Key Points
-
High phosphate concentrations are associated with an increased incidence of cardiovascular complications and mortality in the general population and in patients with chronic kidney disease (CKD)
-
Serum phosphate concentrations are influenced by circadian rhythm and diet
-
The predictive value of a single serum phosphate measurement for cardiovascular risk is greatest for fasting morning concentrations
-
Fibroblast growth factor 23 is a key hormone involved in phosphate homeostasis and an independent predictor of cardiovascular events with a potential causal role in the development of these events
-
Frequently neglected sources of phosphate are bone in patients with CKD and food additives in all populations
-
Definitive proof of a clinical benefit of dietary or pharmacological interventions that lower phosphate exposure is lacking, mainly owing to a paucity of well-designed clinical trials
Abstract
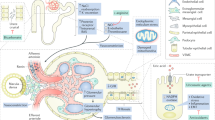
The importance of phosphate homeostasis in chronic kidney disease (CKD) has been recognized for decades, but novel insights — which are frequently relevant to everyday clinical practice — continue to emerge. Epidemiological data consistently indicate an association between hyperphosphataemia and poor clinical outcomes. Moreover, compelling evidence suggests direct toxicity of increased phosphate concentrations. Importantly, serum phosphate concentration has a circadian rhythm that must be considered when interpreting patient phosphate levels. Detailed understanding of dietary sources of phosphate, including food additives, can enable phosphate restriction without risking protein malnutrition. Dietary counselling provides an often underestimated opportunity to target the increasing exposure to dietary phosphate of both the general population and patients with CKD. In patients with secondary hyperparathyroidism, bone can be an important source of serum phosphate, and adequate appreciation of this fact should impact treatment. Dietary and pharmotherapeutic interventions are efficacious strategies to lower phosphate intake and serum concentration. However, strong evidence that targeting serum phosphate improves patient outcomes is currently lacking. Future studies are, therefore, required to investigate the effects of modern dietary and pharmacological interventions on clinically meaningful end points.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Berndt, T. & Kumar, R. Novel mechanisms in the regulation of phosphorus homeostasis. Physiology (Bethesda) 24, 17–25 (2009).
Raggi, P. et al. The ADVANCE study: a randomized study to evaluate the effects of cinacalcet plus low-dose vitamin D on vascular calcification in patients on hemodialysis. Nephrol. Dial. Transplant. 26, 1327–1339 (2011).
Pulskens, W. P. et al. Deregulated renal calcium and phosphate transport during experimental kidney failure. PLoS ONE 10, e0142510 (2015).
Jubiz, W., Canterbury, J. M., Reiss E. & Tyler, F. H. Circadian rhythm in serum parathyroid hormone concentration in human subjects: correlation with serum calcium, phosphate, albumin, and growth hormone levels. J. Clin. Invest. 51, 2040–2046 (1972).
Markowitz, M., Rotkin, L. & Rosen, J. F. Circadian rhythms of blood minerals in humans. Science 213, 672–674 (1981).
Portale, A. A., Halloran, B. P. & Morris, R. C. Jr. Dietary intake of phosphorus modulates the circadian rhythm in serum concentration of phosphorus. Implications for the renal production of 1,25-dihydroxyvitamin D. J. Clin. Invest. 80, 1147–1154 (1987). This early study describes the relationships between dietary intake of phosphate, circadian rhythm and production of 1,25 dihydroxyvitamin D.
Smith, E. R., Cai, M. M., McMahon, L P . & Holt S. G. Biological variability of plasma intact and C-terminal FGF23 measurements. J. Clin. Endocrinol. Metab. 97, 3357–3365 (2012).
Kremsdorf, R. A. et al. Effects of a high-protein diet on regulation of phosphorus homeostasis. J. Clin. Endocrinol. Metab. 98, 1207–1213 (2013).
el-Hajj Fuleihan, G. et al. The parathyroid hormone circadian rhythm is truly endogenous — a general clinical research center study. J. Clin. Endocrinol. Metab. 82, 281–286 (1997).
Trivedi, H. Szabo, A., Zhao, S., Cantor, T. & Raff, H. Circadian variation of mineral and bone parameters in end-stage renal disease. J. Nephrol. 28, 351–359 (2015).
Smith, E. R., McMahon, L. P. & Holt, S. G. Fibroblast growth factor 23. Ann. Clin. Biochem. 51, 203–227 (2014).
Osuka, S. & Razzaque, M. S. Can features of phosphate toxicity appear in normophosphatemia? J. Bone Miner. Metab. 30, 10–18 (2012).
Sullivan, C. M., Leon, J. B. & Sehgal, A. R. Phosphorus-containing food additives and the accuracy of nutrient databases: implications for renal patients. J. Ren. Nutr. 17, 350–354 (2007). This study highlights food additives as an important source of dietary phosphate.
Calvo, M. S., Kumar, R. & Heath, H. Persistently elevated parathyroid hormone secretion and action in young women after four weeks of ingesting high phosphorus, low calcium diets. J. Clin. Endocrinol. Metab. 70, 1334–1340 (1990).
Karp, H. J. Vaihia, K. P., Kärkkäinen, M. U., Niemistö, M. J. & Lamberg-Allardt, C. J. Acute effects of different phosphorus sources on calcium and bone metabolism in young women: a whole-foods approach. Calcif. Tissue Int. 80, 251–258 (2007).
Moe, S. M. et al. Vegetarian compared with meat dietary protein source and phosphorus homeostasis in chronic kidney disease. Clin. J. Am. Soc. Nephrol. 6, 257–264 (2011). This carefully conducted, key study demonstrates the much lower bioavailability of phosphate coming from vegetable sources than from meat sources.
Isakova, T. et al. Daily variability in mineral metabolites in CKD and effects of dietary calcium and calcitriol. Clin. J. Am. Soc. Nephrol. 7, 820–828 (2012).
Micozkadioglu, H. et al. Circadian rhythm of serum phosphate, calcium and parathyroid hormone levels in hemodialysis patients. Clin. Lab. 59, 79–84 (2013).
Ix, J. H. Anderson, C. A., Smits, G., Persky, M. S. & Block, G. A. Effect of dietary phosphate intake on the circadian rhythm of serum phosphate concentrations in chronic kidney disease: a crossover study. Am. J. Clin. Nutr. 100, 1392–1397 (2014).
Ferrari, S. L., Bonjour, J. P. & Rizzoli, R. Fibroblast growth factor-23 relationship to dietary phosphate and renal phosphate handling in healthy young men. J. Clin. Endocrinol. Metab. 90, 1519–1524 (2005).
Vervloet, M. G. et al. Effects of dietary phosphate and calcium intake on fibroblast growth factor-23. Clin. J. Am. Soc. Nephrol. 6, 383–389 (2011).
Dhayat, N. A. et al. Fibroblast growth factor 23 and markers of mineral metabolism in individuals with preserved renal function. Kidney Int. 90, 648–657 (2016).
Weinman, E. J., Light, P. D. & Suki, W. N. Gastrointestinal phosphate handling in CKD and its association with cardiovascular disease. Am. J. Kidney Dis. 62, 1006–1011 (2013).
Selamet, U. et al. Relationship of dietary phosphate intake with risk of end-stage renal disease and mortality in chronic kidney disease stages 3-5: The Modification of Diet in Renal Disease Study. Kidney Int. 89, 176–184 (2015). This post hoc analysis of the landmark MDRD trial undermines the assumption that dietary phosphate restriction improves clinical outcomes in the general population.
Sigrist, M. et al. Responsiveness of FGF-23 and mineral metabolism to altered dietary phosphate intake in chronic kidney disease (CKD): results of a randomized trial. Nephrol. Dial. Transplant. 28, 161–169 (2013).
Karavetian, M. de Vries, N., Rizk, R. & Elzein, H. Dietary educational interventions for management of hyperphosphatemia in hemodialysis patients: a systematic review and meta-analysis. Nutr. Rev. 72, 471–482 (2014).
Boaz, M. & Smetana, S. Regression equation predicts dietary phosphorus intake from estimate of dietary protein intake. J. Am. Diet. Assoc. 96, 1268–1270 (1996).
Kalantar-Zadeh, K. et al. Understanding sources of dietary phosphorus in the treatment of patients with chronic kidney disease. Clin. J. Am. Soc. Nephrol. 5, 519–530 (2010).
Ramirez, J. A. et al. The absorption of dietary phosphorus and calcium in hemodialysis patients. Kidney Int. 30, 753–759 (1986).
Schlemmer, U. Frølich, W., Prieto, R. M. & Grases, F. Phytate in foods and significance for humans: food sources, intake, processing, bioavailability, protective role and analysis. Mol. Nutr. Food Res. 53 (Suppl. 2), S330–S375 (2009).
Gupta, R. K., Gangoliya, S. S. & Singh, N. K. Reduction of phytic acid and enhancement of bioavailable micronutrients in food grains. J. Food Sci. Technol. 52, 676–684 (2015).
Uribarri, J. & Calvo, M. S. Hidden sources of phosphorus in the typical American diet: does it matter in nephrology? Semin. Dial. 16, 186–188 (2003).
McCutcheon, J. Campbell, K., Ferguson, M., Day, S. & Rossi, M. Prevalence of phosphorus-based additives in the Australian food supply: a challenge for dietary education? J. Ren. Nutr. 25, 440–444 (2015).
Calvo, M. S. Dietary considerations to prevent loss of bone and renal function. Nutrition 16, 564–566 (2000).
Leon, J. B., Sullivan, C. M. & Sehgal, A. R. The prevalence of phosphorus-containing food additives in top-selling foods in grocery stores. J. Ren. Nutr. 23, 265–270.e2 (2013).
Moser, M. et al. Phosphorus content of popular beverages. Am. J. Kidney Dis. 65, 969–971 (2015).
Lindley, E. et al. Use of a standard urine assay for measuring the phosphate content of beverages. J. Ren. Nutr. 24, 353–356 (2014).
Benini, O., D'Alessandro, C., Gianfaldoni, D. & Cupisti, A. Extra-phosphate load from food additives in commonly eaten foods: a real and insidious danger for renal patients. J. Ren. Nutr. 21, 303–308 (2011).
Cupisti, A., Benini, O., Ferretti, V., Gianfaldoni, D. & Kalantar-Zadeh, K. Novel differential measurement of natural and added phosphorus in cooked ham with or without preservatives. J. Ren. Nutr. 22, 533–540 (2012).
Sherman, R. A. & Mehta, O. Dietary phosphorus restriction in dialysis patients: potential impact of processed meat, poultry, and fish products as protein sources. Am. J. Kidney Dis. 54, 18–23 (2009). This paper provides data that could help clinicians to avoid protein malnutrition in their patients when restricting their phosphate intake.
Carrigan, A. et al. Contribution of food additives to sodium and phosphorus content of diets rich in processed foods. J. Ren. Nutr. 24, 13–19.e1 (2014).
Carrero, J. J., Burrowes, J. & Wanner, C. A long road to travel: adherence to dietary recommendations and adequate dietary phosphorus control. J. Ren. Nutr. 26, 133–135 (2016).
Karalis, M. Food and Drug Administration petition on food labeling: an update from the American Dietetic Association and National Kidney Foundation. J. Ren. Nutr. 17, 423–424 (2007).
Sherman, R. A., Ravella, S. & Kapoian, T. A dearth of data: the problem of phosphorus in prescription medications. Kidney Int. 87, 1097–1099 (2015).
Sultana, J. et al. Medication is an additional source of phosphate intake in chronic kidney disease patients. Nutr. Metab. Cardiovasc. Dis. 25, 959–967 (2015).
Streja, E. et al. Hyperphosphatemia is a combined function of high serum PTH and high dietary protein intake in dialysis patients. Kidney Int. Suppl. 3, 462–468 (2013).
Block, G., Do, T. P., Collins, A. J., Cooper, K. C. & Bradbury, B. D. Co-trending of parathyroid hormone and phosphate in patients receiving hemodialysis. Clin. Nephrol. 85, 142–151 (2016). This study in a very large haemodialysis population suggests that PTH-driven phosphate liberation from bone might determine serum phosphate concentration in these patients.
London, G., Coyne, D., Hruska, K., Malluche, H. H. & Martin, K. J. The new kidney disease: improving global outcomes (KDIGO) guidelines — expert clinical focus on bone and vascular calcification. Clin. Nephrol. 74, 423–432 (2010).
Zitt, E. et al. Serum phosphorus reduction in dialysis patients treated with cinacalcet for secondary hyperparathyroidism results mainly from parathyroid hormone reduction. Clin. Kidney J. 6, 287–294 (2013).
Cooper, K., Quarles, D., Kubo, Y., Tomlin, H. & Goodman, W. Relationship between reductions in parathyroid hormone and serum phosphorus during the management of secondary hyperparathyroidism with calcimimetics in hemodialysis patients. Nephron. Clin. Pract. 121, c124–c130 (2012).
Frazao, J. M. et al. Is serum phosphorus control related to parathyroid hormone control in dialysis patients with secondary hyperparathyroidism? BMC Nephrol. 13, 76 (2012).
Tonelli, M. et al. Relation between serum phosphate level and cardiovascular event rate in people with coronary disease. Circulation 112, 2627–2633 (2005).
Vart, P., Nigatu, Y. T., Jaglan, A., van Zon, S. K. & Shafique, K. Joint effect of hypertension and elevated serum phosphorus on the risk of mortality in National Health and Nutrition Examination Survey-III. J. Am. Heart Assoc. 4, e001706 (2015).
Foley, R. N., Collins, A. J., Ishani, A. & Kalra, P. A. Calcium-phosphate levels and cardiovascular disease in community-dwelling adults: the Atherosclerosis Risk in Communities (ARIC) Study. Am. Heart J. 156, 556–563 (2008).
Dhingra, R. et al. Relations of serum phosphorus and calcium levels to the incidence of cardiovascular disease in the community. Arch. Intern. Med. 167, 879–885 (2007). This study convincingly demonstrates the prognostic value of high serum phosphate levels, even within the normal range in the general population.
Sim, J. J. et al. Phosphorus and risk of renal failure in subjects with normal renal function. Am. J. Med. 126, 311–318 (2013).
O'Seaghdha, C. M., Hwang, S. J., Muntner, P., Melamed, M. L. & Fox, C. S. Serum phosphorus predicts incident chronic kidney disease and end-stage renal disease. Nephrol. Dial. Transplant. 26, 2885–2890 (2011).
Eddington, H. et al. Serum phosphate and mortality in patients with chronic kidney disease. Clin. J. Am. Soc. Nephrol. 5, 2251–2257 (2010).
Menon, V. et al. Relationship of phosphorus and calcium-phosphorus product with mortality in CKD. Am. J. Kidney Dis. 46, 455–463 (2005).
Chang, W. X. et al. The impact of normal range of serum phosphorus on the incidence of end-stage renal disease by a propensity score analysis. PLoS ONE 11, e0154469 (2016).
Voormolen, N. et al. High plasma phosphate as a risk factor for decline in renal function and mortality in pre-dialysis patients. Nephrol. Dial. Transplant. 22, 2909–2916 (2007).
Kestenbaum, B. et al. Serum phosphate levels and mortality risk among people with chronic kidney disease. J. Am. Soc. Nephrol. 16, 520–528 (2005).
Block, G. A., Hulbert-Shearon, T. E., Levin, N. W. & Port, F. K. Association of serum phosphorus and calcium × phosphate product with mortality risk in chronic hemodialysis patients: a national study. Am. J. Kidney Dis. 31, 607–617 (1998).
Slinin, Y., Foley, R. N. & Collins, A. J. Calcium, phosphorus, parathyroid hormone, and cardiovascular disease in hemodialysis patients: the USRDS waves 1, 3, and 4 study. J. Am. Soc. Nephrol. 16, 1788–1793 (2005).
Naves-Diaz, M. et al. Calcium, phosphorus, PTH and death rates in a large sample of dialysis patients from Latin America. The CORES Study. Nephrol. Dial. Transplant. 26, 1938–1947 (2011).
Young, E. W. et al. Predictors and consequences of altered mineral metabolism: the Dialysis Outcomes and Practice Patterns Study. Kidney Int. 67, 1179–1187 (2005).
Ganesh, S. K., Stack, A. G., Levin, N. W., Hulbert-Shearon, T. & Port, F. K. Association of elevated serum PO4, Ca × PO4 product, and parathyroid hormone with cardiac mortality risk in chronic hemodialysis patients. J. Am. Soc. Nephrol. 12, 2131–2138 (2001).
Floege, J. et al. Serum iPTH, calcium and phosphate, and the risk of mortality in a European haemodialysis population. Nephrol. Dial. Transplant. 26, 1948–1955 (2011).
Palmer, S. C. et al. Serum levels of phosphorus, parathyroid hormone, and calcium and risks of death and cardiovascular disease in individuals with chronic kidney disease: a systematic review and meta-analysis. JAMA 305, 1119–1127 (2011). The findings of this meta-analysis indicate that phosphate control should be prioritized over that of calcium and even more so over PTH control when making clinical decisions.
Kalantar-Zadeh, K. et al. Survival predictability of time-varying indicators of bone disease in maintenance hemodialysis patients. Kidney Int. 70, 771–780 (2006).
Covic, A. et al. Systematic review of the evidence underlying the association between mineral metabolism disturbances and risk of all-cause mortality, cardiovascular mortality and cardiovascular events in chronic kidney disease. Nephrol. Dial. Transplant. 24, 1506–1523 (2009).
Fouque, D. et al. Control of mineral metabolism and bone disease in haemodialysis patients: which optimal targets? Nephrol. Dial. Transplant. 28, 360–367 (2013).
Crouthamel, M. H. et al. Sodium-dependent phosphate cotransporters and phosphate-induced calcification of vascular smooth muscle cells: redundant roles for PiT-1 and PiT-2. Arterioscler. Thromb. Vasc. Biol. 33, 2625–2632 (2013).
Rahabi-Layachi, H. Ourouda, R., Boullier, A., Massy, Z. A. & Amant, C. Distinct effects of inorganic phosphate on cell cycle and apoptosis in human vascular smooth muscle cells. J. Cell. Physiol. 230, 347–355 (2015).
Di Marco, G. S. et al. Increased inorganic phosphate induces human endothelial cell apoptosis in vitro. Am. J. Physiol. Renal Physiol. 294, F1381–F1387 (2008).
Di Marco, G. S. et al. High phosphate directly affects endothelial function by downregulating annexin II. Kidney Int. 83, 213–222 (2013).
Six, I. et al. Effects of phosphate on vascular function under normal conditions and influence of the uraemic state. Cardiovasc. Res. 96, 130–139 (2012). This experimental study provides strong evidence of a direct toxic effect of phosphate.
Mozar, A. et al. High extracellular inorganic phosphate concentration inhibits RANK–RANKL signaling in osteoclast-like cells. J. Cell. Physiol. 215, 47–54 (2008).
Ciceri, P. et al. A new in vitro model to delay high phosphate-induced vascular calcification progression. Mol. Cell. Biochem. 410, 197–206 (2015).
Rangrez, A. Y. et al. Inorganic phosphate accelerates the migration of vascular smooth muscle cells: evidence for the involvement of miR-223. PLoS ONE 7, e47807 (2012).
M'Baya-Moutoula, E., Louvet, L., Metzinger-Le Meuth, V., Massy, Z. A. & Metzinger, L. High inorganic phosphate concentration inhibits osteoclastogenesis by modulating miR-223. Biochim. Biophys. Acta 1852, 2202–2212 (2015).
Graciolli, F. G. et al. Phosphorus overload and PTH induce aortic expression of Runx2 in experimental uraemia. Nephrol. Dial. Transplant. 24, 1416–1421 (2009).
Cozzolino, M. et al. Sevelamer hydrochloride attenuates kidney and cardiovascular calcifications in long-term experimental uremia. Kidney Int. 64, 1653–1661 (2003).
Nikolov, I. G. et al. Lanthanum carbonate, like sevelamer–HCl, retards the progression of vascular calcification and atherosclerosis in uremic apolipoprotein E-deficient mice. Nephrol. Dial. Transplant. 27, 505–513 (2012).
Maizel, J. et al. Effects of sevelamer treatment on cardiovascular abnormalities in mice with chronic renal failure. Kidney Int. 84, 491–500 (2013).
Shuto, E. et al. Dietary phosphorus acutely impairs endothelial function. J. Am. Soc. Nephrol. 20, 1504–1512 (2009).
Levin, A. et al. Prevalence of abnormal serum vitamin D, PTH, calcium, and phosphorus in patients with chronic kidney disease: results of the study to evaluate early kidney disease. Kidney Int. 71, 31–38 (2007).
Kooman, J. P., Kotanko, P., Schols, A. M., Shiels, P. G. & Stenvinkel, P. Chronic kidney disease and premature ageing. Nat. Rev. Nephrol. 10, 732–742 (2014).
Vervloet, M. G. et al. Bone: a new endocrine organ at the heart of chronic kidney disease and mineral and bone disorders. Lancet Diabetes Endocrinol. 2, 427–436 (2014).
Bover, J. et al. Pro: cardiovascular calcifications are clinically relevant. Nephrol. Dial. Transplant. 30, 345–351 (2015).
Isakova, T. et al. Fibroblast growth factor 23 is elevated before parathyroid hormone and phosphate in chronic kidney disease. Kidney Int. 79, 1370–1378 (2011).
Farrow, E. G., Davis, S. I., Summers, L. J. & White, K. E. Initial FGF23-mediated signaling occurs in the distal convoluted tubule. J. Am. Soc. Nephrol. 20, 955–960 (2009).
Hu, M. C. et al. Klotho: a novel phosphaturic substance acting as an autocrine enzyme in the renal proximal tubule. FASEB J. 24, 3438–3450 (2010).
Faul, C. et al. FGF23 induces left ventricular hypertrophy. J. Clin. Invest. 121, 4393–4408 (2011). This landmark study provides compelling evidence for a causal role of FGF-23 in the development of left ventricular hypertrophy.
Grabner, A. et al. Activation of cardiac fibroblast growth factor receptor 4 causes left ventricular hypertrophy. Cell Metab. 22, 1020–1032 (2015).
Shalhoub, V. et al. FGF23 neutralization improves chronic kidney disease-associated hyperparathyroidism yet increases mortality. J. Clin. Invest. 122, 2543–2553 (2012).
Hagel, M. et al. First selective small molecule inhibitor of FGFR4 for the treatment of hepatocellular carcinomas with an activated FGFR4 signaling pathway. Cancer Discov. 5, 424–437 (2015).
Wolf, M. Forging forward with 10 burning questions on FGF23 in kidney disease. J. Am. Soc. Nephrol. 21, 1427–1435 (2010).
Scialla, J. J. & Wolf, M. Roles of phosphate and fibroblast growth factor 23 in cardiovascular disease. Nat. Rev. Nephrol. 10, 268–278 (2014).
Scialla, J. J. et al. Mineral metabolites and CKD progression in African Americans. J. Am. Soc. Nephrol. 24, 125–135 (2013).
Cozzolino, M. et al. Vascular calcification in chronic kidney disease: a changing scenario. J. Nephrol. 24 (Suppl. 18), S3–S10 (2011).
Cozzolino, M., Brancaccio, D., Gallieni, M. & Slatopolsky, E. Pathogenesis of vascular calcification in chronic kidney disease. Kidney Int. 68, 429–436 (2005).
Kidney Disease: Improving Global Outcomes (KDIGO) CKD–MBD Work Group. KDIGO clinical practice guideline for the diagnosis, evaluation, prevention, and treatment of Chronic Kidney Disease–Mineral and Bone Disorder (CKD–MBD). Kidney Int. Suppl. 113, S1–S130 (2009). The clinical practice guideline for CKD–MBD that is currently used worldwide.
Adema, A. Y. et al. Dietary and pharmacological modification of fibroblast growth factor-23 in chronic kidney disease. J. Ren. Nutr. 24, 143–150 (2014).
Penne, E. L. et al. Short-term effects of online hemodiafiltration on phosphate control: a result from the randomized controlled Convective Transport Study (CONTRAST). Am. J. Kidney Dis. 55, 77–87 (2010).
EVOLVE Trial Investigators et al. Effect of cinacalcet on cardiovascular disease in patients undergoing dialysis. N. Engl. J. Med. 367, 2482–2494 (2012). This paper presents the results of the EVOLVE prospective randomized controlled trial, which investigated the effect of cinacalcet on patient-level outcomes in a haemodialysis cohort.
Shinaberger, C. S. et al. Is controlling phosphorus by decreasing dietary protein intake beneficial or harmful in persons with chronic kidney disease? Am. J. Clin. Nutr. 88, 1511–1518 (2008).
Scialla, J. J. et al. Plant protein intake is associated with fibroblast growth factor 23 and serum bicarbonate levels in patients with chronic kidney disease: the Chronic Renal Insufficiency Cohort study. J. Ren. Nutr. 22, 379–388.e1 (2012).
Sullivan, C. et al. Effect of food additives on hyperphosphatemia among patients with end-stage renal disease: a randomized controlled trial. JAMA 301, 629–635 (2009).
Mayne, T. J. et al. Results of a pilot program to improve phosphorus outcomes in hemodialysis patients. J. Ren. Nutr. 22, 472–479 (2012).
D'Alessandro, C., Piccoli, G. B. & Cupisti, A. The “phosphorus pyramid”: a visual tool for dietary phosphate management in dialysis and CKD patients. BMC Nephrol. 16, 9 (2015).
Klahr, S. et al. The effects of dietary protein restriction and blood-pressure control on the progression of chronic renal disease. N. Engl. J. Med. 330, 877–884 (1994). This landmark trial on prevention of CKD progression is among the most influential trials ever performed in nephrology.
Levey, A. S. et al. Effect of dietary protein restriction on the progression of kidney disease: long-term follow-up of the Modification of Diet in Renal Disease (MDRD) Study. Am. J. Kidney Dis. 48, 879–888 (2006).
Evenepoel, P. & Vervloet, M. G. Dietary phosphorus restriction in predialysis chronic kidney disease: time for a cease-fire? Kidney Int. 89, 21–23 (2016).
Eckberg, K. et al. Impact of westernization on fibroblast growth factor 23 levels among individuals of African ancestry. Nephrol. Dial. Transplant. 30, 630–635 (2015).
Lenglet, A. et al. Use of nicotinamide to treat hyperphosphatemia in dialysis patients. Drugs R. D. 13, 165–173 (2013).
Lenglet, A. et al. Efficacy and safety of nicotinamide in haemodialysis patients: the NICOREN study. Nephrol. Dial. Transplant. http://dx.doi.org/10.1093/ndt/gfw042 (2016).
Jamal, S. A. et al. Effect of calcium-based versus non-calcium-based phosphate binders on mortality in patients with chronic kidney disease: an updated systematic review and meta-analysis. Lancet 382, 1268–1277 (2013).
Cozzolino, M., Mazzaferro, S. & Brandenburg, V. The treatment of hyperphosphataemia in CKD: calcium-based or calcium-free phosphate binders? Nephrol. Dial. Transplant. 26, 402–407 (2011).
D'Haese, P. C. et al. A multicenter study on the effects of lanthanum carbonate (Fosrenol) and calcium carbonate on renal bone disease in dialysis patients. Kidney Int. Suppl. 85, S73–S78 (2003).
de Roij van Zuijdewijn, C. L. et al. Serum magnesium and sudden death in European hemodialysis patients. PLoS ONE 10, e0143104 (2015).
De Schutter, T. M. et al. Effect of a magnesium-based phosphate binder on medial calcification in a rat model of uremia. Kidney Int. 83, 1109–1117 (2013).
Block, G. A. et al. Effects of phosphate binders in moderate CKD. J. Am. Soc. Nephrol. 23, 1407–1415 (2012). The only study of sufficient size that has investigated the efficacy of phosphate binders versus placebo in non-dialysis CKD.
Di Iorio, B. et al. Mortality in kidney disease patients treated with phosphate binders: a randomized study. Clin. J. Am. Soc. Nephrol. 7, 487–493 (2012).
Di Iorio, B. et al. Sevelamer versus calcium carbonate in incident hemodialysis patients: results of an open-label 24-month randomized clinical trial. Am. J. Kidney Dis. 62, 771–778 (2013).
Suki, W. N. et al. Effects of sevelamer and calcium-based phosphate binders on mortality in hemodialysis patients. Kidney Int. 72, 1130–1137 (2007).
Cannata-Andia, J. B. et al. Use of phosphate-binding agents is associated with a lower risk of mortality. Kidney Int. 84, 998–1008 (2013).
Patel, L., Bernard, L. M. & Elder, G. J. Sevelamer versus calcium-based binders for treatment of hyperphosphatemia in CKD: a meta-analysis of randomized controlled trials. Clin. J. Am. Soc. Nephrol. 11, 232–244 (2015). This meta-analysis provides a very comprehensive summary of data on the efficacy of calcium-based phosphate binders compared with sevelamer.
Iimori, S. et al. Effects of sevelamer hydrochloride on mortality, lipid abnormality and arterial stiffness in hemodialyzed patients: a propensity-matched observational study. Clin. Exp. Nephrol. 16, 930–937 (2012).
Hill, K. M. et al. Oral calcium carbonate affects calcium but not phosphorus balance in stage 3–4 chronic kidney disease. Kidney Int. 83, 959–966 (2013).
Acknowledgements
Board members of the ERA–EDTA Working Group on Chronic Kidney Disease–Mineral and Bone Disorders: David Goldsmith, Pieter Evenepoel, Vincent Brandenburg, Jordi Bover, Sandro Mazzaferro, Pablo Ureña-Torres, and Adrian Covic. Board members of the European Renal Nutrition Working Group: Juan Jesus Carrero, Vincenzo Bellizzi, Philippe Chauveau, Pablo Molina, Daniel Teta, Christoph Wanner, and Piet Ter Wee.
Author information
Authors and Affiliations
Consortia
Contributions
All authors researched the data for the article, wrote the manuscript, contributed to discussions of the content and reviewed or edited the manuscript before submission.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- Hyperphosphataemia
-
A serum phosphate concentration higher than the upper limit of the normal range, that is > 4.5 mg/dl (> 1.5 mmol/l).
- Paracellular route
-
Transport of ions or other substances between two adjacent cells that are part of a cell layer.
- Transcellular route
-
Transport of ions or other substances through a cell by passing through both the apical and basolateral membrane.
- Phosphaturia
-
Renal excretion of phosphate in the urine.
- Circadian rhythm
-
A day–night pattern of variation in a biological process.
- Hyperdynamic bone disease
-
Bone disease resulting from an abnormally high rate of bone formation by osteoblasts and increased bone degradation by osteoclasts.
- Calcimimetics
-
A class of agents that increase the sensitivity of the calcium-sensing receptor for extracellular calcium. Calcimimetics are used to treat hyperparathyroidism.
- Protein–energy wasting
-
Depletion of body stores owing to insufficient nutrient intake, chronic inflammation and acute or chronic catabolism that can eventually lead to cachexia and death. An insufficient dialysis dose can contribute to protein–energy wasting in patients receiving maintenance dialysis.
- CKD–mineral and bone disorder
-
A syndrome characterized by the co-occurrence of derangements in the levels of calcium, phosphate and the hormones that regulate these minerals, with fractures, vascular calcifications and cardiovascular disease in patients with CKD.
- Osteogenic transdifferentiation
-
The phenotype switch of vascular smooth muscle cells to osteoblast-like bone-forming cells.
- Left ventricular hypertrophy
-
A usually pathological growth of cardiomyocytes that occurs following chronic exposure of the heart to mechanical or non-mechanical stress and leads to heart failure.
- Adynamic bone disease
-
Bone disease resulting from an abnormally low rate of bone formation by osteoblasts and decreased bone degradation by osteoclasts.
Rights and permissions
About this article
Cite this article
Vervloet, M., Sezer, S., Massy, Z. et al. The role of phosphate in kidney disease. Nat Rev Nephrol 13, 27–38 (2017). https://doi.org/10.1038/nrneph.2016.164
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneph.2016.164
This article is cited by
-
Longer serum phosphorus time in range associated with lower mortality risk among peritoneal dialysis patients: a multicenter retrospective cohort study
BMC Nephrology (2024)
-
Determination of phosphate in food based on molybdenum yellow derivatization coupled with resonance Rayleigh scattering method
Analytical Sciences (2024)
-
Existing and emerging technologies for the removal of orthophosphate from wastewater by agricultural waste adsorbents: a review
Biomass Conversion and Biorefinery (2023)
-
Construction of nanohybrid Tb@CDs/GSH-CuNCs as a ratiometric probe to detect phosphate anion based on aggregation-induced emission and FRET mechanism
Microchimica Acta (2023)
-
Reversal Of Arterial Disease by modulating Magnesium and Phosphate (ROADMAP-study): rationale and design of a randomized controlled trial assessing the effects of magnesium citrate supplementation and phosphate-binding therapy on arterial stiffness in moderate chronic kidney disease
Trials (2022)