Key Points
-
Studies in the past decade have provided data on the presence and distribution of voltage-gated ion channels in the dendrites of many types of neuron, but general rules that can explain their distribution are still missing. Uncovering these rules will be an important step towards a 'functional proteomics' of nerve cells, which will be essential in defining neuronal phenotypes.
-
The properties of the dendritic tree have important implications for neuronal function. But if dendrites were strictly passive, this would limit their ability to contribute to synaptic integration. So, the presence of active conductances and their precise distributions profoundly modify the functional properties of the dendritic tree.
-
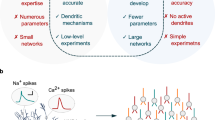
The localization of at least four classes of voltage-gated ion channels — Na+, Ih, KA and Ca2+ channels — has been studied in a variety of cells from several brain regions, using both direct approaches and theoretical modelling. Channel distribution is often heterogeneous within a single neuron and across different cell types. A key goal is to explain the functional properties of each neuronal type as a function of this heterogeneity.
-
So far, attempts to account for specific neuronal phenotypes on the basis of channel distributions are in their infancy, but basic rules are beginning to emerge. More importantly, this type of analysis makes it possible to formulate specific predictions about neuronal function that depend on the channels that are found in the dendrites of a given cell — predictions that can be tested experimentally. Progress in this field will depend on obtaining more detailed information about the distribution of more channel subtypes in more types of neuron.
Abstract
A key goal in neuroscience is to explain how the operations of a neuron emerge from sets of active channels with specific dendritic distributions. If general principles can be identified for these distributions, dendritic channels should reflect the computational role of a given cell type within its functional neural circuit. Here, we discuss insights from experimental and computational data on the distribution of voltage-gated channels in dendrites, and attempt to derive rules for how their interactions implement different dendritic functions. We propose that this type of analysis will be important for understanding behavioural processes in terms of single-neuron properties, and that it constitutes a step towards a 'functional proteomics' of nerve cells, which will be essential for defining neuronal phenotypes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Magee, J. C. in Dendrites (eds Stuart, G., Spruston, N. & Häusser, M.) 139–160 (Oxford Univ. Press, New York, 1999).
Häusser, M., Spruston, N. & Stuart, G. J. Diversity and dynamics of dendritic signaling. Science 290, 739–744 (2000).A comprehensive review of the experimental evidence for the ways in which signal processing can be carried out in dendrites.
Johnston, D. et al. Dendritic potassium channels in hippocampal pyramidal neurons. J. Physiol. (Lond.) 525, 75–81 (2000).
Reyes, A. Influence of dendritic conductances on the input–output properties of neurons. Annu. Rev. Neurosci. 24, 653–675 (2001).
Segev, I. & London, M. Untangling dendrites with quantitative models. Science 290, 744–750 (2000).A clear, state-of-the-art account, largely from a modelling point of view, of how dendrites are fundamental in neuronal information processing.
Rall, W. & Shepherd, G. M. Theoretical reconstruction of field potentials and dendrodendritic synaptic interactions in olfactory bulb. J. Neurophysiol. 31, 884–915 (1968).
Single, S. & Borst, A. Dendritic integration and its role in computing image velocity. Science 281, 1848–1850 (1998).
Livingstone, M. S. Mechanisms of direction selectivity in macaque V1. Neuron 20, 509–526 (1998).
Agmon-Snir, H., Carr, C. E. & Rinzel, J. The role of dendrites in auditory coincidence detection. Nature 393, 268–272 (1998).A pioneering study that provides direct experimental evidence of a neuronal function that cannot be carried out without dendrites.
Rall, W. in Neural Theory and Modeling (ed. Reiss, R.) 73–97 (Stanford Univ. Press, Stanford, 1964).
Agmon-Snir, H. & Segev, I. Signal delay and input synchronization in passive dendritic structures. J. Neurophysiol. 70, 2066–2085 (1993).
Stuart, G. J. & Sakmann, B. Active propagation of somatic action potentials into neocortical pyramidal cell dendrites. Nature 367, 69–72 (1994).
Magee, J. C. & Johnston, D. Characterization of single voltage-gated Na+ and Ca2+ channels in apical dendrites of rat CA1 pyramidal neurons. J. Physiol. (Lond.) 487, 67–90 (1995).A pioneering, accurate characterization of properties and distributions of dendritic Na+ and Ca2+ channels in hippocampal pyramidal neurons.
Bischofberger, J. & Jonas, P. Action potential propagation into the presynaptic dendrites of rat mitral cells. J. Physiol. (Lond.) 504, 359–365 (1997).
Xiong, W. & Chen, W. R. Dynamic gating of spike propagation in the mitral cell lateral dendrites. Neuron 34, 115–126 (2002).This paper and reference 65 report action potential backpropagation and its modulation by synaptic activity for both apical and lateral dendrites in mitral cells.
Margrie, T. W., Sakmann, B. & Urban, N. N. Action potential propagation in mitral cell lateral dendrites is decremental and controls recurrent and lateral inhibition in the mammalian olfactory bulb. Proc. Natl Acad. Sci. USA 98, 319–324 (2001).
Martina, M., Vida, I. & Jonas, P. Distal initiation and active propagation of action potentials in interneuron dendrites. Science 287, 295–300 (2000).
Steinhauser, C., Tennigkeit, M., Matthies, H. & Gundel, J. Properties of the fast sodium channels in pyramidal neurones isolated from the CA1 and CA3 areas of the hippocampus of postnatal rats. Pflugers Arch. 415, 756–761 (1990).
Buzsaki, G., Penttonen, M., Nadasdy, Z. & Bragin, A. Pattern and inhibition-dependent invasion of pyramidal cell dendrites by fast spikes in the hippocampus in vivo. Proc. Natl Acad. Sci. USA 93, 9921–9925 (1996).
Williams, S. R. & Stuart, G. J. Action potential backpropagation and somato-dendritic distribution of ion channels in thalamocortical neurons. J. Neurosci. 20, 1307–1317 (2000).
Stuart, G. & Häusser, M. Initiation and spread of sodium action potentials in cerebellar Purkinje cells. Neuron 13, 703–712 (1994).
Callaway, J. C. & Ross, W. N. Spatial distribution of synaptically activated sodium concentration changes in cerebellar Purkinje neurons. J. Neurophysiol. 77, 145–152 (1997).
Safronov, B. V. Spatial distribution of Na+ and K+ channels in spinal dorsal horn neurones: role of the soma, axon and dendrites in spike generation. Prog. Neurobiol. 59, 217–241 (1999).
Velte, T. J. & Masland, R. H. Action potentials in the dendrites of retinal ganglion cells. J. Neurophysiol. 81, 1412–1417 (1999).
Häusser, M., Stuart, G., Racca, C. & Sakmann, B. Axonal initiation and active dendritic propagation of action potentials in substantia nigra neurons. Neuron 15, 637–647 (1995).
Ketelaars, S. O., Gorter, J. A., Van Vliet, E. A., Lopes da Silva, F. H. & Wadman, W. J. Sodium currents in isolated rat CA1 pyramidal and dentate granule neurones in the post-status epilepticus model of epilepsy. Neuroscience 105, 109–120 (2001).
Jefferys, J. G. Initiation and spread of action potentials in granule cells maintained in vitro in slices of guinea-pig hippocampus. J. Physiol. (Lond.) 289, 375–388 (1979).
Riazanski, V. et al. Functional and molecular analysis of transient voltage-dependent K+ currents in rat hippocampal granule cells. J. Physiol. (Lond.) 537, 391–406 (2001).
Hoffman, D. A., Magee, J. C., Colbert, C. M. & Johnston, D. K+ channel regulation of signal propagation in dendrites of hippocampal pyramidal neurons. Nature 387, 869–875 (1997).
Bekkers, J. M. Distribution and activation of voltage-gated potassium channels in cell-attached and outside-out patches from large layer 5 cortical pyramidal neurons of the rat. J. Physiol. (Lond.) 525, 611–620 (2000).This paper and reference 31 characterize the properties and distribution of K A and K DR K+ currents. They represent new reference points for models of deep neocortical neurons.
Korngreen, A. & Sakmann, B. Voltage-gated K+ channels in layer 5 neocortical pyramidal neurones from young rats: subtypes and gradients. J. Physiol. (Lond.) 525, 621–639 (2000).
Strecker, T. & Heinemann, U. Redistribution of K+ channels into dendrites is unlikely to account for developmental down regulation of A-currents in rat dentate gyrus granule cells. Neurosci. Lett. 164, 209–212 (1993).
Etzion, Y. & Grossman, Y. Potassium currents modulation of calcium spike firing in dendrites of cerebellar Purkinje cells. Exp. Brain Res. 122, 283–294 (1998).
Miyasho, T. et al. Low-threshold potassium channels and a low-threshold calcium channel regulate Ca2+ spike firing in the dendrites of cerebellar Purkinje neurons: a modeling study. Brain Res. 891, 106–115 (2001).
Wolff, M., Vogel, W. & Safronov, B. V. Uneven distribution of K+ channels in soma, axon and dendrites of rat spinal neurones: functional role of the soma in generation of action potentials. J. Physiol. (Lond.) 509, 767–776 (1998).
Magee, J. C. Dendritic hyperpolarization-activated currents modify the integrative properties of hippocampal CA1 pyramidal neurons. J. Neurosci. 18, 7613–7624 (1998).
Williams, S. R. & Stuart, G. J. Site independence of EPSP time course is mediated by dendritic Ih in neocortical pyramidal neurons. J. Neurophysiol. 83, 3177–3182 (2000).
Berger, T., Larkum, M. E. & Luscher, H. R. High Ih channel density in the distal apical dendrite of layer V pyramidal cells increases bidirectional attenuation of EPSPs. J. Neurophysiol. 85, 855–868 (2001).One of few studies on the properties and distribution of a dendritic channel up to 800 μm from the soma in a neocortical neuron.
Tseng, G. F. & Haberly, L. B. Deep neurons in piriform cortex. II. Membrane properties that underlie unusual synaptic responses. J. Neurophysiol. 62, 386–400 (1989).
McCormick, D. A. & Pape, H. C. Properties of a hyperpolarization-activated cation current and its role in rhythmic oscillation in thalamic relay neurones. J. Physiol. (Lond.) 431, 291–318 (1990).
Cadetti, L. & Belluzzi, O. Hyperpolarisation-activated current in glomerular cells of the rat olfactory bulb. Neuroreport 12, 3117–3120 (2001).
Williams, S. R., Christensen, S. R., Stuart, G. J. & Häusser M. Membrane potential bistability is controlled by the hyperpolarization-activated current IH in rat cerebellar Purkinje neurons in vitro. J. Physiol. (Lond.) 539, 469–483 (2002).
Spruston, N. & Johnston, D. Perforated patch-clamp analysis of the passive membrane properties of three classes of hippocampal neurons. J. Neurophysiol. 67, 508–529 (1992).
Lubke, J., Frotscher, M. & Spruston, N. Specialized electrophysiological properties of anatomically identified neurons in the hilar region of the rat fascia dentata. J. Neurophysiol. 79, 1518–1534 (1998).
Luthi, A. & McCormick, D. A. H-current: properties of a neuronal and network pacemaker. Neuron 21, 9–12 (1998).
Magee, J. C. Dendritic Ih normalizes temporal summation in hippocampal CA1 neurons. Nature Neurosci. 2, 508–514 (1999).
Williams, S. R. & Stuart, G. J. Dependence of EPSP efficacy on synapse location in neocortical pyramidal neurons. Science 295, 1907–1910 (2002).
Mao, B. Q., Hamzei-Sichani, F., Aronov, D., Froemke, R. C. & Yuste, R. Dynamics of spontaneous activity in neocortical slices. Neuron 32, 883–898 (2001).
Johnston, D., Magee, J. C., Colbert, C. M. & Cristie, B. R. Active properties of neuronal dendrites. Annu. Rev. Neurosci. 19, 165–186 (1996).
Westenbroek, R. E., Ahlijanian, M. K. & Catterall, W. A. Clustering of L-type Ca2+ channels at the base of major dendrites in hippocampal pyramidal neurons. Nature 347, 281–284 (1990).
Mills, L. R. et al. N-type Ca2+ channels are located on somata, dendrites, and a subpopulation of dendritic spines on live hippocampal pyramidal neurons. J. Neurosci. 14, 6815–6824 (1994).
Markram, H., Helm, P. J. & Sakmann, B. Dendritic calcium transients evoked by single back-propagating action potentials in rat neocortical pyramidal neurons. J. Physiol. (Lond.) 485, 1–20 (1995).
Markram, H. & Sakmann, B. Calcium transients in dendrites of neocortical neurons evoked by single subthreshold excitatory postsynaptic potentials via low-voltage-activated calcium channels. Proc. Natl Acad. Sci. USA 91, 5207–5211 (1994).
Oakley, J. C., Schwindt, P. C. & Crill, W. E. Initiation and propagation of regenerative Ca2+-dependent potentials in dendrites of layer 5 pyramidal neurons. J. Neurophysiol. 86, 503–513 (2001).
Mori, K., Nowycky, M. C. & Shepherd, G. M. Electrophysiological analysis of mitral cells in the isolated turtle olfactory bulb. J. Physiol. (Lond.) 314, 281–294 (1981).
Wang, X., McKenzie, J. S. & Kemm, R. E. Whole-cell calcium currents in acutely isolated olfactory bulb output neurons of the rat. J. Neurophysiol. 75, 1138–1151 (1996).
Isaacson, J. S. & Strowbridge, B. W. Olfactory reciprocal synapses: dendritic signaling in the CNS. Neuron 20, 749–761 (1998).
Llinas, R. & Sugimori, M. Electrophysiological properties of in vitro Purkinje cell dendrites in mammalian cerebellar slices. J. Physiol. (Lond.) 305, 197–213 (1980).
Lev-Ram, V., Miyakawa, H., Lasser-Ross, N. & Ross, W. N. Calcium transients in cerebellar Purkinje neurons evoked by intracellular stimulation. J. Neurophysiol. 68, 1167–1177 (1992).
Usowicz, M. M., Sugimori, M., Cherksey, B. & Llinas, R. P-type calcium channels in the somata and dendrites of adult cerebellar Purkinje cells. Neuron 9, 1185–1199 (1992).
Budde, T., Munsch, T. & Pape, H. C. Distribution of L-type calcium channels in rat thalamic neurones. Eur. J. Neurosci. 10, 586–597 (1998).
Mouginot, D., Bossu, J. L. & Gahwiler, B. H. Low-threshold Ca2+ currents in dendritic recordings from Purkinje cells in rat cerebellar slice cultures. J. Neurosci. 17, 160–170 (1997).
Fisher, R. E., Gray, R. & Johnston, D. Properties and distribution of single voltage-gated calcium channels in adult hippocampal neurons. J. Neurophysiol. 64, 91–104 (1990).
Attwell, D. & Laughlin, S. B. An energy budget for signaling in the grey matter of the brain. J. Cereb. Blood Flow Metab. 21, 1133–1145 (2001).A lucid account of how neuronal encoding might be constrained by energetic requirements, with particular reference to synaptic activity in cortical dendrites.
Chen, W. R., Midtgaard, J. & Shepherd, G. M. Forward and backward propagation of dendritic impulses and their synaptic control in mitral cells. Science 278, 463–467 (1997).
Nowycky, M. C., Mori, K. & Shepherd, G. M. Blockade of synaptic inhibition reveals long-lasting synaptic excitation in isolated turtle olfactory bulb. J. Neurophysiol. 46, 649–658 (1981).
Larkum, M. E., Zhu, J. J. & Sakmann, B. Dendritic mechanisms underlying the coupling of the dendritic with the axonal action potential initiation zone of adult rat layer 5 pyramidal neurons. J. Physiol. (Lond.) 533, 447–466 (2001).
Migliore, M., Cook, E. P., Jaffe, D. B., Turner, D. A. & Johnston, D. Computer simulations of morphologically reconstructed CA3 hippocampal neurons. J. Neurophysiol. 73, 1157–1168 (1995).
Llinas, R. R. The intrinsic electrophysiological properties of mammalian neurons: insights into central nervous system function. Science 242, 1654–1664 (1988).
Hines, M. L. & Carnevale, N. T. The NEURON simulation environment. Neural Comput. 9, 1179–1209 (1997).
Migliore, M., Hoffman, D., Magee, J. C. & Johnston, D. Role of an A-type K+ conductance in the back-propagation of action potentials in the dendrites of hippocampal neurons. J. Comput. Neurosci. 7, 5–15 (1999).A realistic model of a CA1 neuron showing how the experimentally observed dendritic distribution of K A might modulate the backpropagation of action potentials and its association with a synaptic input.
Acknowledgements
Our work has been supported by the National Institute on Deafness and Other Communication Disorders, the National Institute of Mental Health, the National Institute of Neurological Disorders and Stroke, the National Institute on Aging and the National Science Foundation (Human Brain Project), and by the University Research Initiative (Department of Defense). We thank M. Hines, W. Chen, A. Davison and T. Morse for valuable discussions.
Author information
Authors and Affiliations
Corresponding author
Related links
Related links
FURTHER INFORMATION
Encyclopedia of Life Sciences
Glossary
- COINCIDENCE DETECTION
-
The ability to sense the simultaneous occurrence of synaptic inputs at different points on the same cell.
- INPUT RESISTANCE
-
The voltage change elicited by the injection of current into a cell, divided by the amount of current injected.
- α-FUNCTION
-
The functional form G(t) = (t/α)e−αt is known as the α-function, and is widely used to represent the time course of a synaptic conductance. The value of α determines the rise and decay times.
- APICAL AND BASAL DENDRITES
-
Cell types such as mitral cells and cortical pyramidal neurons have dendritic trees that are divided into two parts — an apical tree that ascends across layers and a basal tree that extends laterally.
- MEMBRANE TIME CONSTANT
-
A quantity that depends on the capacitance and resistance of the cell membrane, and which sets a timescale for changes in voltage. A small time constant means that the membrane potential can change rapidly.
- CURRENT–VOLTAGE RELATIONSHIP
-
A plot of the changes in ionic current as a function of membrane voltage.
- NUCLEATED PATCHES
-
A special configuration of patch-clamp recording in which a membrane patch is pulled out of the cell together with the nucleus. The external face of the membrane still faces the extracellular medium, and the nucleus lies enclosed by this balloon-like patch.
- TEMPORAL SUMMATION
-
The way in which synaptic events add in time. One of the basic elements of synaptic integration.
- BACKPROPAGATING ACTION POTENTIALS
-
Although action potentials typically travel down the axon towards the presynaptic terminal, they can also be initiated at the axon and propagate back into the dendrites, shaping the integration of synaptic activity and influencing the induction of synaptic plasticity.
- PERFORANT PATH
-
Axons of entorhinal cortex neurons that terminate largely in the hippocampal dentate gyrus. Some fibres from the entorhinal path reach the distal end of apical dendrites of CA1 neurons.
- SCHAFFER COLLATERALS
-
Axons of the CA3 pyramidal cells of the hippocampus that form synapses with the apical dendrites of CA1 neurons.
- PARALLEL FIBRES
-
The axons of cerebellar granule cells. Parallel fibres emerge from the molecular layer of the cerebellar cortex towards the periphery, where they extend branches perpendicular to the main axis of the Purkinje neurons and form the so-called en passant synapses with this cell type.
- CLIMBING FIBRES
-
Cerebellar afferents that arise from the inferior olivary nucleus, each of which forms multiple synapses with a single Purkinje cell.
Rights and permissions
About this article
Cite this article
Migliore, M., Shepherd, G. Emerging rules for the distributions of active dendritic conductances. Nat Rev Neurosci 3, 362–370 (2002). https://doi.org/10.1038/nrn810
Issue Date:
DOI: https://doi.org/10.1038/nrn810
This article is cited by
-
A patient-derived mutation of epilepsy-linked LGI1 increases seizure susceptibility through regulating Kv1.1
Cell & Bioscience (2023)
-
Positive and biphasic extracellular waveforms correspond to return currents and axonal spikes
Communications Biology (2023)
-
Degeneracy in the emergence of spike-triggered average of hippocampal pyramidal neurons
Scientific Reports (2020)
-
Effects of the Involvement of Calcium Channels on Neuronal Hyperexcitability Related to Alzheimer’s Disease: A Computational Model
Neurophysiology (2020)
-
NeuroPath2Path: Classification and elastic morphing between neuronal arbors using path-wise similarity
Neuroinformatics (2020)