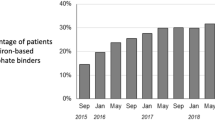
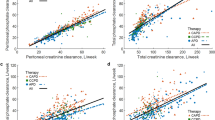
Abstract
The ideal serum level of phosphate in patients on dialysis, and the benefits of controlling levels of phosphate in serum remain unclear despite observational studies that associate phosphate levels with mortality. In the absence of robust data from trials, current guidelines are necessarily based on opinion. Oral phosphate binders are required by the majority of patients on dialysis, and all of these binders can control serum levels of phosphate to similar degrees. Patient preference and adherence to prescribed therapy is at least as important as the efficacy of the prescribed binder. Avoidance of calcium-containing binders has become accepted practice where the alternatives are affordable, but incontrovertible evidence in favor of this approach is lacking. Use of sevelamer and lanthanum avoids calcium loading, but at considerable financial cost and with no reliable patient outcome data to prove their value. Additional approaches to aid control of serum levels of phosphate include blockade of gastrointestinal phosphate absorption and possibly binding of salivary phosphate. Importantly, the role of phosphate control in determining patient outcomes must be quantified, which is likely to require a large randomized, controlled study of two levels of phosphate control. Without such a study we will continue to rely on observational data with all its uncertainties and potential to mislead.
Key Points
-
The ideal serum level of phosphate and the benefits of controlling phosphate levels in serum remain unclear, and available guidelines are largely based on opinion
-
All phosphate binders can control phosphate levels in serum to a similar degree; however, patient preference and adherence to prescribed therapy must be considered together with the efficacy of the prescribed binder
-
Most clinicians avoid calcium-containing binders where the alternatives are affordable, but evidence to support this strategy is limited
-
Calcium loading can be avoided with sevelamer and lanthanum, but these drugs are expensive and no reliable patient outcome data exist to prove their value
-
Alternative approaches to control of phosphate absorption include blockade of gastrointestinal phosphate and possibly binding of salivary phosphate
-
Randomized, controlled trials are required to determine the ideal range for control of phosphate in serum
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Cancer Research UK. Ovarian cancer survival statistics [online], (2011).
Ansell, D. et al. UK Renal Registry 12th Annual Report 117–144 (The Renal Association, Bristol, UK, 2009).
Block, G. A., Hulbert-Shearon, T. E., Levin, N. W. & Port, F. K. Association of serum phosphorus and calcium x phosphate product with mortality risk in chronic hemodialysis patients: a national study. Am. J. Kidney Dis. 31, 607–617 (1998).
Mathew, S. et al. The mechanism of phosphorus as a cardiovascular risk factor in CKD. J. Am. Soc. Nephrol. 19, 1092–1105 (2008).
Cozzolino, M., Ciceri, P., Volpi, E. M., Olivi, L. & Messa, P. G. Pathophysiology of calcium and phosphate metabolism impairment in chronic kidney disease. Blood Purif. 27, 338–344 (2009).
Hruska, K. A., Saab, G., Mathew, S. & Lund, R. Renal osteodystrophy, phosphate homeostasis, and vascular calcification. Semin. Dial. 20, 309–315 (2007).
Uribarri, J. Phosphorus additives in food and their effect in dialysis patients. Clin. J. Am. Soc. Nephrol. 4, 1290–1292 (2009).
Uribarri, J. Phosphorus homeostasis in normal health and in chronic kidney disease patients with special emphasis on dietary phosphorus intake. Semin. Dial. 20, 295–301 (2007).
Zisman, A. L. & Wolf, M. Recent advances in the rapidly evolving field of fibroblast growth factor 23 in chronic kidney disease. Curr. Opin. Nephrol. Hypertens. 19, 335–342 (2010).
Kooienga, L. Phosphorus balance with daily dialysis. Semin. Dial. 20, 342–345 (2007).
Kolek, O. I. et al. 1α, 25-Dihydroxyvitamin D3 upregulates FGF23 gene expression in bone: the final link in a renal-gastrointestinal-skeletal axis that controls phosphate transport. Am. J. Physiol. Gastrointest. Liver Physiol. 289, G1036–G1042 (2005).
Larsson, T., Nisbeth, U., Ljunggren, O., Juppner, H. & Jonsson, K. B. Circulating concentration of FGF-23 increases as renal function declines in patients with chronic kidney disease, but does not change in response to variation in phosphate intake in healthy volunteers. Kidney Int. 64, 2272–2279 (2003).
Gutiérrez, O. et al. Fibroblast growth factor-23 mitigates hyperphosphatemia but accentuates calcitriol deficiency in chronic kidney disease. J. Am. Soc. Nephrol. 16, 2205–2215 (2005).
Perwad, F. et al. Dietary and serum phosphorus regulate fibroblast growth factor 23 expression and 1,25-dihydroxyvitamin D metabolism in mice. Endocrinology 146, 5358–5364 (2005).
Antoniucci, D. M., Yamashita, T. & Portale, A. A. Dietary phosphorus regulates serum fibroblast growth factor-23 concentrations in healthy men. J. Clin. Endocrinol. Metab. 91, 3144–3149 (2006).
Liu, S. et al. Fibroblast growth factor 23 is a counter-regulatory phosphaturic hormone for vitamin D. J. Am. Soc. Nephrol. 17, 1305–1315 (2006).
Imanishi, Y. et al. FGF-23 in patients with end-stage renal disease on hemodialysis. Kidney Int. 65, 1943–1946 (2004).
Shigematsu, T. et al. Possible involvement of circulating fibroblast growth factor 23 in the development of secondary hyperparathyroidism associated with renal insufficiency. Am. J. Kidney Dis. 44, 250–256 (2004).
Shimada, T. et al. Circulating fibroblast growth factor 23 in patients with end-stage renal disease treated by peritoneal dialysis is intact and biologically active. J. Clin. Endocrinol. Metab. 95, 578–585 (2009).
Fliser, D. et al. Fibroblast growth factor 23 (FGF23) predicts progression of chronic kidney disease: the Mild to Moderate Kidney Disease (MMKD) Study. J. Am. Soc. Nephrol. 18, 2600–2608 (2007).
Hsu, H. J. & Wu, M. S. Fibroblast growth factor 23: a possible cause of left ventricular hypertrophy in hemodialysis patients. Am. J. Med. Sci. 337, 116–122 (2009).
Gutiérrez, O. M. et al. Fibroblast growth factor 23 and left ventricular hypertrophy in chronic kidney disease. Circulation 119, 2545–2552 (2009).
Gutiérrez, O. M. et al. Fibroblast growth factor 23 and mortality among patients undergoing hemodialysis. N. Engl. J. Med. 359, 584–592 (2008).
Ben-Dov, I. Z. et al. The parathyroid is a target organ for FGF23 in rats. J. Clin. Invest. 117, 4003–4008 (2007).
Komaba, H. & Fukagawa, M. FGF23-parathyroid interaction: implications in chronic kidney disease. Kidney Int. 77, 292–298 (2009).
Koh, N. et al. Severely reduced production of klotho in human chronic renal failure kidney. Biochem. Biophys. Res. Commun. 280, 1015–1020 (2001).
Komaba, H. et al. Depressed expression of Klotho and FGF receptor 1 in hyperplastic parathyroid glands from uremic patients. Kidney Int. 77, 232–238 (2010).
Galitzer, H., Ben-Dov, I. Z., Silver, J. & Naveh-Many, T. Parathyroid cell resistance to fibroblast growth factor 23 in secondary hyperparathyroidism of chronic kidney disease. Kidney Int. 77, 211–218 (2010).
Wang, Z. et al. Protein carbamylation links inflammation, smoking, uremia and atherogenesis. Nat. Med. 13, 1176–1184 (2007).
Adragão, T. et al. A plain X-ray vascular calcification score is associated with arterial stiffness and mortality in dialysis patients. Nephrol. Dial. Transplant. 24, 997–1002 (2009).
Adragão, T. et al. A simple vascular calcification score predicts cardiovascular risk in haemodialysis patients. Nephrol. Dial. Transplant. 19, 1480–1488 (2004).
Isakova, T. et al. Phosphorus binders and survival on hemodialysis. J. Am. Soc. Nephrol. 20, 388–396 (2009).
Wills, M. R. & Savory, J. Aluminium poisoning: dialysis encephalopathy, osteomalacia, and anaemia. Lancet 2, 29–34 (1983).
González-Revaldería, J. et al. Biochemical and hematological changes in low-level aluminum intoxication. Clin. Chem. Lab. Med. 38, 221–225 (2000).
Becaria, A., Campbell, A. & Bondy, S. C. Aluminum as a toxicant. Toxicol. Ind. Health 18, 309–320 (2002).
Rüster, M., Abendroth, K., Lehmann, G. & Stein, G. Aluminum deposition in the bone of patients with chronic renal failure—detection of aluminum accumulation without signs of aluminum toxicity in bone using acid solochrome azurine. Clin. Nephrol. 58, 305–312 (2002).
Jaffe, J. A., Liftman, C. & Glickman, J. D. Frequency of elevated serum aluminum levels in adult dialysis patients. Am. J. Kidney Dis. 46, 316–319 (2005).
Andrade, L. G. et al. Dialysis encephalopathy secondary to aluminum toxicity, diagnosed by bone biopsy. Nephrol. Dial. Transplant. 20, 2581–2582 (2005).
Feith, G. W. Oral phosphate binders in patients with kidney failure. N. Engl. J. Med. 363, 989–990 (2010).
Slatopolsky, E., Weerts, C., Stokes, T., Windus, D. & Delmez, J. Alternative phosphate binders in dialysis patients: calcium carbonate. Semin. Nephrol. 6, 35–41 (1986).
Schaefer, K., Umlauf, E. & von Herrath, D. Reduced risk of hypercalcemia for hemodialysis patients by administering calcitriol at night. Am. J. Kidney Dis. 19, 460–464 (1992).
Stamatakis, M. K., Alderman, J. M. & Meyer-Stout, P. J. Influence of pH on in vitro disintegration of phosphate binders. Am. J. Kidney Dis. 32, 808–812 (1998).
Sheikh, M. S. et al. Reduction of dietary phosphorus absorption by phosphorus binders. A theoretical, in vitro, and in vivo study. J. Clin. Invest. 83, 66–73 (1989).
Pflanz, S., Henderson, I. S., McElduff, N. & Jones, M. C. Calcium acetate versus calcium carbonate as phosphate-binding agents in chronic haemodialysis. Nephrol. Dial. Transplant. 9, 1121–1124 (1994).
Harris, D. C. & Yuill, L. Calcium alginate versus aluminum hydroxide in patients on hemodialysis. Nephron 65, 324–325 (1993).
Passlick, J., Wilhelm, M., Busch, T., Grabensee, B. & Ohnesorge, F. K. Calcium alginate, an aluminum-free phosphate binder, in patients on CAPD. Clin. Nephrol. 32, 96–100 (1989).
Takahashi, N. et al. Effect of histamine H2-receptor antagonist on the phosphorus-binding abilities of calcium carbonate and calcium lactate in hemodialysis patients. J. Am. Soc. Nephrol. 10, 1090–1094 (1999).
Birck, R., Zimmermann, E., Wassmer, S., Nowack, R. & van der Woude, F. J. Calcium ketoglutarate versus calcium acetate for treatment of hyperphosphataemia in patients on maintenance haemodialysis: a cross-over study. Nephrol. Dial. Transplant. 14, 1475–1479 (1999).
Macia, M. & Coronel, F. Serum phosphate control: what about calcium salts of keto-amino acids? Nephrol. Dial. Transplant. 10, 2159–2160 (1995).
Schaefer, K., von Herrath, D., Asmus, G. & Umlauf, E. The beneficial effect of ketoacids on serum phosphate and parathyroid hormone in patients with chronic uremia. Clin. Nephrol. 30, 93–96 (1988).
Nilsson, P., Johansson, S. G. & Danielson, B. G. Magnesium studies in hemodialysis patients before and after treatment with low dialysate magnesium. Nephron 37, 25–29 (1984).
Hutchison, A. J. et al. Hypercalcaemia, hypermagnesaemia, hyperphosphataemia and hyperaluminaemia in CAPD: improvement in serum biochemistry by reduction in dialysate calcium and magnesium concentrations. Nephron 72, 52–58 (1996).
Hutchison, A. J., Merchant, M., Boulton, H. F., Hinchcliffe, R. & Gokal, R. Calcium and magnesium mass transfer in peritoneal dialysis patients using 1.25 mmol/L calcium, 0.25 mmol/L magnesium dialysis fluid. Perit. Dial. Int. 13, 219–223 (1993).
Meema, H. E., Oreopoulos, D. G. & Rapoport, A. Serum magnesium level and arterial calcification in end-stage renal disease. Kidney Int. 32, 388–394 (1987).
Montezano, A. C. et al. Vascular smooth muscle cell differentiation to an osteogenic phenotype involves TRPM7 modulation by magnesium. Hypertension 56, 453–462 (2010).
Gorgels, T. G. et al. Dietary magnesium, not calcium, prevents vascular calcification in a mouse model for pseudoxanthoma elasticum. J. Mol. Med. 88, 467–475 (2010).
Turgut, F. et al. Magnesium supplementation helps to improve carotid intima media thickness in patients on hemodialysis. Int. Urol. Nephrol. 40, 1075–1082 (2008).
Autissier, V., Damment, S. J. & Henderson, R. A. Relative in vitro efficacy of the phosphate binders lanthanum carbonate and sevelamer hydrochloride. J. Pharm. Sci. 96, 2818–2827 (2007).
Damment, S. J. & Pennick, M. Clinical pharmacokinetics of the phosphate binder lanthanum carbonate. Clin. Pharmacokinet. 47, 553–563 (2008).
Damment, S. J. & Pennick, M. Systemic lanthanum is excreted in the bile of rats. Toxicol. Lett. 171, 69–77 (2007).
Persy, V. P., Behets, G. J., Bervoets, A. R., De Broe, M. E. & D'Haese, P. C. Lanthanum: a safe phosphate binder. Semin. Dial. 19, 195–199 (2006).
Altmann, P., Barnett, M. E. & Finn, W. F. Cognitive function in stage 5 chronic kidney disease patients on hemodialysis: no adverse effects of lanthanum carbonate compared with standard phosphate-binder therapy. Kidney Int. 71, 252–259 (2007).
Albaaj, F. & Hutchison, A. J. Lanthanum carbonate for the treatment of hyperphosphataemia in renal failure and dialysis patients. Expert Opin. Pharmacother. 6, 319–328 (2005).
Goldsmith, D. R., Scott, L. J., Cvetkovic, R. S. & Plosker, G. L. Sevelamer hydrochloride: a review of its use for hyperphosphataemia in patients with end-stage renal disease on haemodialysis. Drugs 68, 85–104 (2008).
Burke, S., Amin, N., Incerti, C., Plone, M. & Watson, N. Sevelamer hydrochloride (Renagel), a nonabsorbed phosphate-binding polymer, does not interfere with digoxin or warfarin pharmacokinetics. J. Clin. Pharmacol. 41, 193–198 (2001).
Burke, S. K., Slatopolsky, E. A. & Goldberg, D. I. RenaGel, a novel calcium- and aluminium-free phosphate binder, inhibits phosphate absorption in normal volunteers. Nephrol. Dial. Transplant. 12, 1640–1644 (1997).
Wrong, O. & Harland, C. Sevelamer. Nephrol. Dial. Transplant. 23, 2108 (2008).
Plone, M. A., Petersen, J. S., Rosenbaum, D. P. & Burke, S. K. Sevelamer, a phosphate-binding polymer, is a non-absorbed compound. Clin. Pharmacokinet. 41, 517–523 (2002).
Rosenbaum, D. P., Holmes-Farley, S. R., Mandeville, W. H., Pitruzzello, M. & Goldberg, D. I. Effect of RenaGel, a non-absorbable, cross-linked, polymeric phosphate binder, on urinary phosphorus excretion in rats. Nephrol. Dial. Transplant. 12, 961–964 (1997).
Burke, S. K., Amin, N. S., Incerti, C., Plone, M. A. & Lee, J. W. Sevelamer hydrochloride (Renagel), a phosphate-binding polymer, does not alter the pharmacokinetics of two commonly used antihypertensives in healthy volunteers. J. Clin. Pharmacol. 41, 199–205 (2001).
Kays, M. B., Overholser, B. R., Mueller, B. A., Moe, S. M. & Sowinski, K. M. Effects of sevelamer hydrochloride and calcium acetate on the oral bioavailability of ciprofloxacin. Am. J. Kidney Dis. 42, 1253–1259 (2003).
Pieper, A. K. et al. The effect of sevelamer on the pharmacokinetics of cyclosporin A and mycophenolate mofetil after renal transplantation. Nephrol. Dial. Transplant. 19, 2630–2633 (2004).
Delmez, J. et al. A randomized, double-blind, crossover design study of sevelamer hydrochloride and sevelamer carbonate in patients on hemodialysis. Clin. Nephrol. 68, 386–391 (2007).
Pai, A. B. & Shepler, B. M. Comparison of sevelamer hydrochloride and sevelamer carbonate: risk of metabolic acidosis and clinical implications. Pharmacotherapy 29, 554–561 (2009).
Fan, S. et al. A randomized, crossover design study of sevelamer carbonate powder and sevelamer hydrochloride tablets in chronic kidney disease patients on haemodialysis. Nephrol. Dial. Transplant. 24, 3794–3799 (2009).
Chertow, G. M. et al. Poly[allylamine hydrochloride] (RenaGel): a noncalcemic phosphate binder for the treatment of hyperphosphatemia in chronic renal failure. Am. J. Kidney Dis. 29, 66–71 (1997).
Al-Baaj, F., Speake, M. & Hutchison, A. J. Control of serum phosphate by oral lanthanum carbonate in patients undergoing haemodialysis and continuous ambulatory peritoneal dialysis in a short-term, placebo-controlled study. Nephrol. Dial. Transplant. 20, 775–782 (2005).
McIntyre, C. W. et al. Iron-magnesium hydroxycarbonate (fermagate): a novel non-calcium-containing phosphate binder for the treatment of hyperphosphatemia in chronic hemodialysis patients. Clin. J. Am. Soc. Nephrol. 4, 401–409 (2009).
Locatelli, F. et al. Effect of MCI-196 on serum phosphate and cholesterol levels in haemodialysis patients with hyperphosphataemia: a double-blind, randomized, placebo-controlled study. Nephrol. Dial. Transplant. 25, 574–581 (2010).
Johnson, D. W. Sevelamer versus calcium-based phosphate binders in chronic kidney disease: what should we conclude from the evidence to date? Nephrol. Dial. Transplant. 24, 2970–2972 (2009).
Navaneethan, S. D., Palmer, S. C., Craig, J. C., Elder, G. J. & Strippoli, G. F. Benefits and harms of phosphate binders in CKD: a systematic review of randomized controlled trials. Am. J. Kidney Dis. 54, 619–637 (2009).
Chertow, G. M., Burke, S. K. & Raggi, P. Sevelamer attenuates the progression of coronary and aortic calcification in hemodialysis patients. Kidney Int. 62, 245–252 (2002).
Jamal, S. A., Fitchett, D., Lok, C. E., Mendelssohn, D. C. & Tsuyuki, R. T. The effects of calcium-based versus non-calcium-based phosphate binders on mortality among patients with chronic kidney disease: a meta-analysis. Nephrol. Dial. Transplant. 24, 3168–3174 (2009).
Sprague, S. M. et al. Lanthanum carbonate vs. sevelamer hydrochloride for the reduction of serum phosphorus in hemodialysis patients: a crossover study. Clin. Nephrol. 72, 252–258 (2009).
Arenas, M. D. et al. A comparative study of 2 new phosphate binders (sevelamer and lanthanum carbonate) in routine clinical practice. J. Nephrol. 23, 683–692 (2010).
Suki, W. N. et al. Effects of sevelamer and calcium-based phosphate binders on mortality in hemodialysis patients. Kidney Int. 72, 1130–1137 (2007).
Wilson, R., Zhang, P., Smyth, M. & Pratt, R. Assessment of survival in a 2-year comparative study of lanthanum carbonate versus standard therapy. Curr. Med. Res. Opin. 25, 3021–3028 (2009).
[No authors listed] KDIGO clinical practice guideline for the diagnosis, evaluation, prevention, and treatment of Chronic Kidney Disease-Mineral and Bone Disorder (CKD-MBD). Kidney Int. Suppl. 76, S1–S130 (2009).
Parham, R., Riley, S., Hutchison, A. & Horne, R. Patients' satisfaction with information about phosphate-binding medication. J. Ren. Care 35 (Suppl. 1), 86–93 (2009).
Chiu, Y. W. et al. Pill burden, adherence, hyperphosphatemia, and quality of life in maintenance dialysis patients. Clin. J. Am. Soc. Nephrol. 4, 1089–1096 (2009).
Arenas, M. D. et al. Phosphorus binders: preferences of patients on haemodialysis and its impact on treatment compliance and phosphorus control [Spanish]. Nefrologia 30, 522–530 (2010).
Hutchison, A. J. & Laville, M. Switching to lanthanum carbonate monotherapy provides effective phosphate control with a low tablet burden. Nephrol. Dial. Transplant. 23, 3677–3684 (2008).
Hutchison, A. J. et al. Long-term efficacy and tolerability of lanthanum carbonate: results from a 3-year study. Nephron Clin. Pract. 102, c61–c71 (2006).
Hutchison, A. J., Barnett, M. E., Krause, R. & Siami, G. A. Lanthanum carbonate treatment, for up to 6 years, is not associated with adverse effects on the liver in patients with chronic kidney disease stage 5 receiving hemodialysis. Clin. Nephrol. 71, 286–295 (2009).
Hutchison, A. J., Barnett, M. E., Krause, R., Kwan, J. T. & Siami, G. A. Long-term efficacy and safety profile of lanthanum carbonate: results for up to 6 years of treatment. Nephron Clin. Pract. 110, c15–c23 (2008).
[No authors listed] Sevelamer: constipation and occlusion. Complications, sometimes fatal. Prescrire Int. 17, 111 (2008).
Madan, P., Bhayana, S., Chandra, P. & Hughes, J. I. Lower gastrointestinal bleeding: association with sevelamer use. World J. Gastroenterol. 14, 2615–2616 (2008).
Friedman, E. A. Calcium-based phosphate binders are appropriate in chronic renal failure. Clin. J. Am. Soc. Nephrol. 1, 704–709 (2006).
Moe, S. M. & Chertow, G. M. The case against calcium-based phosphate binders. Clin. J. Am. Soc. Nephrol. 1, 697–703 (2006).
Bushinsky, D. A. Phosphate binders: hold the calcium? Clin. J. Am. Soc. Nephrol. 1, 695–696 (2006).
Spiegel, D. M. The role of magnesium binders in chronic kidney disease. Semin. Dial. 20, 333–336 (2007).
O'Donovan, R., Baldwin, D., Hammer, M., Moniz, C. & Parsons, V. Substitution of aluminium salts by magnesium salts in control of dialysis hyperphosphataemia. Lancet 1, 880–882 (1986).
Spiegel, D. M. & Farmer, B. Long-term effects of magnesium carbonate on coronary artery calcification and bone mineral density in hemodialysis patients: a pilot study. Hemodial. Int. 13, 453–459 (2009).
Spiegel, D. M., Farmer, B., Smits, G. & Chonchol, M. Magnesium carbonate is an effective phosphate binder for chronic hemodialysis patients: a pilot study. J. Ren. Nutr. 17, 416–422 (2007).
Baradaran, A. & Nasri, H. Correlation of serum magnesium with serum parathormone levels in patients on regular hemodialysis. Saudi J. Kidney Dis. Transpl. 17, 344–350 (2006).
Wei, M., Esbaei, K., Bargman, J. & Oreopoulos, D. G. Relationship between serum magnesium, parathyroid hormone, and vascular calcification in patients on dialysis: a literature review. Perit. Dial. Int. 26, 366–373 (2006).
Navarro, J. F., Mora, C. & Garcia, J. Serum magnesium and parathyroid hormone levels in dialysis patients. Kidney Int. 57, 2654 (2000).
de Francisco, A. L. et al. Evaluation of calcium acetate/magnesium carbonate as a phosphate binder compared with sevelamer hydrochloride in haemodialysis patients: a controlled randomized study (CALMAG study) assessing efficacy and tolerability. Nephrol. Dial. Transplant. 25, 3707–3717 (2010).
Hutchison, A. J. et al. Efficacy, tolerability, and safety of lanthanum carbonate in hyperphosphatemia: a 6-month, randomized, comparative trial versus calcium carbonate. Nephron Clin. Pract. 100, c8–c19 (2005).
Finn, W. F. & SPD 405–307 Lanthanum Study Group. Lanthanum carbonate versus standard therapy for the treatment of hyperphosphatemia: safety and efficacy in chronic maintenance hemodialysis patients. Clin. Nephrol. 65, 191–202 (2006).
Hutchison, A. J. et al. Long-term efficacy and tolerability of lanthanum carbonate: results from a 3-year study. Nephron Clin. Pract. 102, c61–c71 (2006).
Molony, D. A. & Murthy, B. Accumulation of metals and minerals from phosphate binders. Blood Purif. 23 (Suppl. 1), 2–11 (2005).
Lacour, B. et al. Chronic renal failure is associated with increased tissue deposition of lanthanum after 28-day oral administration. Kidney Int. 67, 1062–1069 (2005).
Damment, S. J. Enlightenment on liver lanthanum exposure. Kidney Int. 70, 1372–1373 (2006).
D'Haese, P. C. et al. A multicenter study on the effects of lanthanum carbonate (Fosrenol) and calcium carbonate on renal bone disease in dialysis patients. Kidney Int. Suppl. 63, S73–S78 (2003).
Brezina, B., Qunibi, W. Y. & Nolan, C. R. Acid loading during treatment with sevelamer hydrochloride: mechanisms and clinical implications. Kidney Int. Suppl. S39–S45 (2004).
Ketteler, M. et al. Efficacy and tolerability of sevelamer carbonate in hyperphosphatemic patients who have chronic kidney disease and are not on dialysis. Clin. J. Am. Soc. Nephrol. 3, 1125–1130 (2008).
Pierce, D. et al. The effect of sevelamer carbonate and lanthanum carbonate on the pharmacokinetics of oral calcitriol. Nephrol. Dial. Transplant. 26, 1615–1621 (2010).
Block, G. A., Brillhart, S. L., Persky, M. S., Amer, A. & Slade, A. J. Efficacy and safety of SBR759, a new iron-based phosphate binder. Kidney Int. 77, 897–903 (2010).
Katai, K. et al. Nicotinamide inhibits sodium-dependent phosphate cotransport activity in rat small intestine. Nephrol. Dial. Transplant. 14, 1195–1201 (1999).
Takahashi, Y. et al. Nicotinamide suppresses hyperphosphatemia in hemodialysis patients. Kidney Int. 65, 1099–1104 (2004).
Young, D. O., Cheng, S. C., Delmez, J. A. & Coyne, D. W. The effect of oral niacinamide on plasma phosphorus levels in peritoneal dialysis patients. Perit. Dial. Int. 29, 562–567 (2009).
Knip, M. et al. Safety of high-dose nicotinamide: a review. Diabetologia 43, 1337–1345 (2000).
Kirchner, S. et al. Luminal fructose inhibits rat intestinal sodium-phosphate cotransporter gene expression and phosphate uptake. Am. J. Clin. Nutr. 87, 1028–1038 (2008).
Hilfiker, H. et al. Characterization of a murine type II sodium-phosphate cotransporter expressed in mammalian small intestine. Proc. Natl Acad. Sci. USA 95, 14564–14569 (1998).
White, K. E., Biber, J., Murer, H. & Econs, M. J. Chromosomal localization of two human genes involved in phosphate homeostasis: the type IIb sodium-phosphate cotransporter and stanniocalcin-2. Somat. Cell Mol. Genet. 24, 357–362 (1998).
Shibasaki, Y. et al. Targeted deletion of the tybe IIb Na(+)-dependent Pi-co-transporter, NaPi-IIb, results in early embryonic lethality. Biochem. Biophys. Res. Commun. 381, 482–486 (2009).
Sabbagh, Y. et al. Intestinal npt2b plays a major role in phosphate absorption and homeostasis. J. Am. Soc. Nephrol. 20, 2348–2358 (2009).
Marks, J. et al. Intestinal phosphate absorption and the effect of vitamin D: a comparison of rats with mice. Exp. Physiol. 91, 531–537 (2006).
Savica, V. et al. Salivary phosphate-binding chewing gum reduces hyperphosphatemia in dialysis patients. J. Am. Soc. Nephrol. 20, 639–644 (2009).
Arenas, M. D. et al. Challenge of phosphorus control in hemodialysis patients: a problem of adherence? J. Nephrol. 23, 525–534 (2010).
[No authors listed] The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N. Engl. J. Med. 329, 977–986 (1993).
Acknowledgements
C. P. Vega, University of California, Irvine, CA, is the author of and is solely responsible for the content of the learning objectives, questions and answers of the Medscape, LLC-accredited continuing medical education activity associated with this article.
Author information
Authors and Affiliations
Contributions
All authors contributed equally to researching data for the article, discussion of the content and reviewing and editing the manuscript before submission. A. J. Hutchison wrote the article.
Corresponding author
Ethics declarations
Competing interests
A. J. Hutchison has received research grants from and previously been a consultant for Amgen. He is a consultant for Fresenius Healthcare. A. J. Hutchison has received research grants from, is a speaker for, and was previously a consultant to, Shire Pharmaceuticals. The other authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Hutchison, A., Smith, C. & Brenchley, P. Pharmacology, efficacy and safety of oral phosphate binders. Nat Rev Nephrol 7, 578–589 (2011). https://doi.org/10.1038/nrneph.2011.112
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneph.2011.112
This article is cited by
-
Sucroferric oxyhydroxide for hyperphosphatemia: a review of real-world evidence
Journal of Nephrology (2022)
-
Safety and efficacy of sucroferric oxyhydroxide in pediatric patients with chronic kidney disease
Pediatric Nephrology (2021)
-
A randomized controlled trial of different serum phosphate ranges in subjects on hemodialysis
BMC Nephrology (2019)
-
Efficacy and safety of nicotinamide in the management of hyperphosphatemia in pediatric patients on regular hemodialysis
Pediatric Nephrology (2016)
-
Effect of oral ferric citrate on serum phosphorus in hemodialysis patients: multicenter, randomized, double-blind, placebo-controlled study
Journal of Nephrology (2015)