Abstract
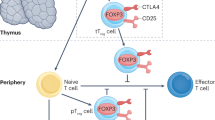
Several types of T cells with immunosuppressive properties have been identified, but FOXP3+ regulatory T (TREG) cells have emerged as a dominant cell type; they are critically involved in the induction and maintenance of immune tolerance. Manipulation of this cell type for the induction of transplant tolerance including renal transplant tolerance has attracted considerable attention. Studies in this area have demonstrated unexpected complexities, and attempts to translate TREG cells towards clinical utility have met with unanticipated difficulties. In this Review, a broad overview is provided on recent progress in the study of TREG cells, focusing on challenges, opportunities, and emerging approaches in exploiting TREG cells for the induction of transplant tolerance.
Key Points
-
Regulatory T (TREG) cells are indispensable in transplant tolerance
-
TREG cells are heterogeneous and are not inherently stable; their suppressive programs can be turned off or they may be reprogrammed to become T-effector cells
-
Some effector cell types, such as memory T cells, can evade TREG-cell-mediated suppression
-
Mechanisms that regulate the in vivo induction and stability of TREG cells are poorly defined
-
The mechanism by which FOXP3+ TREG cells interact with other types of regulatory cells in transplant tolerance is unknown
-
Potential complications of immunodeficiency exist from nonspecific expansion of TREG cells
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Lechler, R., Garden, O. A. & Turka, L. A. The complementary roles of deletion and regulation in transplantation tolerance. Nat. Rev. Immunol. 3, 147–158 (2003).
Strom, T. B. & Koulmanda, M. Recently discovered T cell subsets cannot keep their commitments. J. Am. Soc. Nephrol. 20, 1677–1680 (2009).
Zhou, X., Bailey-Bucktrout, S., Jeker, L. T. & Bluestone, J. A. Plasticity of CD4(+)Foxp3(+) T cells. Curr. Opin. Immunol. 21, 281–285 (2009).
Vu, M. D. et al. OX40 costimulation turns off Foxp3+ Tregs. Blood 110, 2501–2510 (2007).
Sakaguchi, S. Naturally arising CD4+ regulatory T cells for immunologic self-tolerance and negative control of immune responses. Annu. Rev. Immunol. 22, 531–562 (2004).
Groux, H. et al. A CD4+ T-cell subset inhibits antigen-specific T-cell responses and prevents colitis. Nature 389, 737–742 (1997).
Zhang, Z. X., Yang, L., Young, K. J., DuTemple, B. & Zhang, L. Identification of a previously unknown antigen-specific regulatory T cells and its mechanism of suppression. Nat. Med. 6, 782–789 (2000).
Seino, K.-I. et al. Requirement for natural killer T (NKT) cells in the induction of allograft tolerance. Proc. Natl Acad. Sci. USA 98, 2577–2581 (2001).
Wood, K. J. & Sakaguchi, S. Regulatory T cells in transplantation tolerance. Nat. Rev. Immunol. 3, 199–210 (2003).
Hori, S., Nomura, T. & Sakaguchi, S. Control of regulatory T cell development by the transcription factor Foxp3. Science 299, 1057–1061 (2003).
Setoguchi, R., Hori, S., Takahashi, T. & Sakaguchi, S. Homeostatic maintenance of natural Foxp3+ CD25+ CD4+ regulatory T cells by interleukin (IL)-2 and induction of autoimmune disease by IL-2 neutralization. J. Exp. Med. 201, 723–735 (2005).
Curotto de Lafaille, M. A. & Lafaille, J. J. Natural and adaptive foxp3+ regulatory T cells: more of the same or a division of labor? Immunity 30, 626–635 (2009).
Molinero, L. L. et al. CARMA1 controls an early checkpoint in the thymic development of FoxP3+ regulatory T cells. J. Immunol. 182, 6736–6743 (2009).
Gao, W. et al. Contrasting effects of cyclosporine and rapamycin on de novo generation of antigen-specific regulatory T cells. Am. J. Transplant. 7, 1722–1732 (2007).
Liu, Y. et al. A critical function for TGF-beta signaling in the development of natural CD4+CD25+Foxp3+ regulatory T cells. Nat. Immunol. 9, 632–640 (2008).
Chen, W. et al. Conversion of peripheral CD4+CD25- naive T cells to CD4+CD25+ regulatory T cells by TGF-β induction of transcription factor Foxp3. J. Exp. Med. 198, 1875–1886 (2003).
Fontenot, J. D. et al. Regulatory T cell lineage specification by the forkhead transcription factor foxp3. Immunity 22, 329–341 (2005).
Waldmann, H. Transplantation tolerance—where do we stand? Nat. Med. 5, 1245–1248 (1999).
Benson, M. J., Pino-Lagos, K., Rosemblatt, M. & Noelle, R. J. All-trans retinoic acid mediates enhanced T reg cell growth, differentiation, and gut homing in the face of high levels of co-stimulation. J. Exp. Med. 204, 1765–1774 (2007).
Gao, W. et al. Treg versus Th17 lymphocyte lineages are cross-regulated by LIF versus IL-6. Cell Cycle 8, 1444–1450 (2009).
Francisco, L. M. et al. PD-L1 regulates the development, maintenance, and function of induced regulatory T cells. J. Exp. Med. 206, 3015–3029 (2009).
Wei, J. et al. Antagonistic nature of T helper 1/2 developmental programs in opposing peripheral induction of Foxp3+ regulatory T cells. Proc. Natl Acad. Sci. USA 104, 18169–18174 (2007).
Haxhinasto, S., Mathis, D. & Benoist, C. The AKT-mTOR axis regulates de novo differentiation of CD4+Foxp3+ cells. J. Exp. Med. 205, 565–574 (2008).
Lal, G. & Bromberg, J. S. Epigenetic mechanisms of regulation of Foxp3 expression. Blood 114, 3727–3735 (2009).
Wong, J., Mathis, D. & Benoist, C. TCR-based lineage tracing: no evidence for conversion of conventional into regulatory T cells in response to a natural self-antigen in pancreatic islets. J. Exp. Med. 204, 2039–2045 (2007).
Shevach, E. M. Mechanisms of foxp3+ T regulatory cell-mediated suppression. Immunity 30, 636–645 (2009).
D'Alessio, F. R. et al. CD4+CD25+Foxp3+ Tregs resolve experimental lung injury in mice and are present in humans with acute lung injury. J. Clin. Invest. 119, 2898–2913 (2009).
Joffre, O. et al. Prevention of acute and chronic allograft rejection with CD4+CD25+Foxp3+ regulatory T lymphocytes. Nat. Med. 14, 88–92 (2008).
Deaglio, S. et al. Adenosine generation catalyzed by CD39 and CD73 expressed on regulatory T cells mediates immune suppression. J. Exp. Med. 204, 1257–1265 (2007).
Wing, K. et al. CTLA-4 control over Foxp3+ regulatory T cell function. Science 322, 271–275 (2008).
Bettni, M. & Vignali, D. A. Regulatory T cells and inhibitory cytokines in autoimmunity. Curr. Opin. Immunol. 21, 612–618 (2009).
Collison, L. W. et al. The inhibitory cytokine IL-35 contributes to regulatory T cell function. Nature 450, 566–569 (2007).
You, S. et al. Adaptive TGF-beta-dependent regulatory T cells control autoimmune diabetes and are a privileged target of anti-CD3 antibody treatment. Proc. Natl Acad. Sci. USA 104, 6335–6340 (2007).
Veldhoen, M., Hocking, R. J., Atkins, C. J., Locksley, R. M. & Stockinger, B. TGF-beta in the context of an inflammatory cytokine milieu supports de novo differentiation of IL-17 producing cells. Immunity 24, 179–189 (2006).
Zhou, X. et al. Instability of the transcription factor Foxp3 leads to the generation of pathogenic memory T cells in vivo. Nat. Immunol. 10, 1000–1007 (2009).
Wan, Y. Y. & Flavell, R. A. Regulatory T-cell functions are subverted and converted owing to attenuated Foxp3 expression. Nature 445, 766–770 (2007).
Yang, X. O. et al. Molecular antagonism and plasticity of regulatory and inflammatory T cell programs. Immunity 29, 44–56 (2008).
Zhou, X. et al. Selective miRNA disruption in Treg cells leads to uncontrolled autoimmunity. J. Exp. Med. 205, 1983–1991 (2008).
Radhakrishnan, S. et al. Reprogrammed FoxP3+ T regulatory cells become IL-17+ antigen-specific autoimmune effectors in vitro and in vivo. J. Immunol. 181, 3137–3147 (2008).
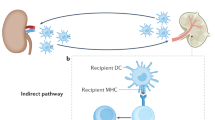
Li, X. C., Rothstein, D. M. & Sayegh, M. H. Costimulatory pathways in transplantation: challenges and new developments. Immunol. Rev. 229, 271–293 (2009).
Lu, L. F. et al. Foxp3-dependent microRNA155 confers competitive fitness to regulatory T cells by targeting SOCS1 protein. Immunity 30, 80–91 (2009).
Auchincloss, H. Jr & Sultan, H. Antigen processing and presentation in transplantation. Curr. Opin. Immunol. 8, 681–687 (1996).
Rulifson, I. C., Szot, G. L., Palmer, E. & Bluestone, J. A. Inability to induce tolerance through direct antigen presentation. Am. J. Transplant. 2, 510–519 (2002).
Hara, M. et al. IL-10 is required for regulatory T cells to mediate tolerance to alloantigens in vivo. J. Immunol. 166, 3789–3796 (2001).
Feuerer, M. et al. Lean, but not obese, fat is enriched for a unique population of regulatory T cells that affect metabolic parameters. Nat. Med. 15, 930–939 (2009).
Graca, L., Cobbold, S. P. & Waldmann, H. Identification of regulatory T cells in tolerated allografts. J. Exp. Med. 195, 1641–1646 (2002).
Zhang, N. et al. Regulatory T cells sequentially migrate from inflamed tissues to draining lymph nodes to suppress the alloimmune response. Immunity 30, 458–469 (2009).
Surh, C. D., Boyman, O., Purton, J. F. & Sprent, J. Homeostasis of memory T cells. Immunol. Rev. 211, 154–163 (2006).
Qin, S. et al. “Infectious” transplantation tolerance. Science 259, 974–977 (1993).
Honey, K., Cobbold, S. P. & Waldmann, H. CD40 ligand blockade induces CD4+ T cell tolerance and linked suppression. J. Immunol. 163, 4805–4810 (1999).
Yang, J. et al. Allograft rejection mediated by memory T cells is resistant to regulation. Proc. Natl Acad. Sci. USA 104, 19954–19959 (2007).
Korn, T. et al. Myelin-specific regulatory T cells accumulate in the CNS but fail to control autoimmune inflammation. Nat. Med. 13, 423–431 (2007).
Roy, S. et al. NK cells lyse T regulatory cells that expand in response to an intracellular pathogen. J. Immunol. 180, 1729–1736 (2008).
Allan, S. E. et al. Activation-induced FOXP3 in human T effector cells does not suppress proliferation or cytokine production. Int. Immunol. 19, 345–354 (2007).
Seddiki, N. et al. Expression of interleukin (IL)-2 and IL-7 receptors discriminates between human regulatory and activated T cells. J. Exp. Med. 203, 1693–1700 (2006).
Liu, W. et al. CD127 expression inversely correlates with FoxP3 and suppressive function of human CD4+ T reg cells. J. Exp. Med. 203, 1701–1711 (2006).
Miyara, M. et al. Functional delineation and differentiation dynamics of human CD4+ T cells expressing the FoxP3 transcription factor. Immunity 30, 899–911 (2009).
Allan, S. E. et al. The role of 2 FOXP3 isoforms in the generation of human CD4+ Tregs. J. Clin. Invest. 115, 3276–3284 (2005).
Bayer, A. L., Yu, A., Adeegbe, D. & Malek, T. R. Essential role for interleukin-2 for CD4+CD25+ T regulatory cell development during the neonatal period. J. Exp. Med. 201, 769–777 (2005).
Zheng, X. X. et al. Favorably tipping the balance between cytopathic and regulatory T cells to create transplantation tolerance. Immunity 19, 503–514 (2003).
Webster, K. E. et al. In vivo expansion of T reg cells with IL-2-mAb complexes: induction of resistance to EAE and long-term acceptance of islet allografts without immunosuppression. J. Exp. Med. 206, 751–760 (2009).
Acknowledgements
X. C. Li and L. A. Turka are supported by the NIH, USA.
Author information
Authors and Affiliations
Contributions
X. C. Li researched data for the article and contributed to the discussion of content, writing the article, and reviewing/editing the manuscript before submission. L. A. Turka contributed to the discussion of content, writing the article, and reviewing/editing the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
L. A. Turka declares that he has acted as a consultant for GlaxoSmithKline, Biogen IDEC and Bristol–Myers Squibb, and is a stockholder/director for Novartis. X. C. Li declares no competing interests.
Rights and permissions
About this article
Cite this article
Li, X., Turka, L. An update on regulatory T cells in transplant tolerance and rejection. Nat Rev Nephrol 6, 577–583 (2010). https://doi.org/10.1038/nrneph.2010.101
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneph.2010.101
This article is cited by
-
IL-6 Directed Therapy in Transplantation
Current Transplantation Reports (2021)
-
HLA in transplantation
Nature Reviews Nephrology (2018)
-
Donor antigen-primed regulatory T cells permit liver regeneration and phenotype correction in hemophilia A mouse by allogeneic bone marrow stem cells
Stem Cell Research & Therapy (2015)
-
The involvement of T regulatory lymphocytes in a cohort of lupus nephritis patients: a pilot study
Internal and Emergency Medicine (2015)
-
Lack of the protein tyrosine phosphatase PTPN22 strengthens transplant tolerance to pancreatic islets in mice
Diabetologia (2015)