Abstract
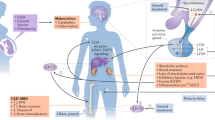
As children with chronic kidney disease (CKD) have a long lifespan, optimal control of bone and mineral homeostasis is essential not only for the prevention of debilitating skeletal complications and for achieving adequate growth but also for preserving long-term cardiovascular health. As the growing skeleton is highly dynamic and at particular risk of deterioration, close control of bone and mineral homeostasis is required in children with CKD. However, assessment of bone disease is hampered by the limited validity of biochemical parameters—major controversy exists on key issues such as parathyroid hormone target ranges and the lack of useful imaging techniques. The role of newly discovered factors in bone and mineral homeostasis, such as fibroblast growth factor 23, is not yet established. Even though scientific evidence is limited in children with CKD, ergocalciferol or cholecalciferol supplementation and the use of calcium-free phosphate binders is recommended. The new drug cinacalcet is highly promising; however, pediatric experience is still limited to observational data and the effect of cinacalcet on longitudinal growth and pubertal development is unknown. Randomized, controlled trials are underway, including studies of cinacalcet pharmacokinetics and pharmacodynamics in infants.
Key Points
-
Chronic kidney disease (CKD) mineral and bone disorder (MBD) is highly dynamic in growing children and requires close monitoring of age-dependent target values and repeated therapeutic adaptations
-
The validity of biochemical and imaging tools in children with CKD is uncertain
-
Ergocalciferol or cholecalciferol supplementation is advised in children who have CKD and a 25-hydroxyvitamin D level of <75 nmol/l
-
In children, sevelamer carbonate and paricalcitol or doxercalciferol should improve control of CKD-MBD; however, this effect has not been conclusively demonstrated
-
Cinacalcet could help control bone and mineral homeostasis in children with CKD; however, caution is advised until the results of current pediatric clinical research trials are available
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Moe, S. et al. Definition, evaluation, and classification of renal osteodystrophy: a position statement from Kidney Disease: Improving Global Outcomes (KDIGO). Kidney Int. 69, 1945–1953 (2006).
Borzych, D. et al. The bone and mineral disorder of children undergoing chronic peritoneal dialysis. Kidney Int. 78, 1295–1304 (2010).
Oh, J. et al. Advanced coronary and carotid arteriopathy in young adults with childhood-onset chronic renal failure. Circulation 106, 100–105 (2002).
Goodman, W. G. et al. Coronary-artery calcification in young adults with end-stage renal disease who are undergoing dialysis. N. Engl. J. Med. 342, 1478–1483 (2000).
McDonald, S. P. & Craig, J. C. Long-term survival of children with end-stage renal disease. N. Engl. J. Med. 350, 2654–2662 (2004).
Shroff, R. et al. Vitamin D deficiency is associated with short stature and may influence blood pressure control in paediatric renal transplant recipients. Pediatr. Nephrol. http://dx.doi.org/10.1007/s00467-011-1920-z.
Gwinner, W. et al. Early calcification of renal allografts detected by protocol biopsies: causes and clinical implications. Am. J. Transplant. 5, 1934–1941 (2005).
Ramirez, J. A. et al. The absorption of dietary phosphorus and calcium in hemodialysis patients. Kidney Int. 30, 753–759 (1986).
Bushinsky, D. A. Contribution of intestine, bone, kidney, and dialysis to extracellular fluid calcium content. Clin. J. Am. Soc. Nephrol. 5 (Suppl. 1), S12–S22 (2010).
Soldin, S. J., Brugnara, C. & Wong, E. C. (eds) Pediatric reference ranges (AACC-Press, Washington, 2003).
National Kidney Foundation. KDOQI Clinical Practice Guidelines for Nutrition in Children with CKD: 2008 Update. Am. J. Kidney Dis. 53 (Suppl. 2), S1–S124 (2009).
Brodehl, J. Assessment and interpretation of the tubular threshold for phosphate in infants and children. Pediatr. Nephrol. 8, 645 (1994).
Clayton, B. E., Jenkins, P. & Round, J. M. (eds) Paediatric chemical pathology: clinical tests and references ranges (Blackwell Scientific, Oxford 1980).
Sitzmann, F. C. in Pädiatrie (ed. Sitzmann, F. C.), 193 (Hippokrates Verlag, Stuttgart, 1995).
Häusler, M., Schäfer, C., Osterwinter, C. & Jahnen-Dechent, W. The physiologic development of fetuin-a serum concentrations in children. Pediatr. Res. 66, 660–664 (2009).
Wigger, M. et al. Fetuin-A serum concentrations in healthy children. Ann. Clin. Biochem. 46, 511–513 (2009).
Picca, S. et al. Parathyroid hormone levels in pubertal uremic adolescents treated with growth hormone. Pediatr. Nephrol. 19, 71–76 (2004).
Bhan, I. et al. Post-transplant hypophosphatemia: tertiary 'hyper-phosphatoninism'? Kidney Int. 70, 1486–1494 (2006).
Klaus, G. et al. Prevention and treatment of renal osteodystrophy in children on chronic renal failure: European guidelines. Pediatr. Nephrol. 21, 151–159 (2006).
Salusky, I. B. et al. Biochemical markers of renal osteodystrophy in pediatric patients undergoing CAPD/CCPD. Kidney Int. 45, 253–258 (1994).
Gal-Moscovici, A. & Popovtzer, M. M. New worldwide trends in presentation of renal osteodystrophy and its relationship to parathyroid hormone levels. Clin. Nephrol. 63, 284–289 (2005).
John, M. R. et al. A novel immunoradiometric assay detects full-length human PTH but not amino-terminally truncated fragments: implications for PTH measurements in renal failure. J. Clin. Endocrinol. Metab. 84, 4287–4290 (1999).
Salusky, I. B. et al. Similar predictive value of bone turnover using first- and second-generation immunometric PTH assays in pediatric patients treated with peritoneal dialysis. Kidney Int. 63, 1801–1808 (2003).
Wesseling-Perry, K. et al. Response of different PTH assays to therapy with sevelamer or CaCO3 and active vitamin D sterols. Pediatr. Nephrol. 24, 1355–1361 (2009).
Slatopolsky, E. et al. A novel mechanism for skeletal resistance in uremia. Kidney Int. 58, 753–761 (2000).
Waller, S. C., Ridout, D., Cantor, T. & Rees, L. Parathyroid hormone and growth in children with chronic renal failure. Kidney Int. 67, 2338–2345 (2005).
Cantor, T., Yang, Z., Caraiani, N. & Ilamathi, E. Lack of comparability of intact parathyroid hormone measurements among commercial assays for end-stage renal disease patients: implication for treatment decisions. Clin. Chem. 52, 1771–1776 (2006).
Gardham, C. et al. Variability of parathyroid hormone and other markers of bone mineral metabolism in patients receiving hemodialysis. Clin. J. Am. Soc. Nephrol. 5, 1261–1267 (2010).
Kidney Disease: Improving Global Outcomes (KDIGO) CKD-MBD Work Group. KDIGO clinical practice guideline for the diagnosis, evaluation, prevention, and treatment of Chronic Kidney Disease-Mineral and Bone Disorder (CKD-MBD). Kidney Int. Suppl. 113, S1–S130 (2009).
National Kidney Foundation. K/DOQI Clinical Practice guidelines for bone metabolism and disease in children with chronic kidney disease. Am. J. Kidney Dis. 46 (Suppl. 1), 1–122 (2005).
Kuizon, B. D. et al. Diminished linear growth during intermittent calcitriol therapy in children undergoing CCPD. Kidney Int. 53, 205–211 (1998).
Schmitt, C. P., Ardissino, G., Testa, S., Claris-Appiani, A. & Mehls, O. Growth in children with chronic renal failure on intermittent versus daily calcitriol. Pediatr. Nephrol. 18, 440–444 (2003).
Waller, S. C., Ridout, D., Cantor, T. & Rees, L. Parathyroid hormone and growth in children with chronic renal failure. Kidney Int. 67, 2338–2345 (2005).
Waller, S., Shroff, R., Freemont, A. J. & Rees, L. Bone histomorphometry in children prior to commencing renal replacement therapy. Pediatr. Nephrol. 23, 1523–1529 (2008).
Cansick, J., Waller, S., Ridout, D. & Rees, L. Growth and PTH in prepubertal children on long-term dialysis. Pediatr. Nephrol. 22, 1349–1354 (2007).
Fischbach, M. et al. Daily on line haemodiafiltration promotes catch-up growth in children on chronic dialysis. Nephrol. Dial. Transplant. 25, 867–873 (2010).
Salusky, I. B. et al. Intermittent calcitriol therapy in secondary hyperparathyroidism: a comparison between oral and intraperitoneal administration. Kidney Int. 54, 907–914 (1998).
Bakkaloglu, S. A. et al. Value of the new bone classification system in pediatric renal osteodystrophy. Clin. J. Am. Soc. Nephrol. 5, 1860–1866 (2010).
Salusky, I. B. et al. Sevelamer controls parathyroid hormone-induced bone disease as efficiently as calcium carbonate without increasing serum calcium levels during therapy with active vitamin D sterols. J. Am. Soc. Nephrol. 16, 2501–2508 (2005).
Piscitelli, J., Cabansag, M. R. & Silverstein, D. M. Correlation among markers of renal osteodystrophy in pediatric hemodialysis patients. J. Pediatr. Endocrinol. Metab. 12, 879–886 (1999).
Christoforidis, A. et al. Comparative study of quantitative ultrasonography and dual-energy X-ray absorptiometry for evaluating renal osteodystrophy in children with chronic kidney disease. J. Bone Miner. Metab. 29, 321–327 (2011).
Siomou, E. et al. Serum osteoprotegerin, RANKL and fibroblast growth factor-23 in children with chronic kidney disease. Pediatr. Nephrol. 26, 1105–1114 (2011).
Ziólkowska, H. et al. Bone biopsy results and serum bone turnover parameters in uremic children. Acta Paediatr. 89, 666–671 (2000).
Fahrleitner-Pammer, A. et al. Bone markers predict cardiovascular events in chronic kidney disease. J. Bone Miner. Res. 23, 1850–1858 (2008).
Baber, U. et al. Non-traditional risk factors predict coronary calcification in chronic kidney disease in a population-based cohort. Kidney Int. 73, 615–621 (2008).
Mohandas, R. & Segal, M. S. Endothelial progenitor cells and endothelial vesicles—what is the significance for patients with chronic kidney disease. Blood Purif. 29, 158–162 (2010).
Bacchetta, J. et al. The influence of glomerular filtration rate and age on fibroblast growth factor 23 serum levels in pediatric chronic kidney disease. J. Clin. Endocrinol. Metab. 95, 1741–1748 (2010).
van Husen, M. et al. Fibroblast growth factor 23 and bone metabolism in children with chronic kidney disease. Kidney Int. 78, 200–206 (2010).
Isakova, T. et al. Fibroblast growth factor 23 is elevated before parathyroid hormone and phosphate in chronic kidney disease. Kidney Int. 79, 1370–1378 (2011).
Petkovich, M. & Jones, G. CYP24A1 and kidney disease. Curr. Opin. Nephrol. Hypertens. 20, 337–344 (2011).
Wesseling-Perry, K. et al. Calcitriol and doxercalciferol are equivalent in controlling bone turnover, suppressing parathyroid hormone, and increasing fibroblast growth factor-23 in secondary hyperparathyroidism. Kidney Int. 79, 112–119 (2011).
Larsson, T., Nisbeth, U., Ljunggren, O., Jüppner, H. & Jonsson, K. B. Circulating concentration of FGF-23 increases as renal function declines in patients with chronic kidney disease, but does not change in response to variation in phosphate intake in healthy volunteers. Kidney Int. 64, 2272–2279 (2003).
Gutiérrez, O. M. et al. Fibroblast growth factor 23 and mortality among patients undergoing hemodialysis. N. Engl. J. Med. 359, 584–592 (2008).
Wolf, M. et al. Elevated fibroblast growth factor 23 is a risk factor for kidney transplant loss and mortality. J. Am. Soc. Nephrol. 22, 956–966 (2011).
Gutiérrez, O. M. et al. Fibroblast growth factor 23 and left ventricular hypertrophy in chronic kidney disease. Circulation 119, 2545–2552 (2009).
Kanbay, M. et al. Fibroblast growth factor 23 and fetuin A are independent predictors for the coronary artery disease extent in mild chronic kidney disease. Clin. J. Am. Soc. Nephrol. 5, 1780–1786 (2010).
Manghat, P. et al. Association of bone turnover markers and arterial stiffness in pre-dialysis chronic kidney disease (CKD). Bone 48, 1127–1132 (2011).
Wesseling-Perry, K., Tsai, E. W., Ettenger, R. B., Jüppner, H. & Salusky, I. B. Mineral abnormalities and long-term graft function in pediatric renal transplant recipients: a role for FGF-23? Nephrol. Dial. Transplant. http://dx.doi.org/10.1093/ndt/gfr126.
Wesseling-Perry, K. et al. Relationship between plasma fibroblast growth factor-23 concentration and bone mineralization in children with renal failure on peritoneal dialysis. J. Clin. Endocrinol. Metab. 94, 511–517 (2009).
Russo, D. & Battaglia, Y. Clinical significance of FGF-23 in patients with CKD. Int. J. Nephrol. 2011, 364890 (2011).
Schönau, E. The peak bone mass concept: is it still relevant? Pediatr. Nephrol. 19, 825–831 (2004).
Weber, L. T. & Mehls, O. Limitations of dual x-ray absorptiometry in children with chronic kidney disease. Pediatr. Nephrol. 25, 3–5 (2010).
Bacchetta, J. et al. Bone assessment in children with chronic kidney disease: data from two new bone imaging techniques in a single-center pilot study. Pediatr. Nephrol. 26, 587–595 (2011).
Leonard, M. B. et al. Effects of sex, race, and puberty on cortical bone and the functional muscle bone unit in children, adolescents, and young adults. J. Clin. Endocrinol. Metab. 95, 1681–1689 (2010).
Ziólkowska, H. et al. Bone biopsy results and serum bone turnover parameters in uremic children. Acta Paediatr. 89, 666–671 (2000).
Salusky, I. B. et al. Biochemical markers of renal osteodystrophy in pediatric patients undergoing CAPD/CCPD. Kidney Int. 45, 253–258 (1994).
Glorieux, F. H. et al. Normative data for iliac bone histomorphometry in growing children. Bone 26, 103–109 (2000).
LaClair, R. E. et al. Prevalence of calcidiol deficiency in CKD: a cross-sectional study across latitudes in the United States. Am. J. Kidney Dis. 45, 1026–1033 (2005).
Hari, P. et al. Vitamin D insufficiency and effect of cholecalciferol in children with chronic kidney disease. Pediatr. Nephrol. 25, 2483–2488 (2010).
Ali, F. N., Arguelles, L. M., Langman, C. B. & Price, H. E. Vitamin D deficiency in children with chronic kidney disease: uncovering an epidemic. Pediatrics 123, 791–796 (2009).
Shroff, R. et al. Vitamin D deficiency is associated with short stature and may influence blood pressure control in paediatric renal transplant recipients. Pediatr. Nephrol. http://dx.doi.org/10.1007/s00467-011-1920-z.
Holick, M. F. Vitamin D deficiency. N. Engl. J. Med. 357, 266–281 (2007).
Matias, P. J. et al. Cholecalciferol supplementation in hemodialysis patients: effects on mineral metabolism, inflammation, and cardiac dimension parameters. Clin. J. Am. Soc. Nephrol. 5, 905–911 (2010).
Menon, S., Valentini, R. P., Hidalgo, G., Peschansky, L. & Mattoo, T. K. Vitamin D insufficiency and hyperparathyroidism in children with chronic kidney disease. Pediatr. Nephrol. 23, 1831–1836 (2008).
Shroff, R. et al. A bimodal association of vitamin D levels and vascular disease in children on dialysis. J. Am. Soc. Nephrol. 19, 1239–1246 (2008).
Jones, G. Pharmacokinetics of vitamin D toxicity. Am. J. Clin. Nutr. 88, 582S–586S (2008).
Teng, M. et al. Survival of patients undergoing hemodialysis with paricalcitol or calcitriol therapy. N. Engl. J. Med. 349, 446–456 (2003).
Tentori, F. et al. Mortality risk among hemodialysis patients receiving different vitamin D analogs. Kidney Int. 70, 1858–1865 (2006).
Nuijten, M., Andress, D. L., Marx, S. E., Curry, A. S. & Sterz, R. Cost effectiveness of paricalcitol versus a non-selective vitamin D receptor activator for secondary hyperparathyroidism in the UK: a chronic kidney disease markov model. Clin. Drug Investig. 30, 545–557 (2010).
Sprague, S. M., Llach, F., Amdahl, M., Taccetta, C. & Batlle, D. Paricalcitol versus calcitriol in the treatment of secondary hyperparathyroidism. Kidney Int. 63, 1483–1490 (2003).
Llach, F. et al. Suppression of parathyroid hormone secretion in hemodialysis patients by a novel vitamin D analogue: 19-nor-1,25-dihydroxyvitamin D2. Am. J. Kidney Dis. 32 (2 Suppl. 2), S48–S54 (1998).
Palmer, S. C. et al. Vitamin D compounds for people with chronic kidney disease requiring dialysis. Cochrane Database of Systematic Reviews Issue 7. Art. No.: CD005633. doi:10.1002/14651858.CD005633.pub2 (2009).
Palmer, S. C. et al. Vitamin D compounds for people with chronic kidney disease not requiring dialysis. Cochrane Database of Systematic Reviews Issue 7 Art. No.: CD008175 doi:10.1002/14651858.CD008175 (2009).
Finch, J. L., Brown, A. J. & Slatopolsky, E. Differential effects of 1,25-dihydroxy-vitamin D3 and 19-nor-1,25-dihydroxy-vitamin D2 on calcium and phosphorus resorption in bone. J. Am. Soc. Nephrol. 10, 980–985 (1999).
Brown, A. J., Finch, J. & Slatopolsky, E. Differential effects of 19-nor-1,25-dihydroxyvitamin D(2) and 1,25-dihydroxyvitamin D(3) on intestinal calcium and phosphate transport. J. Lab. Clin. Med. 139, 279–284 (2002).
Mizobuchi, M., Finch, J. L., Martin, D. R. & Slatopolsky, E. Differential effects of vitamin D receptor activators on vascular calcification in uremic rats. Kidney Int. 72, 709–715 (2007).
Becker, L. E. et al. Effect of paricalcitol and calcitriol on aortic wall remodeling in uninephrectomized ApoE knockout mice. Am. J. Physiol. Renal Physiol. 300, F772–F782 (2011).
Repo, J. M. et al. Paricalcitol aggravates perivascular fibrosis in rats with renal insufficiency and low calcitriol. Kidney Int. 72, 977–984 (2007).
Greenbaum, L. A. et al. Intravenous paricalcitol for treatment of secondary hyperparathyroidism in children on hemodialysis. Am. J. Kidney Dis. 49, 814–823 (2007).
Seeherunvong, W. et al. Paricalcitol versus calcitriol treatment for hyperparathyroidism in pediatric hemodialysis patients. Pediatr. Nephrol. 21, 1434–1439 (2006).
Tsuruoka, S., Wakaumi, M., Sugimoto, K., Saito, T. & Fujimura, A. Chronotherapy of high-dose active vitamin D3 in haemodialysis patients with secondary hyperparathyroidsm: a repeated dosing study. Br. J. Clin. Pharmacol. 55, 531–537 (2003).
Pieper, A. K. et al. A randomized crossover trial comparing sevelamer with calcium acetate in children with CKD. Am. J. Kidney Dis. 47, 625–635 (2006).
Gonzalez, E., Schomberg, J., Amin, N., Salusky, I. B. & Zaritsky, J. Sevelamer carbonate increases serum bicarbonate in pediatric dialysis patients. Pediatr. Nephrol. 25, 373–375 (2010).
Chertow, G. M., Burke, S. K. & Raggi, P. Sevelamer attenuates the progression of coronary and aortic calcification in hemodialysis patients. Kidney Int. 62, 245–252 (2002).
Kakuta, T. et al. Effect of sevelamer and calcium-based phosphate binders on coronary artery calcification and accumulation of circulating advanced glycation end products in hemodialysis patients. Am. J. Kidney Dis. 57, 422–431 (2011).
Block, G. A., Raggi, P., Bellasi, A., Kooienga, L. & Spiegel, D. M. Mortality effect of coronary calcification and phosphate binder choice in incident hemodialysis patients. Kidney Int. 71, 438–441 (2007).
Navaneethan, S. D., Palmer, S. C., Craig, J. C., Elder, G. J. & Strippoli, G. F. Benefits and harms of phosphate binders in CKD: a systematic review of randomized controlled trials. Am. J. Kidney Dis. 54, 619–637 (2009).
Hutchison, A. J., Barnett, M. E., Krause, R. J. & Siami, G. A. Lanthanum carbonate treatment, for up to 6 years, is not associated with adverse effects on the liver in patients with chronic kidney disease stage 5 receiving hemodialysis. Clin. Nephrol. 71, 286–295 (2009).
Lacour, B. et al. Chronic renal failure is associated with increased tissue deposition of lanthanum after 28-day oral administration. Kidney Int. 67, 1062–1069 (2005).
Davis, R. L. & Abraham, J. L. Lanthanum deposition in a dialysis patient. Nephrol. Dial. Transplant. 24, 3247–3250 (2009).
Spasovski, G. B. et al. Evolution of bone and plasma concentration of lanthanum in dialysis patients before, during 1 year of treatment with lanthanum carbonate and after 2 years of follow-up. Nephrol. Dial. Transplant. 21, 2217–2224 (2006).
Strippoli, G. F., Tong, A., Palmer, S. C., Elder, G. & Craig, J. C. Calcimimetics for secondary hyperparathyroidism in chronic kidney disease patients. Cochrane Database of Systematic Reviews Issue 18 Art. No.: CD006254 doi:10.1002/14651858.CD006254 (2006).
Meola, M., Petrucci, I. & Barsotti, G. Long-term treatment with cinacalcet and conventional therapy reduces parathyroid hyperplasia in severe secondary hyperparathyroidism. Nephrol. Dial. Transplant. 24, 982–989 (2009).
Raggi, P. et al. The ADVANCE study: a randomized study to evaluate the effects of cinacalcet plus low-dose vitamin D on vascular calcification in patients on hemodialysis. Nephrol. Dial. Transplant. 26, 1327–1339 (2011).
Muscheites, J. et al. Cinacalcet for secondary hyperparathyroidism in children with end-stage renal disease. Pediatr. Nephrol. 23, 1823–1829 (2008).
Silverstein, D. M. et al. Cinacalcet is efficacious in pediatric dialysis patients. Pediatr. Nephrol. 23, 1817–1822 (2008).
Bacchetta, J. et al. Precocious puberty and unlicensed paediatric drugs for severe hyperparathyroidism. Nephrol. Dial. Transplant. 24, 2595–2598 (2009).
European Medicines Agency European Assessment Report MIMPARA [online], (2009).
Wu, S., Palese, T., Mishra, O. P., Delivoria-Papadopoulos, M. & De Luca, F. Effects of Ca2+ sensing receptor activation in the growth plate. FASEB J. 18, 143–145 (2004).
Rodriguez, L., Cheng, Z., Chen, T. H., Tu, C. & Chang, W. Extracellular calcium and parathyroid hormone-related peptide signaling modulate the pace of growth plate chondrocyte differentiation. Endocrinology 146, 4597–4608 (2005).
Schmitt, C. P. et al. Intermittent administration of parathyroid hormone (1–37) improves growth and bone mineral density in uremic rats. Kidney Int. 57, 1484–1492 (2000).
Lee, K. et al. Parathyroid hormone-related peptide delays terminal differentiation of chondrocytes during endochondral bone development. Endocrinology 137, 5109–5118 (1996).
Lanske, B. et al. PTH/PTHrP receptor in early development and Indian hedgehog-regulated bone growth. Science 273, 663–666 (1996).
Garner, S. C., Pi, M., Tu, Q. & Quarles, L. D. Rickets in cation-sensing receptor-deficient mice: an unexpected skeletal phenotype. Endocrinology 142, 3996–4005 (2001).
Kos, C. H. et al. The calcium-sensing receptor is required for normal calcium homeostasis independent of parathyroid hormone. J. Clin. Invest. 111, 1021–1028 (2003).
Tu, Q. et al. Rescue of the skeletal phenotype in CasR-deficient mice by transfer onto the Gcm2 null background. J. Clin. Invest. 111, 1029–1037 (2003).
Mittelman, S. D. et al. A hypocalcemic child with a novel activating mutation of the calcium-sensing receptor gene: successful treatment with recombinant human parathyroid hormone. J. Clin. Endocrinol. Metab. 91, 2474–2479 (2006).
Burren, C. P., Curley, A., Christie, P., Rodda, C. P. & Thakker, R. V. A family with autosomal dominant hypocalcaemia with hypercalciuria (ADHH): mutational analysis, phenotypic variability and treatment challenges. J. Pediatr. Endocrinol. Metab. 18, 689–699 (2005).
Uckun-Kitapci, A., Underwood, L. E., Zhang, J. & Moats-Staats, B. A novel mutation (E767K) in the second extracellular loop of the calcium sensing receptor in a family with autosomal dominant hypocalcemia. Am. J. Med. Genet. 132, 125–129 (2005).
Nakagawa, K. et al. Cinacalcet does not affect longitudinal growth but increases body weight gain in experimental uraemia. Nephrol. Dial. Transplant. 23, 2761–2767 (2008).
Platt, C. et al. Middle-term use of cinacalcet in paediatric dialysis patients. Pediatr. Nephrol. 25, 143–148 (2010).
DrugLib.com Well-designed clinical trials related to Sensipar (cinacalcet) [online], (2011).
Henley, C. 3rd et al. Discovery of a calcimimetic with differential effects on parathyroid hormone and calcitonin secretion. J. Pharmacol. Exp. Ther. 337, 681–691 (2011).
Oh, J. et al. Stimulation of the calcium-sensing receptor stabilizes the podocyte cytoskeleton, improves cell survival, and reduces toxin-induced glomerulosclerosis. Kidney Int. 80, 483–492 (2011).
Finch, J. L. et al. Effect of paricalcitol and cinacalcet on serum phosphate, FGF-23, and bone in rats with chronic kidney disease. Am. J. Physiol. Renal Physiol. 298, F1315–F1322 (2010).
Cunningham, J., Locatelli, F. & Rodriguez, M. Secondary hyperparathyroidism: pathogenesis, disease progression, and therapeutic options. Clin. J. Am. Soc. Nephrol. 6, 913–921 (2011).
Schaefer, B. et al. Long-term control of parathyroid hormone and calcium-phosphate metabolism after parathyroidectomy in children with chronic kidney disease. Nephrol. Dial. Transplant. 25, 2590–2595 (2010).
Author information
Authors and Affiliations
Contributions
Both authors contributed equally to all aspects of this Review.
Corresponding author
Ethics declarations
Competing interests
C. P. Schmitt has been a consultant and speaker for Amgen and is a patent holder/applicant with Amgen. C. P. Schmitt has received grant/research support from Amgen and Fresenius. O. Mehls declares no competing interests.
Rights and permissions
About this article
Cite this article
Schmitt, C., Mehls, O. Mineral and bone disorders in children with chronic kidney disease. Nat Rev Nephrol 7, 624–634 (2011). https://doi.org/10.1038/nrneph.2011.139
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneph.2011.139
This article is cited by
-
Diagnosis and management of mineral and bone disorders in infants with CKD: clinical practice points from the ESPN CKD-MBD and Dialysis working groups and the Pediatric Renal Nutrition Taskforce
Pediatric Nephrology (2023)
-
Phase 1, single-dose study to assess the safety, tolerability, pharmacokinetics, and pharmacodynamics of etelcalcetide in pediatric patients with secondary hyperparathyroidism receiving hemodialysis
Pediatric Nephrology (2021)
-
Bone Disease in CKD in Children
Calcified Tissue International (2021)
-
Targeting optimal PD management in children: what have we learned from the IPPN registry?
Pediatric Nephrology (2021)
-
An open-label, single-dose study to evaluate the safety, tolerability, pharmacokinetics, and pharmacodynamics of cinacalcet in pediatric subjects aged 28 days to < 6 years with chronic kidney disease receiving dialysis
Pediatric Nephrology (2019)