibrosis in a wide range of disease models whereas overexpression of TGF-β1 induces renal fibrosis. TGF-β1 can induce renal fibrosis via activation of both canonical (Smad-based) and non-canonical (non-Smad-based) signalling pathways, which result in activation of myofibroblasts, excessive production of extracellular matrix (ECM) and inhibition of ECM degradation. The role of Smad proteins in the regulation of fibrosis is complex, with competing profibrotic and antifibrotic actions (including in the regulation of mesenchymal transitioning), and with complex interplay between TGF-β/Smads and other signalling pathways. Studies over the past 5 years have identified additional mechanisms that regulate the action of TGF-β1/Smad signalling in fibrosis, including short and long noncoding RNA molecules and epigenetic modifications of DNA and histone proteins. Although direct targeting of TGF-β1 is unlikely to yield a viable antifibrotic therapy due to the involvement of TGF-β1 in other processes, greater understanding of the various pathways by which TGF-β1 controls fibrosis has identified alternative targets for the development of novel therapeutics to halt this most damaging process in CKD.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Eddy, A. A. & Neilson, E. G. Chronic kidney disease progression. J. Am. Soc. Nephrol. 17, 2964–2966 (2006).
Bottinger, E. P. & Bitzer, M. TGF-β signaling in renal disease. J. Am. Soc. Nephrol. 13, 2600–2610 (2002).
Eddy, A. A. Overview of the cellular and molecular basis of kidney fibrosis. Kidney Int. Suppl. (2011) 4, 2–8 (2014).
Sharma, K. et al. Increased renal production of transforming growth factor-β1 in patients with type II diabetes. Diabetes 46, 854–859 (1997).
Yamamoto, T., Nakamura, T., Noble, N. A., Ruoslahti, E. & Border, W. A. Expression of transforming growth factor β is elevated in human and experimental diabetic nephropathy. Proc. Natl Acad. Sci. USA 90, 1814–1818 (1993).
Yoshioka, K. et al. Transforming growth factor-beta protein and mRNA in glomeruli in normal and diseased human kidneys. Lab. Invest. 68, 154–163 (1993).
Border, W. A., Okuda, S., Languino, L. R., Sporn, M. B. & Ruoslahti, E. Suppression of experimental glomerulonephritis by antiserum against transforming growth factor β1. Nature 346, 371–374 (1990).
Isaka, Y. et al. Glomerulosclerosis induced by in vivo transfection of transforming growth factor-beta or platelet-derived growth factor gene into the rat kidney. J. Clin. Invest. 92, 2597–2601 (1993).
Moon, J. A., Kim, H. T., Cho, I. S., Sheen, Y. Y. & Kim, D. K. IN-1130, a novel transforming growth factor-β type I receptor kinase (ALK5) inhibitor, suppresses renal fibrosis in obstructive nephropathy. Kidney Int. 70, 1234–1243 (2006).
Russo, L. M., del Re, E., Brown, D. & Lin, H. Y. Evidence for a role of transforming growth factor (TGF)-β1 in the induction of postglomerular albuminuria in diabetic nephropathy: amelioration by soluble TGF-β type II receptor. Diabetes 56, 380–388 (2007).
Sato, M., Muragaki, Y., Saika, S., Roberts, A. B. & Ooshima, A. Targeted disruption of TGF-β1/Smad3 signaling protects against renal tubulointerstitial fibrosis induced by unilateral ureteral obstruction. J. Clin. Invest. 112, 1486–1494 (2003).
Meng, X. M., Chung, A. C. & Lan, H. Y. Role of the TGF-β/BMP-7/Smad pathways in renal diseases. Clin. Sci. (Lond.) 124, 243–254 (2013).
Zhang, Y. E. Non-Smad pathways in TGF-β signaling. Cell Res. 19, 128–139 (2009).
Yu, L., Border, W. A., Huang, Y. & Noble, N. A. TGF-β isoforms in renal fibrogenesis. Kidney Int. 64, 844–856 (2003).
Xu, P., Liu, J. & Derynck, R. Post-translational regulation of TGF-β receptor and Smad signaling. FEBS Lett. 586, 1871–1884 (2012).
Sanderson, N. et al. Hepatic expression of mature transforming growth factor β 1 in transgenic mice results in multiple tissue lesions. Proc. Natl Acad. Sci. USA 92, 2572–2576 (1995).
Robertson, I. B. et al. Latent TGF-β-binding proteins. Matrix Biol. 47, 44–53 (2015).
Annes, J. P., Munger, J. S. & Rifkin, D. B. Making sense of latent TGFβ activation. J. Cell Sci. 116, 217–224 (2003).
Probst-Kepper, M., Balling, R. & Buer, J. FOXP3: required but not sufficient. The role of GARP (LRRC32) as a safeguard of the regulatory phenotype. Curr. Mol. Med. 10, 533–539 (2010).
Huang, X. R., Chung, A. C., Wang, X. J., Lai, K. N. & Lan, H. Y. Mice overexpressing latent TGF-β1 are protected against renal fibrosis in obstructive kidney disease. Am. J. Physiol. Renal Physiol. 295, F118–F127 (2008).
Huang, X. R., Chung, A. C., Zhou, L., Wang, X. J. & Lan, H. Y. Latent TGF-β1 protects against crescentic glomerulonephritis. J. Am. Soc. Nephrol. 19, 233–242 (2008).
Border, W. A., Okuda, S., Languino, L. R. & Ruoslahti, E. Transforming growth factor-β regulates production of proteoglycans by mesangial cells. Kidney Int. 37, 689–695 (1990).
Haberstroh, U. et al. TGF-β stimulates rat mesangial cell proliferation in culture: role of PDGF beta-receptor expression. Am. J. Physiol. 264, F199–F205 (1993).
Wilson, H. M. et al. Effect of transforming growth factor-beta 1 on plasminogen activators and plasminogen activator inhibitor-1 in renal glomerular cells. Exp. Nephrol. 1, 343–350 (1993).
Lopez-Hernandez, F. J. & Lopez-Novoa, J. M. Role of TGF-β in chronic kidney disease: an integration of tubular, glomerular and vascular effects. Cell Tissue Res. 347, 141–154 (2012).
Gruden, G., Perin, P. C. & Camussi, G. Insight on the pathogenesis of diabetic nephropathy from the study of podocyte and mesangial cell biology. Curr. Diabetes Rev. 1, 27–40 (2005).
Neelisetty, S. et al. Renal fibrosis is not reduced by blocking transforming growth factor-β signaling in matrix-producing interstitial cells. Kidney Int. 88, 503–514 (2015).
Meng, X. M. et al. Diverse roles of TGF-β receptor II in renal fibrosis and inflammation in vivo and in vitro. J. Pathol. 227, 175–188 (2012).
Gewin, L. et al. TGF-β receptor deletion in the renal collecting system exacerbates fibrosis. J. Am. Soc. Nephrol. 21, 1334–1343 (2010).
Lebrin, F., Deckers, M., Bertolino, P. & Ten Dijke, P. TGF-β receptor function in the endothelium. Cardiovasc. Res. 65, 599–608 (2005).
Das, R. et al. Upregulation of mitochondrial Nox4 mediates TGF-β-induced apoptosis in cultured mouse podocytes. Am. J. Physiol. Renal Physiol. 306, F155–F167 (2014).
Mack, M. & Yanagita, M. Origin of myofibroblasts and cellular events triggering fibrosis. Kidney Int. 87, 297–307 (2015).
Chen, S. J. et al. Stimulation of type I collagen transcription in human skin fibroblasts by TGF-β: involvement of Smad 3. J. Invest. Dermatol. 112, 49–57 (1999).
Yuan, W. & Varga, J. Transforming growth factor-β repression of matrix metalloproteinase-1 in dermal fibroblasts involves Smad3. J. Biol. Chem. 276, 38502–38510 (2001).
Dennler, S. et al. Direct binding of Smad3 and Smad4 to critical TGF β-inducible elements in the promoter of human plasminogen activator inhibitor-type 1 gene. EMBO J. 17, 3091–3100 (1998).
Piek, E. et al. Functional characterization of transforming growth factor β signaling in Smad2- and Smad3-deficient fibroblasts. J. Biol. Chem. 276, 19945–19953 (2001).
Goumans, M. J. & Mummery, C. Functional analysis of the TGFbeta receptor/Smad pathway through gene ablation in mice. Int. J. Dev. Biol. 44, 253–265 (2000).
Inazaki, K. et al. Smad3 deficiency attenuates renal fibrosis, inflammation, and apoptosis after unilateral ureteral obstruction. Kidney Int. 66, 597–604 (2004).
Kim, J. H., Kim, B. K., Moon, K. C., Hong, H. K. & Lee, H. S. Activation of the TGF-β/Smad signaling pathway in focal segmental glomerulosclerosis. Kidney Int. 64, 1715–1721 (2003).
Fujimoto, M. et al. Mice lacking Smad3 are protected against streptozotocin-induced diabetic glomerulopathy. Biochem. Biophys. Res. Commun. 305, 1002–1007 (2003).
Zhou, L. et al. Mechanism of chronic aristolochic acid nephropathy: role of Smad3. Am. J. Physiol. Renal Physiol. 298, F1006–F1017 (2010).
Meng, X. M. et al. Smad2 protects against TGF-β/Smad3-mediated renal fibrosis. J. Am. Soc. Nephrol. 21, 1477–1487 (2010).
Tsuchida, K., Zhu, Y., Siva, S., Dunn, S. R. & Sharma, K. Role of Smad4 on TGF-β-induced extracellular matrix stimulation in mesangial cells. Kidney Int. 63, 2000–2009 (2003).
Meng, X. M. et al. Disruption of Smad4 impairs TGF-β/Smad3 and Smad7 transcriptional regulation during renal inflammation and fibrosis in vivo and in vitro. Kidney Int. 81, 266–279 (2012).
Yan, X. & Chen, Y. G. Smad7: not only a regulator, but also a cross-talk mediator of TGF-β signalling. Biochem. J. 434, 1–10 (2011).
Shi, Y. & Massague, J. Mechanisms of TGF-β signaling from cell membrane to the nucleus. Cell 113, 685–700 (2003).
Fukasawa, H. et al. Down-regulation of Smad7 expression by ubiquitin-dependent degradation contributes to renal fibrosis in obstructive nephropathy in mice. Proc. Natl Acad. Sci. USA 101, 8687–8692 (2004).
Chung, A. C. et al. Disruption of the Smad7 gene promotes renal fibrosis and inflammation in unilateral ureteral obstruction (UUO) in mice. Nephrol. Dial. Transplant. 24, 1443–1454 (2009).
Chen, H. Y. et al. The protective role of Smad7 in diabetic kidney disease: mechanism and therapeutic potential. Diabetes 60, 590–601 (2011).
Liu, G. X. et al. Disruption of Smad7 promotes ANG II-mediated renal inflammation and fibrosis via Sp1-TGF-β/Smad3-NF.κB-dependent mechanisms in mice. PLoS ONE 8, e53573 (2013).
Hou, C. C. et al. Ultrasound-microbubble-mediated gene transfer of inducible Smad7 blocks transforming growth factor-β signaling and fibrosis in rat remnant kidney. Am. J. Pathol. 166, 761–771 (2005).
Ka, S. M. et al. Smad7 gene therapy ameliorates an autoimmune crescentic glomerulonephritis in mice. J. Am. Soc. Nephrol. 18, 1777–1788 (2007).
Ka, S. M. et al. Kidney-targeting Smad7 gene transfer inhibits renal TGF-β/MAD homologue (SMAD) and nuclear factor κB (NF-κB) signalling pathways, and improves diabetic nephropathy in mice. Diabetologia 55, 509–519 (2012).
Liu, G. X. et al. Smad7 inhibits AngII-mediated hypertensive nephropathy in a mouse model of hypertension. Clin. Sci. (Lond.) 127, 195–208 (2014).
Meng, X. M. et al. Treatment of renal fibrosis by rebalancing TGF-β/Smad signaling with the combination of asiatic acid and naringenin. Oncotarget 6, 36984–36997 (2015).
Ma, F. Y., Sachchithananthan, M., Flanc, R. S. & Nikolic-Paterson, D. J. Mitogen activated protein kinases in renal fibrosis. Front. Biosci. (Schol. Ed.) 1, 171–187 (2009).
Adhikary, L. et al. Abnormal p38 mitogen-activated protein kinase signalling in human and experimental diabetic nephropathy. Diabetologia 47, 1210–1222 (2004).
De Borst, M. H. et al. Glomerular and tubular induction of the transcription factor c-Jun in human renal disease. J. Pathol. 213, 219–228 (2007).
Stambe, C., Nikolic-Paterson, D. J., Hill, P. A., Dowling, J. & Atkins, R. C. p38 mitogen-activated protein kinase activation and cell localization in human glomerulonephritis: correlation with renal injury. J. Am. Soc. Nephrol. 15, 326–336 (2004).
Ma, F. Y. et al. A pathogenic role for c-Jun amino-terminal kinase signaling in renal fibrosis and tubular cell apoptosis. J. Am. Soc. Nephrol. 18, 472–484 (2007).
Stambe, C. et al. The role of p38α mitogen-activated protein kinase activation in renal fibrosis. J. Am. Soc. Nephrol. 15, 370–379 (2004).
Ma, F. Y. et al. Blockade of the c-Jun amino terminal kinase prevents crescent formation and halts established anti-GBM glomerulonephritis in the rat. Lab. Invest. 89, 470–484 (2009).
Muller, R. et al. The mitogen-activated protein kinase p38α regulates tubular damage in murine anti-glomerular basement membrane nephritis. PLoS ONE 8, e56316 (2013).
Weigert, C., Brodbeck, K., Klopfer, K., Haring, H. U. & Schleicher, E. D. Angiotensin II induces human TGF-β1 promoter activation: similarity to hyperglycaemia. Diabetologia 45, 890–898 (2002).
Gruden, G. et al. Mechanical stretch-induced fibronectin and transforming growth factor-β1 production in human mesangial cells is p38 mitogen-activated protein kinase-dependent. Diabetes 49, 655–661 (2000).
Ma, F. Y., Tesch, G. H. & Nikolic-Paterson, D. J. ASK1/p38 signaling in renal tubular epithelial cells promotes renal fibrosis in the mouse obstructed kidney. Am. J. Physiol. Renal Physiol. 307, F1263–F1273 (2014).
Chuang, C. T., Guh, J. Y., Lu, C. Y., Chen, H. C. & Chuang, L. Y. S100B is required for high glucose-induced pro-fibrotic gene expression and hypertrophy in mesangial cells. Int. J. Mol. Med. 35, 546–552 (2015).
Naito, T. et al. Angiotensin II induces thrombospondin-1 production in human mesangial cells via p38 MAPK and JNK: a mechanism for activation of latent TGF-β1 . Am. J. Physiol. Renal Physiol. 286, F278–F287 (2004).
Kamato, D. et al. Transforming growth factor-β signalling: role and consequences of Smad linker region phosphorylation. Cell. Signal. 25, 2017–2024 (2013).
Lee, M. K. et al. TGF-β activates Erk MAP kinase signalling through direct phosphorylation of ShcA. EMBO J. 26, 3957–3967 (2007).
Sorrentino, A. et al. The type I TGF-β receptor engages TRAF6 to activate TAK1 in a receptor kinase-independent manner. Nat. Cell Biol. 10, 1199–1207 (2008).
Chin, B. Y., Mohsenin, A., Li, S. X., Choi, A. M. & Choi, M. E. Stimulation of pro-α1(I) collagen by TGF-β1 in mesangial cells: role of the p38 MAPK pathway. Am. J. Physiol. Renal Physiol. 280, F495–F504 (2001).
Mariasegaram, M. et al. Lefty antagonises TGF-β1 induced epithelial–mesenchymal transition in tubular epithelial cells. Biochem. Biophys. Res. Commun. 393, 855–859 (2010).
Bakin, A. V., Rinehart, C., Tomlinson, A. K. & Arteaga, C. L. p38 mitogen-activated protein kinase is required for TGFβ-mediated fibroblastic transdifferentiation and cell migration. J. Cell Sci. 115, 3193–3206 (2002).
Wei, J. et al. AEG-1 participates in TGF-β1-induced EMT through p38 MAPK activation. Cell Biol. Int. 37, 1016–1021 (2013).
Hayashida, T., Decaestecker, M. & Schnaper, H. W. Cross-talk between ERK MAP kinase and Smad signaling pathways enhances TGF-β-dependent responses in human mesangial cells. FASEB J. 17, 1576–1578 (2003).
Tan, R. J., Zhou, D., Zhou, L. & Liu, Y. Wnt/β-catenin signaling and kidney fibrosis. Kidney Int. Suppl. (2011) 4, 84–90 (2014).
Hao, S. et al. Targeted inhibition of β-catenin/CBP signaling ameliorates renal interstitial fibrosis. J. Am. Soc. Nephrol. 22, 1642–1653 (2011).
He, W. et al. Wnt/β-catenin signaling promotes renal interstitial fibrosis. J. Am. Soc. Nephrol. 20, 765–776 (2009).
Poon, R., Nik, S. A., Ahn, J., Slade, L. & Alman, B. A. β-catenin and transforming growth factor β have distinct roles regulating fibroblast cell motility and the induction of collagen lattice contraction. BMC Cell Biol. 10, 38 (2009).
Amini Nik, S., Ebrahim, R. P., Van Dam, K., Cassiman, J. J. & Tejpar, S. TGF-β modulates β-Catenin stability and signaling in mesenchymal proliferations. Exp. Cell Res. 313, 2887–2895 (2007).
Akhmetshina, A. et al. Activation of canonical Wnt signalling is required for TGF-β-mediated fibrosis. Nat. Commun. 3, 735 (2012).
Wang, D., Dai, C., Li, Y. & Liu, Y. Canonical Wnt/β-catenin signaling mediates transforming growth factor-β-driven podocyte injury and proteinuria. Kidney Int. 80, 1159–1169 (2011).
Zhou, B. et al. Interactions between β-catenin and transforming growth factor-β signaling pathways mediate epithelial–mesenchymal transition and are dependent on the transcriptional co-activator cAMP-response element-binding protein (CREB)-binding protein (CBP). J. Biol. Chem. 287, 7026–7038 (2012).
Eger, A. et al. β-catenin and TGFβ signalling cooperate to maintain a mesenchymal phenotype after FosER-induced epithelial to mesenchymal transition. Oncogene 23, 2672–2680 (2004).
Kim, K. K. et al. Epithelial cell α3β1 integrin links β-catenin and Smad signaling to promote myofibroblast formation and pulmonary fibrosis. J. Clin. Invest. 119, 213–224 (2009).
Lam, A. P. & Gottardi, C. J. β-catenin signaling: a novel mediator of fibrosis and potential therapeutic target. Curr. Opin. Rheumatol. 23, 562–567 (2011).
Zeng, F., Singh, A. B. & Harris, R. C. The role of the EGF family of ligands and receptors in renal development, physiology and pathophysiology. Exp. Cell Res. 315, 602–610 (2009).
Liu, N. et al. Genetic or pharmacologic blockade of EGFR inhibits renal fibrosis. J. Am. Soc. Nephrol. 23, 854–867 (2012).
Yang, L., Besschetnova, T. Y., Brooks, C. R., Shah, J. V. & Bonventre, J. V. Epithelial cell cycle arrest in G2/M mediates kidney fibrosis after injury. Nat. Med. 16, 535–543, 1p following 143 (2010).
Chen, J. et al. EGFR signaling promotes TGFβ-dependent renal fibrosis. J. Am. Soc. Nephrol. 23, 215–224 (2012).
Liu, N. et al. EGF receptor inhibition alleviates hyperuricemic nephropathy. J. Am. Soc. Nephrol. 26, 2716–2729 (2015).
Qian, Y. et al. Novel epidermal growth factor receptor inhibitor attenuates angiotensin II-induced kidney fibrosis. J. Pharmacol. Exp. Ther. 356, 32–42 (2016).
Zhuang, S. & Liu, N. EGFR signaling in renal fibrosis. Kidney Int. Suppl. (2011) 4, 70–74 (2014).
Samarakoon, R. et al. Induction of renal fibrotic genes by TGF-β1 requires EGFR activation, 53 and reactive oxygen species. Cell. Signal. 25, 2198–2209 (2013).
Yan, Y. et al. Src inhibition blocks renal interstitial fibroblast activation and ameliorates renal fibrosis. Kidney Int. 89, 68–81 (2015).
Lieberthal, W. & Levine, J. S. Mammalian target of rapamycin and the kidney. I. The signaling pathway. Am. J. Physiol. Renal Physiol. 303, F1–F10 (2012).
Lamming, D. W. et al. Rapamycin-induced insulin resistance is mediated by mTORC2 loss and uncoupled from longevity. Science 335, 1638–1643 (2012).
Lieberthal, W. & Levine, J. S. Mammalian target of rapamycin and the kidney. II. Pathophysiology and therapeutic implications. Am. J. Physiol. Renal Physiol. 303, F180–F191 (2012).
Lieberthal, W. & Levine, J. S. The role of the mammalian target of rapamycin (mTOR) in renal disease. J. Am. Soc. Nephrol. 20, 2493–2502 (2009).
Das, F. et al. Transforming growth factor β integrates Smad 3 to mechanistic target of rapamycin complexes to arrest deptor abundance for glomerular mesangial cell hypertrophy. J. Biol. Chem. 288, 7756–7768 (2013).
Li, J. et al. Rictor/mTORC2 signaling mediates TGFβ1-induced fibroblast activation and kidney fibrosis. Kidney Int. 88, 515–527 (2015).
Cordenonsi, M. et al. Links between tumor suppressors: 53 is required for TGF-β gene responses by cooperating with Smads. Cell 113, 301–314 (2003).
Overstreet, J. M., Samarakoon, R., Meldrum, K. K. & Higgins, P. J. Redox control of p53 in the transcriptional regulation of TGF-β1 target genes through SMAD cooperativity. Cell. Signal. 26, 1427–1436 (2014).
Overstreet, J. M., Samarakoon, R., Cardona-Grau, D., Goldschmeding, R. & Higgins, P. J. Tumor suppressor ataxia telangiectasia mutated functions downstream of TGF-β1 in orchestrating profibrotic responses. FASEB J. 29, 1258–1268 (2015).
Deshpande, S. D. et al. Transforming growth factor-β-induced cross talk between p53 and a microRNA in the pathogenesis of diabetic nephropathy. Diabetes 62, 3151–3162 (2013).
Ghosh, A. K., Bhattacharyya, S. & Varga, J. The tumor suppressor p53 abrogates Smad-dependent collagen gene induction in mesenchymal cells. J. Biol. Chem. 279, 47455–47463 (2004).
Ghosh, A. K., Nagpal, V., Covington, J. W., Michaels, M. A. & Vaughan, D. E. Molecular basis of cardiac endothelial-to-mesenchymal transition (EndMT): differential expression of microRNAs during EndMT. Cell. Signal. 24, 1031–1036 (2012).
Li, R. X., Yiu, W. H. & Tang, S. C. Role of bone morphogenetic protein-7 in renal fibrosis. Front. Physiol. 6, 114 (2015).
Zeisberg, M. et al. BMP-7 counteracts TGF-β1-induced epithelial-to-mesenchymal transition and reverses chronic renal injury. Nat. Med. 9, 964–968 (2003).
Wang, S. N., Lapage, J. & Hirschberg, R. Loss of tubular bone morphogenetic protein-7 in diabetic nephropathy. J. Am. Soc. Nephrol. 12, 2392–2399 (2001).
Simon, M. et al. Expression of bone morphogenetic protein-7 mRNA in normal and ischemic adult rat kidney. Am. J. Physiol. 276, F382–F389 (1999).
Hruska, K. A. et al. Osteogenic protein-1 prevents renal fibrogenesis associated with ureteral obstruction. Am. J. Physiol. Renal Physiol. 279, F130–F143 (2000).
Wang, S. et al. Renal bone morphogenetic protein-7 protects against diabetic nephropathy. J. Am. Soc. Nephrol. 17, 2504–2512 (2006).
Zeisberg, M. et al. Bone morphogenic protein-7 inhibits progression of chronic renal fibrosis associated with two genetic mouse models. Am. J. Physiol. Renal Physiol. 285, F1060–F1067 (2003).
Motazed, R., Colville-Nash, P., Kwan, J. T. & Dockrell, M. E. BMP-7 and proximal tubule epithelial cells: activation of multiple signaling pathways reveals a novel anti-fibrotic mechanism. Pharm. Res. 25, 2440–2446 (2008).
Wang, S. & Hirschberg, R. Bone morphogenetic protein-7 signals opposing transforming growth factor β in mesangial cells. J. Biol. Chem. 279, 23200–23206 (2004).
Veerasamy, M. et al. Differential regulation of E-cadherin and α-smooth muscle actin by BMP 7 in human renal proximal tubule epithelial cells and its implication in renal fibrosis. Am. J. Physiol. Renal Physiol. 297, F1238–F1248 (2009).
Munoz-Felix, J. M., Gonzalez-Nunez, M. & Lopez-Novoa, J. M. ALK1-Smad1/5 signaling pathway in fibrosis development: friend or foe? Cytokine Growth Factor Rev. 24, 523–537 (2013).
Scharpfenecker, M., Floot, B., Russell, N. S., Ten Dijke, P. & Stewart, F. A. Endoglin haploinsufficiency reduces radiation-induced fibrosis and telangiectasia formation in mouse kidneys. Radiother. Oncol. 92, 484–491 (2009).
Munoz-Felix, J. M., Oujo, B. & Lopez-Novoa, J. M. The role of endoglin in kidney fibrosis. Expert Rev. Mol. Med. 16, e18 (2014).
Goligorsky, M. S. Pathogenesis of endothelial cell dysfunction in chronic kidney disease: a retrospective and what the future may hold. Kidney Res. Clin. Pract. 34, 76–82 (2015).
Rodriguez-Pena, A. et al. Endoglin upregulation during experimental renal interstitial fibrosis in mice. Hypertension 40, 713–720 (2002).
Roy-Chaudhury, P., Simpson, J. G. & Power, D. A. Endoglin, a transforming growth factor-beta-binding protein, is upregulated in chronic progressive renal disease. Exp. Nephrol. 5, 55–60 (1997).
Rodriguez-Barbero, A. et al. Endoglin expression in human and rat mesangial cells and its upregulation by TGF-β1. Biochem. Biophys. Res. Commun. 282, 142–147 (2001).
Diez-Marques, L. et al. Expression of endoglin in human mesangial cells: modulation of extracellular matrix synthesis. Biochim. Biophys. Acta 1587, 36–44 (2002).
Iekushi, K. et al. Hepatocyte growth factor attenuates renal fibrosis through TGF-β1 suppression by apoptosis of myofibroblasts. J. Hypertens. 28, 2454–2461 (2010).
Yang, J., Dai, C. & Liu, Y. Hepatocyte growth factor suppresses renal interstitial myofibroblast activation and intercepts Smad signal transduction. Am. J. Pathol. 163, 621–632 (2003).
Mishra, R. et al. AMP-activated protein kinase inhibits transforming growth factor-β-induced Smad3-dependent transcription and myofibroblast transdifferentiation. J. Biol. Chem. 283, 10461–10469 (2008).
Lim, J. Y., Oh, M. A., Kim, W. H., Sohn, H. Y. & Park, S. I. AMP-activated protein kinase inhibits TGF-β-induced fibrogenic responses of hepatic stellate cells by targeting transcriptional coactivator p300. J. Cell. Physiol. 227, 1081–1089 (2012).
Sirin, Y. & Susztak, K. Notch in the kidney: development and disease. J. Pathol. 226, 394–403 (2012).
Zavadil, J., Cermak, L., Soto-Nieves, N. & Bottinger, E. P. Integration of TGF-β/Smad and Jagged1/Notch signalling in epithelial-to-mesenchymal transition. EMBO J. 23, 1155–1165 (2004).
Ueno, T. et al. Aberrant Notch1-dependent effects on glomerular parietal epithelial cells promotes collapsing focal segmental glomerulosclerosis with progressive podocyte loss. Kidney Int. 83, 1065–1075 (2013).
Niimi, H., Pardali, K., Vanlandewijck, M., Heldin, C. H. & Moustakas, A. Notch signaling is necessary for epithelial growth arrest by TGF-β. J. Cell Biol. 176, 695–707 (2007).
Niranjan, T. et al. The Notch pathway in podocytes plays a role in the development of glomerular disease. Nat. Med. 14, 290–298 (2008).
Leung, A. K. The whereabouts of microRNA actions: cytoplasm and beyond. Trends Cell Biol. 25, 601–610 (2015).
Zarjou, A., Yang, S., Abraham, E., Agarwal, A. & Liu, G. Identification of a microRNA signature in renal fibrosis: role of miR-21. Am. J. Physiol. Renal Physiol. 301, F793–F801 (2011).
Rudnicki, M., Beckers, A., Neuwirt, H. & Vandesompele, J. RNA expression signatures and posttranscriptional regulation in diabetic nephropathy. Nephrol. Dial. Transplant. 30 (Suppl. 4), iv35–iv42 (2015).
McClelland, A. D. et al. miR-21 promotes renal fibrosis in diabetic nephropathy by targeting PTEN and SMAD7. Clin. Sci. (Lond.) 129, 1237–1249 (2015).
Zhong, X., Chung, A. C., Chen, H. Y., Meng, X. M. & Lan, H. Y. Smad3-mediated upregulation of miR-21 promotes renal fibrosis. J. Am. Soc. Nephrol. 22, 1668–1681 (2011).
Zhong, X. et al. miR-21 is a key therapeutic target for renal injury in a mouse model of type 2 diabetes. Diabetologia 56, 663–674 (2013).
Chau, B. N. et al. MicroRNA-21 promotes fibrosis of the kidney by silencing metabolic pathways. Sci. Transl. Med. 4, 121ra18 (2012).
Chung, A. C. & Lan, H. Y. MicroRNAs in renal fibrosis. Front. Physiol. 6, 50 (2015).
Yu, F., Guo, Y., Chen, B., Dong, P. & Zheng, J. MicroRNA-17-5p activates hepatic stellate cells through targeting of Smad7. Lab. Invest. 95, 781–789 (2015).
Kato, M. et al. TGF-β activates Akt kinase through a microRNA-dependent amplifying circuit targeting PTEN. Nat. Cell Biol. 11, 881–889 (2009).
Kriegel, A. J. et al. MicroRNA-target pairs in human renal epithelial cells treated with transforming growth factor β1: a novel role of miR-382. Nucleic Acids Res. 38, 8338–8347 (2010).
Li, R. et al. The microRNA miR-433 promotes renal fibrosis by amplifying the TGF-β/Smad3-Azin1 pathway. Kidney Int. 84, 1129–1144 (2013).
Park, J. T. et al. Repression of let-7 by transforming growth factor-β1-induced Lin28 upregulates collagen expression in glomerular mesangial cells under diabetic conditions. Am. J. Physiol. Renal Physiol. 307, F1390–F1403 (2014).
Wang, B. et al. Transforming growth factor-β1-mediated renal fibrosis is dependent on the regulation of transforming growth factor receptor 1 expression by let-7b. Kidney Int. 85, 352–361 (2014).
Tijsen, A. J. et al. The microRNA-15 family inhibits the TGFβ-pathway in the heart. Cardiovasc. Res. 104, 61–71 (2014).
Lakner, A. M. et al. Inhibitory effects of microRNA 19b in hepatic stellate cell-mediated fibrogenesis. Hepatology 56, 300–310 (2012).
Liang, H. et al. The antifibrotic effects and mechanisms of microRNA-26a action in idiopathic pulmonary fibrosis. Mol. Ther. 22, 1122–1133 (2014).
Tu, X. et al. MicroRNA-101 suppresses liver fibrosis by targeting the TGFβ signalling pathway. J. Pathol. 234, 46–59 (2014).
Pan, Z. et al. MicroRNA-101 inhibited postinfarct cardiac fibrosis and improved left ventricular compliance via the FBJ osteosarcoma oncogene/transforming growth factor-β1 pathway. Circulation 126, 840–850 (2012).
Wang, B. et al. Suppression of microRNA-29 expression by TGF-β1 promotes collagen expression and renal fibrosis. J. Am. Soc. Nephrol. 23, 252–265 (2012).
Bowen, T., Jenkins, R. H. & Fraser, D. J. MicroRNAs, transforming growth factor beta-1, and tissue fibrosis. J. Pathol. 229, 274–285 (2013).
Qin, W. et al. TGF-β/Smad3 signaling promotes renal fibrosis by inhibiting miR-29. J. Am. Soc. Nephrol. 22, 1462–1474 (2011).
Yu, J. W. et al. MicroRNA-29b inhibits peritoneal fibrosis in a mouse model of peritoneal dialysis. Lab. Invest. 94, 978–990 (2014).
Lv, L. L. et al. MicroRNA-29c in urinary exosome/microvesicle as a biomarker of renal fibrosis. Am. J. Physiol. Renal Physiol. 305, F1220–F1227 (2013).
Rubattu, S. et al. Pathogenesis of target organ damage in hypertension: role of mitochondrial oxidative stress. Int. J. Mol. Sci. 16, 823–839 (2015).
Jiang, L. et al. A microRNA-30e/mitochondrial uncoupling protein 2 axis mediates TGF-β-induced tubular epithelial cell extracellular matrix production and kidney fibrosis. Kidney Int. 84, 285–296 (2013).
Wu, J. et al. Downregulation of microRNA-30 facilitates podocyte injury and is prevented by glucocorticoids. J. Am. Soc. Nephrol. 25, 92–104 (2014).
Zhao, N. et al. MicroRNA miR145 regulates TGFBR2 expression and matrix synthesis in vascular smooth muscle cells. Circ. Res. 116, 23–34 (2015).
Yang, S. et al. miR-145 regulates myofibroblast differentiation and lung fibrosis. FASEB J. 27, 2382–2391 (2013).
Putta, S. et al. Inhibiting microRNA-192 ameliorates renal fibrosis in diabetic nephropathy. J. Am. Soc. Nephrol. 23, 458–469 (2012).
Chung, A. C., Huang, X. R., Meng, X. & Lan, H. Y. miR-192 mediates TGF-β/Smad3-driven renal fibrosis. J. Am. Soc. Nephrol. 21, 1317–1325 (2010).
Kato, M. et al. MicroRNA-192 in diabetic kidney glomeruli and its function in TGF-β-induced collagen expression via inhibition of E-box repressors. Proc. Natl Acad. Sci. USA 104, 3432–3437 (2007).
Krupa, A. et al. Loss of microRNA-192 promotes fibrogenesis in diabetic nephropathy. J. Am. Soc. Nephrol. 21, 438–447 (2010).
Wang, B. et al. E-cadherin expression is regulated by miR-192/215 by a mechanism that is independent of the profibrotic effects of transforming growth factor-β. Diabetes 59, 1794–1802 (2010).
Oba, S. et al. miR-200b precursor can ameliorate renal tubulointerstitial fibrosis. PLoS ONE 5, e13614 (2010).
Xiong, M. et al. The miR-200 family regulates TGF-β-induced renal tubular epithelial to mesenchymal transition through Smad pathway by targeting ZEB1 and ZEB2 expression. Am. J. Physiol. Renal Physiol. 302, F369–F379 (2012).
Kato, M. et al. A microRNA circuit mediates transforming growth factor-β1 autoregulation in renal glomerular mesangial cells. Kidney Int. 80, 358–368 (2011).
US National Library of Science. ClinicalTrials.gov [online], https://clinicaltrials.gov/ct2/show/results/NCT02136862 (2016).
US National Library of Science. ClinicalTrials.gov [online], https://clinicaltrials.gov/ct2/show/NCT02603224 (2016).
Mercer, T. R. & Mattick, J. S. Structure and function of long noncoding RNAs in epigenetic regulation. Nat. Struct. Mol. Biol. 20, 300–307 (2013).
Zhou, Q. et al. Identification of novel long noncoding RNAs associated with TGF-β/Smad3-mediated renal inflammation and fibrosis by RNA sequencing. Am. J. Pathol. 184, 409–417 (2014).
He, Y. et al. Inhibitory effects of long noncoding RNA MEG3 on hepatic stellate cells activation and liver fibrogenesis. Biochim. Biophys. Acta 1842, 2204–2215 (2014).
Tampe, B. & Zeisberg, M. Evidence for the involvement of epigenetics in the progression of renal fibrogenesis. Nephrol. Dial. Transplant. 29 (Suppl. 1), i1–i8 (2014).
Bomsztyk, K. & Denisenko, O. Epigenetic alterations in acute kidney injury. Semin. Nephrol. 33, 327–340 (2013).
Zeisberg, E. M. & Zeisberg, M. The role of promoter hypermethylation in fibroblast activation and fibrogenesis. J. Pathol. 229, 264–273 (2013).
Bechtel, W. et al. Methylation determines fibroblast activation and fibrogenesis in the kidney. Nat. Med. 16, 544–550 (2010).
Tampe, B. et al. Tet3-mediated hydroxymethylation of epigenetically silenced genes contributes to bone morphogenic protein 7-induced reversal of kidney fibrosis. J. Am. Soc. Nephrol. 25, 905–912 (2014).
Xiao, X., Tang, W., Yuan, Q., Peng, L. & Yu, P. Epigenetic repression of Kruppel-like factor 4 through Dnmt1 contributes to EMT in renal fibrosis. Int. J. Mol. Med. 35, 1596–1602 (2015).
Bian, E. B. et al. Repression of Smad7 mediated by DNMT1 determines hepatic stellate cell activation and liver fibrosis in rats. Toxicol. Lett. 224, 175–185 (2014).
Pan, X., Chen, Z., Huang, R., Yao, Y. & Ma, G. Transforming growth factor β1 induces the expression of collagen type I by DNA methylation in cardiac fibroblasts. PLoS ONE 8, e60335 (2013).
Zhou, V. W., Goren, A. & Bernstein, B. E. Charting histone modifications and the functional organization of mammalian genomes. Nat. Rev. Genet. 12, 7–18 (2011).
Kato, M. & Natarajan, R. Diabetic nephropathy — emerging epigenetic mechanisms. Nat. Rev. Nephrol. 10, 517–530 (2014).
Sun, G. et al. Epigenetic histone methylation modulates fibrotic gene expression. J. Am. Soc. Nephrol. 21, 2069–2080 (2010).
Berger, S. L. Histone modifications in transcriptional regulation. Curr. Opin. Genet. Dev. 12, 142–148 (2002).
Yuan, H. et al. Involvement of p300/CBP and epigenetic histone acetylation in TGF-β1-mediated gene transcription in mesangial cells. Am. J. Physiol. Renal Physiol. 304, F601–F613 (2013).
Inoue, Y. et al. Smad3 is acetylated by p300/CBP to regulate its transactivation activity. Oncogene 26, 500–508 (2007).
Li, J., Qu, X., Ricardo, S. D., Bertram, J. F. & Nikolic-Paterson, D. J. Resveratrol inhibits renal fibrosis in the obstructed kidney: potential role in deacetylation of Smad3. Am. J. Pathol. 177, 1065–1071 (2010).
Noh, H. et al. Histone deacetylase-2 is a key regulator of diabetes- and transforming growth factor-β1-induced renal injury. Am. J. Physiol. Renal Physiol. 297, F729–F739 (2009).
Liu, N. et al. Blocking the class I histone deacetylase ameliorates renal fibrosis and inhibits renal fibroblast activation via modulating TGF-β and EGFR signaling. PLoS ONE 8, e54001 (2013).
Choi, S. Y. et al. Tubastatin A suppresses renal fibrosis via regulation of epigenetic histone modification and Smad3-dependent fibrotic genes. Vascul. Pharmacol. 72, 130–140 (2015).
Yoshikawa, M., Hishikawa, K., Marumo, T. & Fujita, T. Inhibition of histone deacetylase activity suppresses epithelial-to-mesenchymal transition induced by TGF-β1 in human renal epithelial cells. J. Am. Soc. Nephrol. 18, 58–65 (2007).
Palumbo-Zerr, K. et al. Orphan nuclear receptor NR4A1 regulates transforming growth factor-β signaling and fibrosis. Nat. Med. 21, 150–158 (2015).
Rius, M. & Lyko, F. Epigenetic cancer therapy: rationales, targets and drugs. Oncogene 31, 4257–4265 (2012).
Huang, X. Z. et al. Sirt1 activation ameliorates renal fibrosis by inhibiting the TGF-β/Smad3 pathway. J. Cell. Biochem. 115, 996–1005 (2014).
Ponnusamy, M. et al. Activation of Sirtuin-1 promotes renal fibroblast activation and aggravates renal fibrogenesis. J. Pharmacol. Exp. Ther. 354, 142–151 (2015).
Zerr, P. et al. Sirt1 regulates canonical TGF-β signalling to control fibroblast activation and tissue fibrosis. Ann. Rheum. Dis. 75, 226–233 (2014).
Falke, L. L., Gholizadeh, S., Goldschmeding, R., Kok, R. J. & Nguyen, T. Q. Diverse origins of the myofibroblast-implications for kidney fibrosis. Nat. Rev. Nephrol. 11, 233–244 (2015).
Duffield, J. S., Lupher, M., Thannickal, V. J. & Wynn, T. A. Host responses in tissue repair and fibrosis. Annu. Rev. Pathol. 8, 241–276 (2013).
Strutz, F. et al. Identification and characterization of a fibroblast marker: FSP1. J. Cell Biol. 130, 393–405 (1995).
Jinde, K. et al. Tubular phenotypic change in progressive tubulointerstitial fibrosis in human glomerulonephritis. Am. J. Kidney Dis. 38, 761–769 (2001).
Oldfield, M. D. et al. Advanced glycation end products cause epithelial–myofibroblast transdifferentiation via the receptor for advanced glycation end products (RAGE). J. Clin. Invest. 108, 1853–1863 (2001).
Ng, Y. Y. et al. Glomerular epithelial–myofibroblast transdifferentiation in the evolution of glomerular crescent formation. Nephrol. Dial. Transplant. 14, 2860–2872 (1999).
Ng, Y. Y. et al. Tubular epithelial–myofibroblast transdifferentiation in progressive tubulointerstitial fibrosis in 5/6 nephrectomized rats. Kidney Int. 54, 864–876 (1998).
Yamaguchi, Y. et al. Epithelial–mesenchymal transition as a potential explanation for podocyte depletion in diabetic nephropathy. Am. J. Kidney Dis. 54, 653–664 (2009).
Fan, J. M. et al. Transforming growth factor-β regulates tubular epithelial–myofibroblast transdifferentiation in vitro. Kidney Int. 56, 1455–1467 (1999).
Lamouille, S., Xu, J. & Derynck, R. Molecular mechanisms of epithelial–mesenchymal transition. Nat. Rev. Mol. Cell Biol. 15, 178–196 (2014).
Bhowmick, N. A. et al. Transforming growth factor-β1 mediates epithelial to mesenchymal transdifferentiation through a RhoA-dependent mechanism. Mol. Biol. Cell 12, 27–36 (2001).
Li, J. H. et al. Advanced glycation end products induce tubular epithelial–myofibroblast transition through the RAGE-ERK1/2 MAP kinase signaling pathway. Am. J. Pathol. 164, 1389–1397 (2004).
Grgic, I., Duffield, J. S. & Humphreys, B. D. The origin of interstitial myofibroblasts in chronic kidney disease. Pediatr. Nephrol. 27, 183–193 (2012).
LeBleu, V. S. et al. Origin and function of myofibroblasts in kidney fibrosis. Nat. Med. 19, 1047–1053 (2013).
Grande, M. T. et al. Snail1-induced partial epithelial-to-mesenchymal transition drives renal fibrosis in mice and can be targeted to reverse established disease. Nat. Med. 21, 989–997 (2015).
Lovisa, S. et al. Epithelial-to-mesenchymal transition induces cell cycle arrest and parenchymal damage in renal fibrosis. Nat. Med. 21, 998–1009 (2015).
Piera-Velazquez, S., Li, Z. & Jimenez, S. A. Role of endothelial–mesenchymal transition (EndoMT) in the pathogenesis of fibrotic disorders. Am. J. Pathol. 179, 1074–1080 (2011).
van Meeteren, L. A. & ten Dijke, P. Regulation of endothelial cell plasticity by TGF-β. Cell Tissue Res. 347, 177–186 (2012).
Xavier, S. et al. Curtailing endothelial TGF-β signaling is sufficient to reduce endothelial–mesenchymal transition and fibrosis in CKD. J. Am. Soc. Nephrol. 26, 817–829 (2015).
Li, J. et al. Blockade of endothelial–mesenchymal transition by a Smad3 inhibitor delays the early development of streptozotocin-induced diabetic nephropathy. Diabetes 59, 2612–2624 (2010).
Shi, S. et al. Interactions of DPP-4 and integrin β1 influences endothelial-to-mesenchymal transition. Kidney Int. 88, 479–489 (2015).
Bucala, R., Spiegel, L. A., Chesney, J., Hogan, M. & Cerami, A. Circulating fibrocytes define a new leukocyte subpopulation that mediates tissue repair. Mol. Med. 1, 71–81 (1994).
Reich, B. et al. Fibrocytes develop outside the kidney but contribute to renal fibrosis in a mouse model. Kidney Int. 84, 78–89 (2013).
Strieter, R. M., Keeley, E. C., Hughes, M. A., Burdick, M. D. & Mehrad, B. The role of circulating mesenchymal progenitor cells (fibrocytes) in the pathogenesis of pulmonary fibrosis. J. Leukoc. Biol. 86, 1111–1118 (2009).
Wada, T., Sakai, N., Matsushima, K. & Kaneko, S. Fibrocytes: a new insight into kidney fibrosis. Kidney Int. 72, 269–273 (2007).
Chen, G. et al. CXCL16 recruits bone marrow-derived fibroblast precursors in renal fibrosis. J. Am. Soc. Nephrol. 22, 1876–1886 (2011).
Xia, Y., Yan, J., Jin, X., Entman, M. L. & Wang, Y. The chemokine receptor CXCR6 contributes to recruitment of bone marrow-derived fibroblast precursors in renal fibrosis. Kidney Int. 86, 327–337 (2014).
Hong, K. M., Belperio, J. A., Keane, M. P., Burdick, M. D. & Strieter, R. M. Differentiation of human circulating fibrocytes as mediated by transforming growth factor-β and peroxisome proliferator-activated receptor γ. J. Biol. Chem. 282, 22910–22920 (2007).
Nikolic-Paterson, D. J., Wang, S. & Lan, H. Y. Macrophages promote renal fibrosis through direct and indirect mechanisms. Kidney Int. Suppl. (2011) 4, 34–38 (2014).
Huen, S. C., Moeckel, G. W. & Cantley, L. G. Macrophage-specific deletion of transforming growth factor-β1 does not prevent renal fibrosis after severe ischemia-reperfusion or obstructive injury. Am. J. Physiol. Renal Physiol. 305, F477–F484 (2013).
Bertrand, S., Godoy, M., Semal, P. & Van Gansen, P. Transdifferentiation of macrophages into fibroblasts as a result of Schistosoma mansoni infection. Int. J. Dev. Biol. 36, 179–184 (1992).
Pilling, D. & Gomer, R. H. Differentiation of circulating monocytes into fibroblast-like cells. Methods Mol. Biol. 904, 191–206 (2012).
Wang, S. et al. TGF-β/Smad3 signalling regulates the transition of bone marrow-derived macrophages into myofibroblasts during tissue fibrosis. Oncotarget http://dx.doi.org/10.18632/oncotarget.6604 (2015).
Schnaper, H. W., Hayashida, T., Hubchak, S. C. & Poncelet, A. C. TGF-β signal transduction and mesangial cell fibrogenesis. Am. J. Physiol. Renal Physiol. 284, F243–F252 (2003).
Barnes, J. L. & Gorin, Y. Myofibroblast differentiation during fibrosis: role of NAD(P)H oxidases. Kidney Int. 79, 944–956 (2011).
Manickam, N., Patel, M., Griendling, K. K., Gorin, Y. & Barnes, J. L. RhoA/Rho kinase mediates TGF-β1-induced kidney myofibroblast activation through Poldip2/Nox4-derived reactive oxygen species. Am. J. Physiol. Renal Physiol. 307, F159–F171 (2014).
Grande, M. T. & Lopez-Novoa, J. M. Fibroblast activation and myofibroblast generation in obstructive nephropathy. Nat. Rev. Nephrol. 5, 319–328 (2009).
Wu, C. F. et al. Transforming growth factor β-1 stimulates profibrotic epithelial signaling to activate pericyte–myofibroblast transition in obstructive kidney fibrosis. Am. J. Pathol. 182, 118–131 (2013).
Trachtman, H. et al. A phase 1, single-dose study of fresolimumab, an anti-TGF-β antibody, in treatment-resistant primary focal segmental glomerulosclerosis. Kidney Int. 79, 1236–1243 (2011).
US National Library of Science. ClinicalTrials.gov [online], https://clinicaltrials.gov/ct2/show/NCT01113801 (2015).
Rice, L. M. et al. Fresolimumab treatment decreases biomarkers and improves clinical symptoms in systemic sclerosis patients. J. Clin. Invest. 125, 2795–2807 (2015).
Meng, X. M., Tang, P. M., Li, J. & Lan, H. Y. TGF-β/Smad signaling in renal fibrosis. Front. Physiol. 6, 82 (2015).
Rodon, J. et al. First-in-human dose study of the novel transforming growth factor-beta receptor I kinase inhibitor LY2157299 monohydrate in patients with advanced cancer and glioma. Clin. Cancer Res. 21, 553–560 (2015).
Liu, Y. Hepatocyte growth factor in kidney fibrosis: therapeutic potential and mechanisms of action. Am. J. Physiol. Renal Physiol. 287, F7–F16 (2004).
Doi, S. et al. Klotho inhibits transforming growth factor-β1 (TGF-β1) signaling and suppresses renal fibrosis and cancer metastasis in mice. J. Biol. Chem. 286, 8655–8665 (2011).
Sharma, K. et al. Pirfenidone for diabetic nephropathy. J. Am. Soc. Nephrol. 22, 1144–1151 (2011).
Cho, M. E., Smith, D. C., Branton, M. H., Penzak, S. R. & Kopp, J. B. Pirfenidone slows renal function decline in patients with focal segmental glomerulosclerosis. Clin. J. Am. Soc. Nephrol. 2, 906–913 (2007).
Shihab, F. S., Bennett, W. M., Yi, H. & Andoh, T. F. Pirfenidone treatment decreases transforming growth factor-β1 and matrix proteins and ameliorates fibrosis in chronic cyclosporine nephrotoxicity. Am. J. Transplant. 2, 111–119 (2002).
Ai, J. et al. GQ5 hinders renal fibrosis in obstructive nephropathy by selectively inhibiting TGF-β-induced Smad3 phosphorylation. J. Am. Soc. Nephrol. 26, 1827–1838 (2015).
Schiffer, M. et al. Apoptosis in podocytes induced by TGF-β and Smad7. J. Clin. Invest. 108, 807–816 (2001).
Acknowledgements
We apologize to all colleagues whose important findings could not be cited owing to space limitations. The authors' work described in this Review was supported by a Major State Basic Research Development Program of China (973 program, No. 2012CB517705), the Research Grants Council of Hong Kong (GRF 468711, CUHK3/CRF/12R), the Focused Investment Scheme A from Chinese University of Hong Kong, and the National Natural Science Foundation of China (No. 81300580, No. 81570623). D.J.N.-P. is supported by a Senior Research Fellowship from the National Health and Medical Research Council of Australia.
Author information
Authors and Affiliations
Contributions
All authors discussed the article's content. X.-M.M. wrote the manuscript, and D.J.N.-P. and H.Y.L. reviewed and edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
D.J.N.-P. has worked as a consultant and received research funding from Celgene and Gilead Sciences, and is a co-inventor on a patent for a Smad3 inhibitor (Australian Application number 2015900903). X.-M.M. and H.Y.L. declare no competing interests.
Glossary
- CpG islands
-
Short DNA sequences that contain an atypically high frequency of cytosine–phosphate–guanine (CpG) sites; such sites generally lack DNA (cysteine) methylation and associate with the majority of annotated gene promoters.
- CpG shores
-
Regions of comparatively low CpG density that flank traditional CpG islands.
Rights and permissions
About this article
Cite this article
Meng, Xm., Nikolic-Paterson, D. & Lan, H. TGF-β: the master regulator of fibrosis. Nat Rev Nephrol 12, 325–338 (2016). https://doi.org/10.1038/nrneph.2016.48
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneph.2016.48
This article is cited by
-
Metformin and adipose-derived stem cell combination therapy alleviates radiation-induced skin fibrosis in mice
Stem Cell Research & Therapy (2024)
-
Sulforaphane (Sul) reduces renal interstitial fibrosis (RIF) by controlling the inflammation and TGF-β/Smad signaling pathway
Applied Biological Chemistry (2024)
-
The occurrence and development mechanisms of esophageal stricture: state of the art review
Journal of Translational Medicine (2024)
-
LATS2 degradation promoted fibrosis damage and rescued by vitamin K3 in lupus nephritis
Arthritis Research & Therapy (2024)
-
The disruptor of telomeric silencing 1-like (DOT1L) promotes peritoneal fibrosis through the upregulation and activation of protein tyrosine kinases
Molecular Biomedicine (2024)