Key Points
-
ABO-incompatible (ABOi) kidney transplantation is now an established treatment option for patients with end-stage renal disease, but the mechanisms that underlie acceptance of ABOi grafts are not well understood
-
The biology of the ABO system is complex; blood group subtypes and organ-specific patterns of core-chain tissue distribution require particular consideration in the context of ABOi transplantation
-
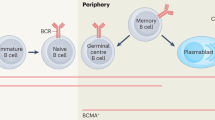
Innovative humanized animal models are expected to provide a better understanding of anti-A/B immune responses and might help to establish innovative therapeutic strategies to counteract blood-group-specific B-cell responses
-
The development of efficient desensitization protocols including apheresis, modulation of B-cell immunity and long-term maintenance immunosuppression has enabled ABOi kidney transplantation to become a safe treatment strategy with favourable outcomes
-
Although the reduction of pretransplant anti-A/B antibody titres below a permissive threshold is a major principle of desensitization, thresholds at which antibody-mediated damage can be predicted have not been defined
-
Tailoring the intensity of preconditioning for ABOi kidney transplant recipients according to their pretransplant anti-A/B antibody titres might be an efficient strategy to minimize the risks associated with enhanced immunosuppression
Abstract
Kidney transplantation across the ABO blood group barrier was long considered a contraindication for transplantation, but in an effort to increase donor pools, specific regimens for ABO-incompatible (ABOi) transplantation have been developed. These regimens are now widely used as an integral part of the available treatment options. Various desensitization protocols, commonly based on transient depletion of preformed anti-A and/or anti-B antibodies and modulation of B-cell immunity, enable excellent transplant outcomes, even in the long-term. Nevertheless, the molecular mechanisms behind transplant acceptance facilitated by a short course of anti-humoral treatment are still incompletely understood. With the evolution of efficient clinical programmes, tailoring of recipient preconditioning based on individual donor–recipient blood type combinations and the levels of pretransplant anti-A/B antibodies has become possible. In the context of low antibody titres and/or donor A2 phenotype, immunomodulation and/or apheresis might be dispensable. A concern still exists, however, that ABOi kidney transplantation is associated with an increased risk of surgical and infectious complications, partly owing to the effects of extracorporeal treatment and intensified immunosuppression. Nevertheless, a continuous improvement in desensitization strategies, with the aim of minimizing the immunosuppressive burden, might pave the way to clinical outcomes that are comparable to those achieved in ABO-compatible transplantation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Hume, D. M., Merrill, J. P., Miller, B. F. & Thorn, G. W. Experiences with renal homotransplantation in the human: report of nine cases. J. Clin. Invest. 34, 327–382 (1955).
Starzl, T. E. et al. Renal homografts in patients with major donor-recipient blood group incompatibilities. Surgery 55, 195–200 (1964).
Cook, D. J., Graver, B. & Terasaki, P. I. ABO incompatibility in cadaver donor kidney allografts. Transplant. Proc. 19, 4549–4552 (1987).
Williams, G. M. et al. Hyperacute renal-homograft rejection in man. N. Engl. J. Med. 279, 611–618 (1968).
Starzl, T. E. et al. The definition of ABO factors in transplantation: relation to other humoral antibody states. Transplant. Proc. 19, 4492–4497 (1987).
Squifflet, J. P. et al. Lessons learned from ABO-incompatible living donor kidney transplantation: 20 years later. Exp. Clin. Transplant. 2, 208–213 (2004).
Alexandre, G. P. et al. Present experiences in a series of 26 ABO-incompatible living donor renal allografts. Transplant. Proc. 19, 4538–4542 (1987).
Landsteiner, K. Über Agglutinationserscheinungen normalen menschlichen Blutes [German]. Wien Klin. Wochenschr. 14, 1132–1134 (1901).
Oriol, R. ABH and related tissue antigens. Biochem. Soc. Trans. 15, 596–599 (1987).
Kelly, R. J., Rouquier, S., Giorgi, D., Lennon, G. G. & Lowe, J. B. Sequence and expression of a candidate for the human Secretor blood group α(1,2)fucosyltransferase gene (FUT2). Homozygosity for an enzyme-inactivating nonsense mutation commonly correlates with the non-secretor phenotype. J. Biol. Chem. 270, 4640–4649 (1995).
Mollicone, R., Cailleau, A. & Oriol, R. Molecular genetics of H, Se, Lewis and other fucosyltransferase genes. Transfus. Clin. Biol. 2, 235–242 (1995).
Rouquier, S. et al. Molecular cloning of a human genomic region containing the H blood group α(1,2)fucosyltransferase gene and two H locus-related DNA restriction fragments. Isolation of a candidate for the human Secretor blood group locus. J. Biol. Chem. 270, 4632–4639 (1995).
Ravn, V. & Dabelsteen, E. Tissue distribution of histo-blood group antigens. APMIS 108, 1–28 (2000).
Clausen, H. & Hakomori, S. ABH and related histo-blood group antigens; immunochemical differences in carrier isotypes and their distribution. Vox Sang. 56, 1–20 (1989).
Yamamoto, F., Clausen, H., White, T., Marken, J. & Hakomori, S. Molecular genetic basis of the histo-blood group ABO system. Nature 345, 229–233 (1990).
Bhende, Y. et al. A “new” blood group character related to the ABO system. Lancet 1, 903–904 (1952).
Malhotra, S., Dhawan, H. K., Jain, A., Sachdev, S. & Marwaha, N. Acute hemolytic transfusion reaction in a patient with Bombay phenotype: implications for ABO grouping. Indian J. Hematol. Blood Transfus. 30, 108–110 (2014).
Townamchai, N. et al. A case of nearly mistaken AB para-Bombay blood group donor transplanted to a group 'O' recipient. BMJ Case Rep. http://dx.doi.org/10.1136/bcr-2014-206374.
Breimer, M. E. et al. Blood group A and B antigen expression in human kidneys correlated to A1/A2/B, Lewis, and secretor status. Transplantation 82, 479–485 (2006).
Oriol, R., Cartron, J. P., Cartron, J. & Mulet, C. Biosynthesis of ABH and Lewis antigens in normal and transplanted kidneys. Transplantation 29, 184–188 (1980).
Holgersson, J. et al. Blood group A glycolipid antigen expression in kidney, ureter, kidney artery, and kidney vein from a blood group A1Lea-b+ human individual. Evidence for a novel blood group A heptaglycosylceramide based on a type 3 carbohydrate chain. J. Biol. Chem. 265, 20790–20798 (1990).
Holgersson, J., Rydberg, L. & Breimer, M. E. Molecular deciphering of the ABO system as a basis for novel diagnostics and therapeutics in ABO incompatible transplantation. Int. Rev. Immunol. 33, 174–94 (2014).
Breimer, M. E. et al. Blood group ABO-incompatible kidney transplantation biochemical and immunochemical studies of blood group A glycolipid antigens in human kidney and characterization of the antibody response (antigen specificity and antibody class) in O recipients receiving A2 grafts. Transplant. Proc. 19, 226–230 (1987).
Tasaki, M. et al. Identification and characterization of major proteins carrying ABO blood group antigens in the human kidney. Transplantation 87, 1125–1133 (2009).
Rydberg, L., Breimer, M. E., Samuelsson, B. E. & Brynger, H. Blood group ABO-incompatible (A2 to O) kidney transplantation in human subjects: a clinical, serologic, and biochemical approach. Transplant. Proc. 19, 4528–4537 (1987).
Rydberg, L., Breimer, M. E., Brynger, H. & Samuelsson, B. E. ABO-incompatible kidney transplantation (A2 to O). Qualitative and semiquantitative studies of the humoral immune response against different blood group A antigens. Transplantation 49, 954–960 (1990).
Nelson, P. W., Helling, T. S., Shield, C. F., Beck, M. & Bryan, C. F. Current experience with renal transplantation across the ABO barrier. Am. J. Surg. 164, 541–544 (1992).
Meloncelli, P. J., West, L. J. & Lowary, T. L. Synthesis and NMR studies on the ABO histo-blood group antigens: synthesis of type III and IV structures and NMR characterization of type I-VI antigens. Carbohydr. Res. 346, 1406–1426 (2011).
Zschiedrich, S. et al. An update on ABO-incompatible kidney transplantation. Transplant. Int. 28, 387–397 (2014).
Breimer, M. E. & Samuelsson, B. E. The specific distribution of glycolipid-based blood group A antigens in human kidney related to A1/A2, Lewis, and secretor status of single individuals. A possible molecular explanation for the successful transplantation of A2 kidneys into O recipients. Transplantation 42, 88–91 (1986).
Breimer, M. E. & Jovall, P. A. Structural characterization of a blood group A heptaglycosylceramide with globo-series structure. The major glycolipid based blood group A antigen of human kidney. FEBS Lett. 179, 165–172 (1985).
Jeyakanthan, M. et al. Chemical basis for qualitative and quantitative differences between ABO blood groups and subgroups: implications for organ transplantation. Am. J. Transplant. http://dx.doi.org/10.1111/ajt.13328.
Baumgarth, N., Tung, J. W. & Herzenberg, L. A. Inherent specificities in natural antibodies: a key to immune defense against pathogen invasion. Springer Semin. Immunopathol. 26, 347–362 (2005).
Rieben, R., Bbuchs, J. P., Flückiger, E. & Nydegger, U. E. Antibodies to histo-blood group substances A and B: agglutination titers, Ig class, and IgG subclasses in healthy persons of different age categories. Transfusion 31, 607–615 (1991).
Andersson, M., Carlin, N., Leontein, K., Lindquist, U. & Slettengren, K. Structural studies of the O-antigenic polysaccharide of Escherichia coli O86, which possesses blood-group B activity. Carbohydr. Res. 185, 211–223 (1989).
Springer, G. F., Horton, R. E. & Forbes, M. Origin of antihuman blood group B agglutinins in germfree chicks. Ann. NY Acad. Sci. 78, 272–275 (1959).
Springer, G. F. & Horton, R. E. Blood group isoantibody stimulation in man by feeding blood group-active bacteria. J. Clin. Invest. 48, 1280–1291 (1969).
Spalter, S. H. et al. Normal human serum contains natural antibodies reactive with autologous ABO blood group antigens. Blood 93, 4418–4424 (1999).
Genberg, H., Kumlien, G., Wennberg, L. & Tyden, G. The efficacy of antigen-specific immunoadsorption and rebound of anti-A/B antibodies in ABO-incompatible kidney transplantation. Nephrol. Dial. Transplant. 26, 2394–2400 (2011).
Rieben, R., Frauenfelder, A. & Nydegger, U. E. Naturally occurring ABO antibodies: long-term stable, individually distinct anti-A IgG spectrotypes. Eur. J. Immunol. 22, 2129–2133 (1992).
Iwasaki, K. et al. Comparative study on signal transduction in endothelial cells after anti-a/b and human leukocyte antigen antibody reaction: implication of accommodation. Transplantation 93, 390–397 (2012).
Fan, X. et al. Induction of human blood group A antigen expression on mouse cells, using lentiviral gene transduction. Hum. Gene Ther. 21, 877–890 (2010).
Ishida, H. et al. Differences in humoral immunity between a non-rejection group and a rejection group after ABO-incompatible renal transplantation. Transplantation 81, 665–671 (2006).
Pecanha, L. M., Snapper, C. M., Finkelman, F. D. & Mond, J. J. Dextran-conjugated anti-Ig antibodies as a model for T cell-independent type 2 antigen-mediated stimulation of Ig secretion in vitro. I. Lymphokine dependence. J. Immunol. 146, 833–839 (1991).
Snapper, C. M. et al. Induction of IgG3 secretion by interferon γ: a model for T cell-independent class switching in response to T cell-independent type 2 antigens. J. Exp. Med. 175, 1367–1371 (1992).
Mond, J. J., Lees, A. & Snapper, C. M. T cell-independent antigens type 2. Annu. Rev. Immunol. 13, 655–692 (1995).
Lang, G. A., Exley, M. A. & Lang, M. L. The CD1d-binding glycolipid α-galactosylceramide enhances humoral immunity to T-dependent and T-independent antigen in a CD1d-dependent manner. Immunology 119, 116–125 (2006).
Tonti, E. et al. Follicular helper NKT cells induce limited B cell responses and germinal center formation in the absence of CD4+ T cell help. J. Immunol. 188, 3217–3222 (2012).
Yamamoto, M. et al. Murine equivalent of the human histo-blood group ABO gene is a cis-AB gene and encodes a glycosyltransferase with both A and B transferase activity. J. Biol. Chem. 276, 13701–13708 (2001).
Fan, X., Ang, A., Tao, K. & West, L. J. Induction of human histo-blood group A antigen expression in mouse cells by gene therapy using lentiviral vectors harbouring human ABH-related glycosyltransferase genes. Transplant. Proc. 37, 265–267 (2005).
Zhou, W. et al. NOD/SCID mice engrafted with human peripheral blood lymphocytes can be a model for investigating B cells responding to blood group A carbohydrate determinant. Transpl. Immunol. 12, 9–18 (2003).
Irei, T. et al. The persistent elimination of B cells responding to blood group A carbohydrates by synthetic group A carbohydrates and B-1 cell differentiation blockade: novel concept in preventing antibody-mediated rejection in ABO-incompatible transplantation. Blood 110, 4567–4575 (2007).
Tazawa, H. et al. Blockade of invariant TCR-CD1d interaction specifically inhibits antibody production against blood group A carbohydrates. Blood 122, 2582–2590 (2013).
Wekerle, T. Taming the ABO barrier in transplantation. Blood 122, 2527–2528 (2013).
Park, W. D. et al. Accommodation in ABO-incompatible kidney allografts, a novel mechanism of self-protection against antibody-mediated injury. Am. J. Transplant. 3, 952–960 (2003).
Bentall, A. et al. Differences in chronic intragraft inflammation between positive crossmatch and ABO-incompatible kidney transplantation. Transplantation 98, 1089–1096 (2014).
Bach, F. H. et al. Accommodation of vascularized xenografts: expression of “protective genes” by donor endothelial cells in a host Th2 cytokine environment. Nat. Med. 3, 196–204 (1997).
Chen Song, S. et al. Complement inhibition enables renal allograft accommodation and long-term engraftment in presensitized nonhuman primates. Am. J. Transplant. 11, 2057–2066 (2011).
Koestner, S. C. et al. Histo-blood group type change of the graft from B to O after ABO mismatched heart transplantation. Lancet 363, 1523–1525 (2004).
Tanabe, T. et al. Decrease of blood type antigenicity over the long-term after ABO-incompatible kidney transplantation. Transpl. Immunol. 25, 1–6 (2011).
Tanabe, T. et al. Endothelial chimerism after ABO-incompatible kidney transplantation. Transplantation 93, 709–716 (2012).
Barbolla, L., Mojena, M., Cienfuegos, J. A. & Escartin, P. Presence of an inhibitor of glycosyltransferase activity in a patient following an ABO incompatible liver transplant. Br. J. Haematol. 69, 93–96 (1988).
West, L. J. et al. ABO-incompatible heart transplantation in infants. N. Engl. J. Med. 344, 793–800 (2001).
West, L. J. ABO-incompatible hearts for infant transplantation. Curr. Opin. Organ Transplant. 16, 548–554 (2011).
West, L. J. et al. Impact on outcomes after listing and transplantation, of a strategy to accept ABO blood group-incompatible donor hearts for neonates and infants. J. Thorac. Cardiovasc. Surg. 131, 455–461 (2006).
Fan, X. et al. Donor-specific B-cell tolerance after ABO-incompatible infant heart transplantation. Nat. Med. 10, 1227–1233 (2004).
Urschel, S. et al. C3d plasma levels and CD21 expressing B-cells in children after ABO-incompatible heart transplantation: Alterations associated with blood group tolerance. J. Heart Lung Transplant. 33, 1149–1156 (2014).
Urschel, S. et al. ABO-incompatible heart transplantation in early childhood: an international multicenter study of clinical experiences and limits. J. Heart Lung Transplant. 32, 285–292 (2013).
Urschel, S. et al. Absence of donor-specific anti-HLA antibodies after ABO-incompatible heart transplantation in infancy: altered immunity or age? Am. J. Transplant. 10, 149–156 (2010).
Jeyakanthan, M. et al. Failure of neonatal B-cell tolerance induction by ABO-incompatible kidney grafts in piglets. Transplantation 96, 519–528 (2013).
Redfield, R. R. et al. Underutilization of A2 ABO incompatible kidney transplantation. Clin. Transplant. 26, 489–494 (2012).
Barnett, A. N. et al. Distribution of ABO blood group antibody titers in pediatric patients awaiting renal transplantation: implications for organ allocation policy. Transplantation 94, 362–368 (2012).
Israni, A. K. et al. New national allocation policy for deceased donor kidneys in the United States and possible effect on patient outcomes. J. Am. Soc. Nephrol. 25, 1842–1848 (2014).
Segev, D. L., Gentry, S. E., Melancon, J. K. & Montgomery, R. A. Characterization of waiting times in a simulation of kidney paired donation. Am. J. Transplant. 5, 2448–2455 (2005).
Takahashi, K. & Saito, K. ABO-incompatible kidney transplantation. Transplant. Rev. (Orlando) 27, 1–8 (2013).
Montgomery, J. R. et al. Outcomes of ABO-incompatible kidney transplantation in the United States. Transplantation 93, 603–609 (2013).
Ferrari, P. et al. ABO-incompatible matching significantly enhances transplant rates in kidney paired donation. Transplantation 96, 821–826 (2013).
Glorie, K., Haase-Kromwijk, B., van de Klundert, J., Wagelmans, A. & Weimar, W. Allocation and matching in kidney exchange programs. Transpl. Int. 27, 333–343 (2013).
Montgomery, R. A. et al. ABO incompatible renal transplantation: a paradigm ready for broad implementation. Transplantation 87, 1246–1255 (2009).
Morath, C. et al. ABO-incompatible kidney transplantation enabled by non-antigen-specific immunoadsorption. Transplantation 93, 827–834 (2012).
Barnett, A. N. et al. Tailored desensitization strategies in ABO blood group antibody incompatible renal transplantation. Transpl. Int. 27, 187–196 (2013).
Masterson, R. et al. ABO incompatible renal transplantation without antibody removal using conventional immunosuppression alone. Am. J. Transplant. 14, 2807–2813 (2014).
Geyer, M. et al. Pre-emptive postoperative antigen-specific immunoadsorption in ABO-incompatible kidney transplantation: necessary or not? Transplantation 84, S40–S43 (2007).
Wilpert, J. et al. Long-term outcome of ABO-incompatible living donor kidney transplantation based on antigen-specific desensitization. An observational comparative analysis. Nephrol. Dial. Transplant. 25, 3778–3786 (2010).
Tanabe, K. Double-filtration plasmapheresis. Transplantation 84, S30–S32 (2007).
Hanafusa, N. et al. The effect of different apheresis modalities on coagulation factor XIII level during antibody removal in ABO-blood type incompatible living related renal transplantation. Transfus. Apher. Sci. 49, 254–258 (2014).
Renner, F. C. et al. Postoperative bleeding after AB0-incompatible living donor kidney transplantation. Transplant. Proc. 42, 4164–4166 (2010).
Yeh, J. H. & Chiu, H. C. Coagulation abnormalities in serial double-filtration plasmapheresis. J. Clin. Apher. 16, 139–142 (2001).
Tyden, G., Kumlien, G. & Fehrman, I. Successful ABO-incompatible kidney transplantations without splenectomy using antigen-specific immunoadsorption and rituximab. Transplantation 76, 730–731 (2003).
Tyden, G., Kumlien, G. & Efvergren, M. Present techniques for antibody removal. Transplantation 84, S27–S29 (2007).
Biglarnia, A. R. et al. Desensitization with antigen-specific immunoadsorption interferes with complement in ABO-incompatible kidney transplantation. Transplantation 93, 87–92 (2012).
Lindberg, L., Theinert, K., Liu, J. & Holgersson, J. Adsorption of chain type-specific ABO antibodies on Sepharose-linked A and B tetrasaccharides. Transfusion 52, 2356–2367 (2012).
Schiesser, M. et al. The reuse of immunoadsorption columns in ABO-incompatible kidney transplantation is efficient: the Swiss experience. Transplantation 99, 1030–1035 (2015).
Wahrmann, M. et al. Anti-A/B antibody depletion by semiselective versus ABO blood group-specific immunoadsorption. Nephrol. Dial. Transplant. 27, 2122–2129 (2012).
Eskandary, F. et al. ABO antibody and complement depletion by immunoadsorption combined with membrane filtration—a randomized, controlled, cross-over trial. Nephrol. Dial. Transplant. 29, 706–714 (2014).
Teschner, S. et al. ABO-incompatible kidney transplantation using regenerative selective immunoglobulin adsorption. J. Clin. Apher. 27, 51–60 (2012).
US National Library of Medicine. ClinicalTrials.gov [online], (2014).
Lentine, K. L. et al. Early clinical complications after ABO-incompatible live-donor kidney transplantation: a national study of Medicare-insured recipients. Transplantation 98, 54–65 (2014).
Sonnenday, C. J. et al. Plasmapheresis, CMV hyperimmune globulin, and anti-CD20 allow ABO-incompatible renal transplantation without splenectomy. Am. J. Transplant. 4, 1315–1322 (2004).
Gloor, J. M. et al. A comparison of splenectomy versus intensive post-transplant antidonor blood group antibody monitoring without splenectomy in ABO-incompatible kidney transplantation. Transplantation 80, 1572–1577 (2005).
Kaplan, B. et al. Successful rescue of refractory, severe antibody mediated rejection with splenectomy. Transplantation 83, 99–100 (2007).
Tzvetanov, I. et al. Rescue splenectomy in a case of humoral rejection in ABO-incompatible simultaneous liver-kidney transplantation. Transplantation 93, e1–e2 (2012).
Kamburova, E. G. et al. A single dose of rituximab does not deplete B cells in secondary lymphoid organs but alters phenotype and function. Am. J. Transplant. 13, 1503–1511 (2013).
Tyden, G. et al. A randomized, doubleblind, placebo-controlled, study of single-dose rituximab as induction in renal transplantation. Transplantation 87, 1325–1329 (2009).
Tyden, G., Ekberg, H., Tufveson, G. & Mjornstedt, L. A randomized, double-blind, placebo-controlled study of single dose rituximab as induction in renal transplantation: a 3-year follow-up. Transplantation 94, e21–e22 (2013).
Clatworthy, M. R. et al. B-cell-depleting induction therapy and acute cellular rejection. N. Engl. J. Med. 360, 2683–2685 (2009).
Kohei, N., Hirai, T., Omoto, K., Ishida, H. & Tanabe, K. Chronic antibody-mediated rejection is reduced by targeting B-cell immunity during an introductory period. Am. J. Transplant. 12, 469–476 (2012).
Ashimine, S. et al. Neither pre-transplant rituximab nor splenectomy affects de novo HLA antibody production after renal transplantation. Kidney Int. 85, 425–430 (2013).
Shirakawa, H. et al. The low dose of rituximab in ABO-incompatible kidney transplantation without a splenectomy: a single-center experience. Clin. Transplant. 25, 878–884 (2011).
Fuchinoue, S. et al. The 5-year outcome of ABO-incompatible kidney transplantation with rituximab induction. Transplantation 91, 853–857 (2011).
Flint, S. M. et al. Successful ABO-incompatible kidney transplantation with antibody removal and standard immunosuppression. Am. J. Transplant. 11, 1016–1024 (2011).
Opelz, G. et al. Three-year outcomes following 1,420 ABO-incompatible living-donor kidney transplants performed after ABO antibody reduction: results from 101 centers. Transplantation 99, 400–404 (2015).
Renner, F. C. et al. Impact of pretransplant intravenous immunoglobulin administration on anti-AB0 antibody levels in AB0-incompatible living donor kidney transplantation. Transplant. Proc. 42, 4003–4005 (2010).
Shimmura, H. et al. Lack of correlation between results of ABO-incompatible living kidney transplantation and anti-ABO blood type antibody titers under our current immunosuppression. Transplantation 80, 985–988 (2005).
Ishida, H. et al. Evaluation of immunosuppressive regimens in ABO-incompatible living kidney transplantation—single center analysis. Am. J. Transplant. 7, 825–831 (2007).
Mannami, M. & Mitsuhata, N. Improved outcomes after ABO-incompatible living-donor kidney transplantation after 4 weeks of treatment with mycophenolate mofetil. Transplantation 79, 1756–1758 (2005).
Wennberg, L., Genberg, H. & Tyden, G. Preapheresis immunosuppressive induction: necessary or harmful? Transplantation 84, S37–S39 (2007).
Uchida, J. et al. Conversion of stable ABO-incompatible kidney transplant recipients from mycophenolate mofetil with standard exposure calcineurin inhibitors (CNIs) to everolimus with very low exposure CNIs—a short-term pilot study. Clin. Transplant. 28, 80–87 (2014).
Catalano, G. et al. Everolimus-based immunosuppression in a case of ABO-incompatible liver transplantation with calcineurin inhibitor-related posterior occipital syndrome. Transpl. Int. 27, e84–e86 (2014).
Oettl, T. et al. Late steroid withdrawal after ABO blood group-incompatible living donor kidney transplantation: high rate of mild cellular rejection. Transplantation 89, 702–706 (2010).
Galliford, J. et al. ABO incompatible living renal transplantation with a steroid sparing protocol. Transplantation 86, 901–906 (2008).
Kato, Y. et al. Early steroid withdrawal protocol with basiliximab, cyclosporine and mycophenolate mofetil in renal-transplant recipients. Int. Immunopharmacol. 6, 1984–1992 (2006).
Kumlien, G., Wilpert, J., Säfwenberg, J. & Tydén, G. Comparing the tube and gel techniques for ABO antibody titration, as performed in three European centers. Transplantation 84, S17–S19 (2007).
Yung, G. P. et al. Flow cytometric measurement of ABO antibodies in ABO-incompatible living donor kidney transplantation. Transplantation 84, S20–S23 (2007).
Bentall, A. et al. No progress in ABO titer measurement: time to aim for a reference? Transplantation 97, e19–e21 (2014).
Kobayashi, T. & Saito, K. A series of surveys on assay for anti-A/B antibody by Japanese ABO-incompatible Transplantation Committee. Xenotransplantation 13, 136–140 (2006).
Kimura, S. et al. Rapid quantitation of immunoglobulin G antibodies specific for blood group antigens A and B by surface plasmon resonance. Transfusion 45, 56–62 (2005).
Stussi, G. et al. Isotype-specific detection of ABO blood group antibodies using a novel flow cytometric method. Br. J. Haematol. 130, 954–963 (2005).
Krishnan, N. S. et al. Application of flow cytometry to monitor antibody levels in ABO incompatible kidney transplantation. Transplantation 86, 474–477 (2008).
Valli, P. V. et al. Changes of circulating antibody levels induced by ABO antibody adsorption for ABO-incompatible kidney transplantation. Am. J. Transplant. 9, 1072–1080 (2009).
Fischer, M. B. et al. Surface plasmon resonance analysis shows an IgG-isotype-specific defect in ABO blood group antibody formation in patients with common variable immunodeficiency. Front. Immunol. 6, 211 (2015).
Gloor, J. M. et al. ABO-incompatible kidney transplantation using both A2 and non-A2 living donors. Transplantation 75, 971–977 (2003).
Tobian, A. A. et al. ABO antibody titer and risk of antibody-mediated rejection in ABO-incompatible renal transplantation. Am. J. Transplant. 10, 1247–1253 (2010).
van Agteren, M. et al. The first fifty ABO blood group incompatible kidney transplantations: the Rotterdam experience. J. Transplant. 2014, 913902 (2014).
Ishida, H., Kondo, T., Shimizu, T., Nozaki, T. & Tanabe, K. Postoperative rebound of antiblood type antibodies and antibody-mediated rejection after ABO-incompatible living-related kidney transplantation. Transpl. Int. 28, 286–296 (2015).
Takahashi, K. & Saito, K. Present status of ABO-incompatible kidney transplantation in Japan. Xenotransplantation 13, 118–122 (2006).
Tyden, G. et al. Implementation of a protocol for ABO-incompatible kidney transplantation—a three-center experience with 60 consecutive transplantations. Transplantation 83, 1153–1155 (2007).
Norden, G. et al. ABO-incompatible live donor renal transplantation using blood group A/B carbohydrate antigen immunoadsorption and anti-CD20 antibody treatment. Xenotransplantation 13, 148–153 (2006).
Haidinger, M. et al. Vienna experience of ABO-incompatible living-donor kidney transplantation [German]. Wien. Klin. Wochenschr. 121, 247–255 (2009).
Habicht, A. et al. Increase of infectious complications in ABO-incompatible kidney transplant recipients—a single centre experience. Nephrol. Dial. Transplant. 26, 4124–4131 (2011).
Toki, D., Ishida, H., Horita, S., Yamaguchi, Y. & Tanabe, K. Blood group O recipients associated with early graft deterioration in living ABO-incompatible kidney transplantation. Transplantation 88, 1186–1193 (2009).
Gloor, J. M. et al. Histologic findings one year after positive crossmatch or ABO blood group incompatible living donor kidney transplantation. Am. J. Transplant. 6, 1841–1847 (2006).
Haas, M. et al. C4d and C3d staining in biopsies of ABO and HLA-incompatible renal allografts: correlation with histologic findings. Am. J. Transplant. 6, 1829–1840 (2006).
Setoguchi, K. et al. Analysis of renal transplant protocol biopsies in ABO-incompatible kidney transplantation. Am. J. Transplant. 8, 86–94 (2008).
Brocker, V. et al. Beyond C4d: the ultrastructural appearances of endothelium in ABO-incompatible renal allografts. Nephrol. Dial. Transplant. 28, 3101–3109 (2013).
Bach, F. H., Turman, M. A., Vercellotti, G. M., Platt, J. L. & Dalmasso, A. P. Accommodation: a working paradigm for progressing toward clinical discordant xenografting. Transplant. Proc. 23, 205–207 (1991).
Haas, M. et al. C4d deposition without rejection correlates with reduced early scarring in ABO-incompatible renal allografts. J. Am. Soc. Nephrol. 20, 197–204 (2009).
Schaefer, B. et al. Bleeding complications in pediatric ABO-incompatible kidney transplantation. Pediatr. Nephrol. 28, 327–332 (2013).
Hwang, J. K. et al. Comparative analysis of ABO-incompatible living donor kidney transplantation with ABO-compatible grafts: a single-center experience in Korea. Transplant. Proc. 45, 2931–2936 (2013).
de Weerd, A. E. et al. ABO-incompatible kidney transplant recipients have a higher bleeding risk after antigen-specific immunoadsorption. Transpl. Int. 28, 25–33 (2015).
Lopau, K., Syamken, K., Rubenwolf, P., Riedmiller, H. & Wanner, C. Impact of mycophenolate mofetil on wound complications and lymphoceles after kidney transplantation. Kidney Blood Press. Res. 33, 52–59 (2010).
Montgomery, R. A. & Locke, J. E. ABO-incompatible transplantation: less may be more. Transplantation 84, S8–S9 (2007).
van den Hoogen, M. W. F. et al. Rituximab as induction therapy after renal transplantation: a randomized, double-blind, placebo-controlled study of efficacy and safety. Am. J. Transplant. 15, 407–416 (2015).
Oettl, T. et al. ABO blood group-incompatible living donor kidney transplantation: a prospective, single-centre analysis including serial protocol biopsies. Nephrol. Dial. Transplant. 24, 298–303 (2009).
Sharif, A. et al. Incidence and outcomes of BK virus allograft nephropathy among ABO and HLA-incompatible kidney transplant recipients. Clin. J. Am. Soc. Nephrol. 7, 1320–1327 (2012).
Hall, E. C., Engels, E. A., Montgomery, R. A. & Segev, D. L. Cancer risk after ABO-incompatible living-donor kidney transplantation. Transplantation 96, 476–479 (2013).
Kobayashi, T. et al. Removal of blood group A/B antigen in organs by ex vivo and in vivo administration of endo-β-galactosidase (ABase) for ABO-incompatible transplantation. Transpl. Immunol. 20, 132–138 (2009).
Hasegawa, Y. et al. Neutralization of blood group A-antigen by a novel anti-A antibody: overcoming ABO-incompatible solid-organ transplantation. Transplantation 85, 378–385 (2008).
Ye, Y. et al. Cardiac allotransplantation across the ABO-blood group barrier by the neutralization of preformed antibodies: the baboon as a model for the human. Lab. Anim. Sci. 44, 121–124 (1994).
Yoneyama, T. et al. Blood group antigen-targeting peptide suppresses anti-blood group antibody binding to antigen in renal glomerular capillaries after ABO-incompatible blood reperfusion. Transplantation 95, 418–425 (2013).
Stegall, M. D. et al. Terminal complement inhibition decreases antibody-mediated rejection in sensitized renal transplant recipients. Am. J. Transplant. 11, 2405–2413 (2011).
Biglarnia, A. R. et al. Prompt reversal of a severe complement activation by eculizumab in a patient undergoing intentional ABO-incompatible pancreas and kidney transplantation. Transpl. Int. 24, e61–e66 (2011).
US National Library of Medicine. ClinicalTrials.gov [online], (2010).
Author information
Authors and Affiliations
Contributions
All authors researched the data, made substantial contributions to discussions of the content, wrote the article and reviewed and edited the manuscript before submission.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Böhmig, G., Farkas, A., Eskandary, F. et al. Strategies to overcome the ABO barrier in kidney transplantation. Nat Rev Nephrol 11, 732–747 (2015). https://doi.org/10.1038/nrneph.2015.144
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneph.2015.144
This article is cited by
-
Association between ABO and Duffy blood types and circulating chemokines and cytokines
Genes & Immunity (2021)
-
Glycosphingolipids of human embryonic stem cells
Glycoconjugate Journal (2017)