Key Points
-
The dentate gyrus in the hippocampus in one of the discrete regions in which new neurons are continuously generated and integrated into local networks in the brains of adult mammals, including humans. Studies in the past decade have led to an understanding of the process of neurogenesis in the dentate gyrus; however, the functions of adult-born dentate granule cells (DGCs) remain unclear.
-
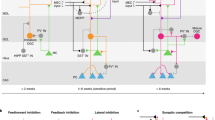
After birth, adult-born DGCs undergo a lengthy process of morphogenesis including the de novo growth of axons and dendrites and the formation of both efferent and afferent synapses. The adult-born DGCs show enhanced excitability and plasticity before they are fully integrated into the brain circuitry.
-
The addition of adult-born DGCs into the local network is extensively regulated by the experiences of the animal, which evoke hippocampal activity. Physiological and pathological conditions that alter hippocampal activity also affect adult neurogenesis in the hippocampus. The experiences of animals, especially those occurring during the maturation of DGCs, can influence the responsiveness of these cells when they become fully mature.
-
A common hypothesis emerging from several computational studies is that adult neurogenesis allows plasticity to be mostly localized to newborn immature DGCs, preserving the information that is represented by mature DGCs. A recent model further proposes that immature DGCs can serve as a pattern integrator by linking events that occur closely in time.
-
Studies combining neurogenesis ablation models with behavioural analyses have yielded inconsistent data concerning the functions of adult hippocampal neurogenesis. Besides the differences in specific experimental conditions, the varied experimental timelines and the types of function assessed are two major reasons for these discrepancies.
-
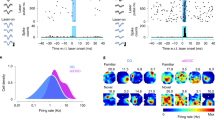
The dentate gyrus plays a crucial role in pattern separation. A role for adult neurogenesis in pattern separation as a function of pattern similarity has recently been revealed.
-
In the future, novel methods that have the cellular and temporal precision to manipulate neurogenesis and behavioural tests that directly assess dentate gyrus-related functions will be required to understand the functional mechanisms of adult neurogenesis.
Abstract
The integration of adult-born neurons into the circuitry of the adult hippocampus suggests an important role for adult hippocampal neurogenesis in learning and memory, but its specific function in these processes has remained elusive. In this article, we summarize recent progress in this area, including advances based on behavioural studies and insights provided by computational modelling. Increasingly, evidence suggests that newborn neurons might be involved in hippocampal functions that are particularly dependent on the dentate gyrus, such as pattern separation. Furthermore, newborn neurons at different maturation stages may make distinct contributions to learning and memory. In particular, computational studies suggest that, before newborn neurons are fully mature, they might function as a pattern integrator by introducing a degree of similarity to the encoding of events that occur closely in time.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Altman, J. & Das, G. D. Post-natal origin of microneurones in the rat brain. Nature 207, 953–956 (1965).
Gage, F. H. Mammalian neural stem cells. Science 287, 1433–1438 (2000).
Enikolopov, G. & Overstreet Wadiche, L. in Adult Neurogenesis (eds. Gage, F. H., Kempermann, G. & Song, H.) 81–100 (Cold Spring Harbor Laboratory Press, New York, 2008).
Kuhn, H. G. & Peterson, D. A. in Adult Neurogenesis (eds. Gage, F. H., Kempermann, G. & Song, H.) 25–47 (Cold Spring Harbor Laboratory Press, New York, 2008).
Zhao, C. in Adult Neurogenesis (eds. Gage, F. H., Kempermann, G. & Song, H.) 101–117 (Cold Spring Harbor Laboratory Press, New York, 2008).
Zhao, C., Deng, W. & Gage, F. H. Mechanisms and functional implications of adult neurogenesis. Cell 132, 645–660 (2008).
Suh, H., Deng, W. & Gage, F. H. Signaling in adult neurogenesis. Annu. Rev. Cell Dev. Biol. 25, 253–275 (2009).
Grubb, M. S., Nissant, A., Murray, K. & Lledo, P. M. Functional maturation of the first synapse in olfaction: development and adult neurogenesis. J. Neurosci. 28, 2919–2932 (2008).
Nissant, A., Bardy, C., Katagiri, H., Murray, K. & Lledo, P. M. Adult neurogenesis promotes synaptic plasticity in the olfactory bulb. Nature Neurosci. 12, 728–730 (2009).
Breton-Provencher, V., Lemasson, M., Peralta, M. R. III & Saghatelyan, A. Interneurons produced in adulthood are required for the normal functioning of the olfactory bulb network and for the execution of selected olfactory behaviors. J. Neurosci. 29, 15245–15257 (2009).
Whitman, M. C. & Greer, C. A. Adult neurogenesis and the olfactory system. Prog. Neurobiol. 89, 162–175 (2009).
Squire, L. R. Memory and the hippocampus: a synthesis from findings with rats, monkeys, and humans. Psychol. Rev. 99, 195–231 (1992).
Sahay, A. & Hen, R. Hippocampal neurogenesis and depression. Novartis Found. Symp. 289, 152–160; discussion 160–164, 193–195 (2008).
David, D. J. et al. Neurogenesis-dependent and -independent effects of fluoxetine in an animal model of anxiety/depression. Neuron 62, 479–493 (2009).
Cameron, H. A., Woolley, C. S., McEwen, B. S. & Gould, E. Differentiation of newly born neurons and glia in the dentate gyrus of the adult rat. Neuroscience 56, 337–344 (1993).
Zhao, C., Teng, E. M., Summers, R. G. Jr, Ming, G. L. & Gage, F. H. Distinct morphological stages of dentate granule neuron maturation in the adult mouse hippocampus. J. Neurosci. 26, 3–11 (2006).
Snyder, J. S. et al. Adult-born hippocampal neurons are more numerous, faster maturing, and more involved in behavior in rats than in mice. J. Neurosci. 29, 14484–14495 (2009).
Esposito, M. S. et al. Neuronal differentiation in the adult hippocampus recapitulates embryonic development. J. Neurosci. 25, 10074–10086 (2005). The first systematic characterization of the process of adult hippocampal neurogenesis regarding the morphological and physiological maturation of adult-born DGCs.
Ge, S. et al. GABA regulates synaptic integration of newly generated neurons in the adult brain. Nature 439, 589–593 (2006).
Hastings, N. B., Seth, M. I., Tanapat, P., Rydel, T. A. & Gould, E. Granule neurons generated during development extend divergent axon collaterals to hippocampal area CA3. J. Comp. Neurol. 452, 324–333 (2002).
Overstreet Wadiche, L., Bromberg, D. A., Bensen, A. L. & Westbrook, G. L. GABAergic signaling to newborn neurons in dentate gyrus. J. Neurophysiol. 94, 4528–4532 (2005).
Markwardt, S. J., Wadiche, J. I. & Overstreet-Wadiche, L. S. Input-specific GABAergic signaling to newborn neurons in adult dentate gyrus. J. Neurosci. 29, 15063–15072 (2009).
Jagasia, R. et al. GABA-cAMP response element-binding protein signaling regulates maturation and survival of newly generated neurons in the adult hippocampus. J. Neurosci. 29, 7966–7977 (2009).
Toni, N. et al. Synapse formation on neurons born in the adult hippocampus. Nature Neurosci. 10, 727–734 (2007).
Toni, N. et al. Neurons born in the adult dentate gyrus form functional synapses with target cells. Nature Neurosci. 11, 901–907 (2008).
Faulkner, R. L. et al. Development of hippocampal mossy fiber synaptic outputs by new neurons in the adult brain. Proc. Natl Acad. Sci. USA 105, 14157–14162 (2008).
Bliss, T. V. & Collingridge, G. L. A synaptic model of memory: long-term potentiation in the hippocampus. Nature 361, 31–39 (1993).
Tashiro, A., Sandler, V. M., Toni, N., Zhao, C. & Gage, F. H. NMDA-receptor-mediated, cell-specific integration of new neurons in adult dentate gyrus. Nature 442, 929–933 (2006).
Ge, S., Yang, C. H., Hsu, K. S., Ming, G. L. & Song, H. A critical period for enhanced synaptic plasticity in newly generated neurons of the adult brain. Neuron 54, 559–566 (2007). This study demonstrated the enhanced plasticity of developing adult-born DGCs by a systematic characterization of retrovirus-labelled DGCs at different time points.
Schmidt-Hieber, C., Jonas, P. & Bischofberger, J. Enhanced synaptic plasticity in newly generated granule cells of the adult hippocampus. Nature 429, 184–187 (2004).
Ambrogini, P. et al. Morpho-functional characterization of neuronal cells at different stages of maturation in granule cell layer of adult rat dentate gyrus. Brain Res. 1017, 21–31 (2004).
Scobie, K. N. et al. Kruppel-like factor 9 is necessary for late-phase neuronal maturation in the developing dentate gyrus and during adult hippocampal neurogenesis. J. Neurosci. 29, 9875–9887 (2009).
Gould, E., Beylin, A., Tanapat, P., Reeves, A. & Shors, T. J. Learning enhances adult neurogenesis in the hippocampal formation. Nature Neurosci. 2, 260–265 (1999).
Epp, J. R., Spritzer, M. D. & Galea, L. A. Hippocampus-dependent learning promotes survival of new neurons in the dentate gyrus at a specific time during cell maturation. Neuroscience 149, 273–285 (2007).
Leuner, B. et al. Learning enhances the survival of new neurons beyond the time when the hippocampus is required for memory. J. Neurosci. 24, 7477–7481 (2004).
Leuner, B., Waddell, J., Gould, E. & Shors, T. J. Temporal discontiguity is neither necessary nor sufficient for learning-induced effects on adult neurogenesis. J. Neurosci. 26, 13437–13442 (2006).
Dupret, D. et al. Spatial learning depends on both the addition and removal of new hippocampal neurons. PLoS Biol. 5, e214 (2007).
Dobrossy, M. D. et al. Differential effects of learning on neurogenesis: learning increases or decreases the number of newly born cells depending on their birth date. Mol. Psychiatry 8, 974–982 (2003).
van Praag, H., Kempermann, G. & Gage, F. H. Neural consequences of environmental enrichment. Nature Rev. Neurosci. 1, 191–198 (2000).
Kempermann, G., Kuhn, H. G. & Gage, F. H. More hippocampal neurons in adult mice living in an enriched environment. Nature 386, 493–495 (1997). The first study to show the regulation of the survival of adult-born DGCs by experience in mice.
Tashiro, A., Makino, H. & Gage, F. H. Experience-specific functional modification of the dentate gyrus through adult neurogenesis: a critical period during an immature stage. J. Neurosci. 27, 3252–3259 (2007). The authors showed that the experiences of mice when the adult-born DGCs are in a hyper-excitable stage affect the subsequent responsiveness of these DGCs to various inputs.
Bruel-Jungerman, E., Laroche, S. & Rampon, C. New neurons in the dentate gyrus are involved in the expression of enhanced long-term memory following environmental enrichment. Eur. J. Neurosci. 21, 513–521 (2005).
Meshi, D. et al. Hippocampal neurogenesis is not required for behavioral effects of environmental enrichment. Nature Neurosci. 9, 729–731 (2006).
Hillman, C. H., Erickson, K. I. & Kramer, A. F. Be smart, exercise your heart: exercise effects on brain and cognition. Nature Rev. Neurosci. 9, 58–65 (2008).
van Praag, H. Exercise and the brain: something to chew on. Trends Neurosci. 32, 283–290 (2009).
van Praag, H., Kempermann, G. & Gage, F. H. Running increases cell proliferation and neurogenesis in the adult mouse dentate gyrus. Nature Neurosci. 2, 266–270 (1999).
van Praag, H., Shubert, T., Zhao, C. & Gage, F. H. Exercise enhances learning and hippocampal neurogenesis in aged mice. J. Neurosci. 25, 8680–8685 (2005).
Muotri, A. R., Zhao, C., Marchetto, M. C. & Gage, F. H. Environmental influence on L1 retrotransposons in the adult hippocampus. Hippocampus 19, 1002–1007 (2009).
Snyder, J. S., Glover, L. R., Sanzone, K. M., Kamhi, J. F. & Cameron, H. A. The effects of exercise and stress on the survival and maturation of adult-generated granule cells. Hippocampus 19, 898–906 (2009).
van Praag, H., Christie, B. R., Sejnowski, T. J. & Gage, F. H. Running enhances neurogenesis, learning, and long-term potentiation in mice. Proc. Natl Acad. Sci. USA 96, 13427–13431 (1999).
Leasure, J. L. & Decker, L. Social isolation prevents exercise-induced proliferation of hippocampal progenitor cells in female rats. Hippocampus 19, 907–912 (2009).
Bruel-Jungerman, E., Davis, S., Rampon, C. & Laroche, S. Long-term potentiation enhances neurogenesis in the adult dentate gyrus. J. Neurosci. 26, 5888–5893 (2006).
Chun, S. K., Sun, W., Park, J. J. & Jung, M. W. Enhanced proliferation of progenitor cells following long-term potentiation induction in the rat dentate gyrus. Neurobiol. Learn. Mem. 86, 322–329 (2006).
Madsen, T. M., Greisen, M. H., Nielsen, S. M., Bolwig, T. G. & Mikkelsen, J. D. Electroconvulsive stimuli enhance both neuropeptide Y receptor Y1 and Y2 messenger RNA expression and levels of binding in the rat hippocampus. Neuroscience 98, 33–39 (2000).
Malberg, J. E., Eisch, A. J., Nestler, E. J. & Duman, R. S. Chronic antidepressant treatment increases neurogenesis in adult rat hippocampus. J. Neurosci. 20, 9104–9110 (2000).
Parent, J. M. et al. Dentate granule cell neurogenesis is increased by seizures and contributes to aberrant network reorganization in the adult rat hippocampus. J. Neurosci. 17, 3727–3738 (1997).
Jessberger, S. et al. Seizure-associated, aberrant neurogenesis in adult rats characterized with retrovirus-mediated cell labeling. J. Neurosci. 27, 9400–9407 (2007).
Parent, J. M., Elliott, R. C., Pleasure, S. J., Barbaro, N. M. & Lowenstein, D. H. Aberrant seizure-induced neurogenesis in experimental temporal lobe epilepsy. Ann. Neurol. 59, 81–91 (2006).
Overstreet-Wadiche, L. S., Bromberg, D. A., Bensen, A. L. & Westbrook, G. L. Seizures accelerate functional integration of adult-generated granule cells. J. Neurosci. 26, 4095–4103 (2006).
Guzowski, J. F. et al. Mapping behaviorally relevant neural circuits with immediate-early gene expression. Curr. Opin. Neurobiol. 15, 599–606 (2005).
Jessberger, S. & Kempermann, G. Adult-born hippocampal neurons mature into activity-dependent responsiveness. Eur. J. Neurosci. 18, 2707–2712 (2003).
Ramirez-Amaya, V., Marrone, D. F., Gage, F. H., Worley, P. F. & Barnes, C. A. Integration of new neurons into functional neural networks. J. Neurosci. 26, 12237–12241 (2006).
Kee, N., Teixeira, C. M., Wang, A. H. & Frankland, P. W. Preferential incorporation of adult-generated granule cells into spatial memory networks in the dentate gyrus. Nature Neurosci. 10, 355–362 (2007).
Trouche, S., Bontempi, B., Roullet, P. & Rampon, C. Recruitment of adult-generated neurons into functional hippocampal networks contributes to updating and strengthening of spatial memory. Proc. Natl Acad. Sci. USA 106, 5919–5924 (2009).
Gould, E., Tanapat, P., Hastings, N. B. & Shors, T. J. Neurogenesis in adulthood: a possible role in learning. Trends Cogn. Sci. 3, 186–192 (1999).
Kempermann, G., Wiskott, L. & Gage, F. H. Functional significance of adult neurogenesis. Curr. Opin. Neurobiol. 14, 186–191 (2004).
Schinder, A. F. & Gage, F. H. A hypothesis about the role of adult neurogenesis in hippocampal function. Physiology (Bethesda) 19, 253–261 (2004).
Marr, D. Simple memory: a theory for archicortex. Philos. Trans. R. Soc. Lond. B Biol. Sci. 262, 23–81 (1971).
McNaughton, B. L. & Morris, R. G. M. Hippocampal synaptic enhancement and information storage within a distributed memory system. Trends Neurosci. 10, 408–415 (1987).
O'Reilly, R. C. & McClelland, J. L. Hippocampal conjunctive encoding, storage, and recall: avoiding a trade-off. Hippocampus 4, 661–682 (1994).
Rolls, E. T. A theory of hippocampal function in memory. Hippocampus 6, 601–620 (1996).
Treves, A. & Rolls, E. T. Computational constraints suggest the need for two distinct input systems to the hippocampal CA3 network. Hippocampus 2, 189–199 (1992).
Bakker, A., Kirwan, C. B., Miller, M. & Stark, C. E. Pattern separation in the human hippocampal CA3 and dentate gyrus. Science 319, 1640–1642 (2008).
Leutgeb, J. K., Leutgeb, S., Moser, M. B. & Moser, E. I. Pattern separation in the dentate gyrus and CA3 of the hippocampus. Science 315, 961–966 (2007).
McHugh, T. J. et al. Dentate gyrus NMDA receptors mediate rapid pattern separation in the hippocampal network. Science 317, 94–99 (2007).
Aimone, J. B. & Wiskott, L. in Adult Neurogenesis (eds. Gage, F. H., Kempermann, G. & Song, H.) 101–117 (Cold Spring Harbor Laboratory Press, New York, 2008).
Chambers, R. A., Potenza, M. N., Hoffman, R. E. & Miranker, W. Simulated apoptosis/neurogenesis regulates learning and memory capabilities of adaptive neural networks. Neuropsychopharmacology 29, 747–758 (2004).
Deisseroth, K. et al. Excitation-neurogenesis coupling in adult neural stem/progenitor cells. Neuron 42, 535–552 (2004).
Crick, C. & Miranker, W. Apoptosis, neurogenesis, and information content in Hebbian networks. Biol. Cybern. 94, 9–19 (2006).
Becker, S. A computational principle for hippocampal learning and neurogenesis. Hippocampus 15, 722–738 (2005).
Wiskott, L., Rasch, M. J. & Kempermann, G. A functional hypothesis for adult hippocampal neurogenesis: avoidance of catastrophic interference in the dentate gyrus. Hippocampus 16, 329–343 (2006).
Weisz, V. I. & Argibay, P. F. A putative role for neurogenesis in neuro-computational terms: inferences from a hippocampal model. Cognition 112, 229–240 (2009).
Aimone, J. B., Wiles, J. & Gage, F. H. Computational influence of adult neurogenesis on memory encoding. Neuron 61, 187–202 (2009). A bottom-up computational model of adult hippocampal neurogenesis. The authors proposed a role for adult-born DGCs with enhanced excitability in pattern integration through their broad tuning properties.
Aimone, J. B., Wiles, J. & Gage, F. H. Potential role for adult neurogenesis in the encoding of time in new memories. Nature Neurosci. 9, 723–727 (2006).
Friedman, W. J. Comment on “Potential role for adult neurogenesis in the encoding of time in new memories”. Hippocampus 17, 503–504 (2007).
Becker, S. & Wojtowicz, J. M. A model of hippocampal neurogenesis in memory and mood disorders. Trends Cogn. Sci. 11, 70–76 (2007).
Dupret, D. et al. Spatial relational memory requires hippocampal adult neurogenesis. PLoS One 3, e1959 (2008).
Saxe, M. D. et al. Ablation of hippocampal neurogenesis impairs contextual fear conditioning and synaptic plasticity in the dentate gyrus. Proc. Natl Acad. Sci. USA 103, 17501–17506 (2006).
Shors, T. J. et al. Neurogenesis in the adult is involved in the formation of trace memories. Nature 410, 372–376 (2001). The first study to show the functional importance of adult neurogenesis. The authors discovered that rats with reduced adult neurogenesis were impaired in learning conditioned response in an eye blink trace conditioning paradigm.
Shors, T. J., Townsend, D. A., Zhao, M., Kozorovitskiy, Y. & Gould, E. Neurogenesis may relate to some but not all types of hippocampal-dependent learning. Hippocampus 12, 578–584 (2002).
Clelland, C. D. et al. A functional role for adult hippocampal neurogenesis in spatial pattern separation. Science 325, 210–213 (2009). The first study to suggest an involvement of adult hippocampal neurogenesis in pattern separation, a proposed function for the dentate gyrus.
Deng, W., Saxe, M. D., Gallina, I. S. & Gage, F. H. Adult-born hippocampal dentate granule cells undergoing maturation modulate learning and memory in the brain. J. Neurosci. 29, 13532–13542 (2009). This study showed that adult-born DGCs contribute to learning and memory before their full maturation, at a stage when they have enhanced excitability.
Zhang, C. L., Zou, Y., He, W., Gage, F. H. & Evans, R. M. A role for adult TLX-positive neural stem cells in learning and behaviour. Nature 451, 1004–1007 (2008).
Jessberger, S. et al. Dentate gyrus-specific knockdown of adult neurogenesis impairs spatial and object recognition memory in adult rats. Learn. Mem. 16, 147–154 (2009).
Madsen, T. M., Kristjansen, P. E., Bolwig, T. G. & Wortwein, G. Arrested neuronal proliferation and impaired hippocampal function following fractionated brain irradiation in the adult rat. Neuroscience 119, 635–642 (2003).
Snyder, J. S., Hong, N. S., McDonald, R. J. & Wojtowicz, J. M. A role for adult neurogenesis in spatial long-term memory. Neuroscience 130, 843–852 (2005).
Imayoshi, I. et al. Roles of continuous neurogenesis in the structural and functional integrity of the adult forebrain. Nature Neurosci. 11, 1153–1161 (2008).
Warner-Schmidt, J. L., Madsen, T. M. & Duman, R. S. Electroconvulsive seizure restores neurogenesis and hippocampus-dependent fear memory after disruption by irradiation. Eur. J. Neurosci. 27, 1485–1493 (2008).
Winocur, G., Wojtowicz, J. M., Sekeres, M., Snyder, J. S. & Wang, S. Inhibition of neurogenesis interferes with hippocampus-dependent memory function. Hippocampus 16, 296–304 (2006).
Ko, H. G. et al. Effect of ablated hippocampal neurogenesis on the formation and extinction of contextual fear memory. Mol. Brain 2, 1 (2009).
Ben Abdallah, N. M., Slomianka, L., Vyssotski, A. L. & Lipp, H. P. Early age-related changes in adult hippocampal neurogenesis in C57 mice. Neurobiol. Aging 31, 151–161.
Seki, T. & Arai, Y. Age-related production of new granule cells in the adult dentate gyrus. Neuroreport 6, 2479–2482 (1995).
Raber, J. et al. Radiation-induced cognitive impairments are associated with changes in indicators of hippocampal neurogenesis. Radiat. Res. 162, 39–47 (2004).
Rola, R. et al. Radiation-induced impairment of hippocampal neurogenesis is associated with cognitive deficits in young mice. Exp. Neurol. 188, 316–330 (2004).
Garthe, A., Behr, J. & Kempermann, G. Adult-generated hippocampal neurons allow the flexible use of spatially precise learning strategies. PLoS One 4, e5464 (2009).
Nakashiba, T., Young, J. Z., McHugh, T. J., Buhl, D. L. & Tonegawa, S. Transgenic inhibition of synaptic transmission reveals role of CA3 output in hippocampal learning. Science 319, 1260–1264 (2008).
Brun, V. H. et al. Impaired spatial representation in CA1 after lesion of direct input from entorhinal cortex. Neuron 57, 290–302 (2008).
Gilbert, P. E., Kesner, R. P. & Lee, I. Dissociating hippocampal subregions: double dissociation between dentate gyrus and CA1. Hippocampus 11, 626–636 (2001).
Saxe, M. D. et al. Paradoxical influence of hippocampal neurogenesis on working memory. Proc. Natl Acad. Sci. USA 104, 4642–4646 (2007).
Zhang, F., Aravanis, A. M., Adamantidis, A., de Lecea, L. & Deisseroth, K. Circuit-breakers: optical technologies for probing neural signals and systems. Nature Rev. Neurosci. 8, 577–581 (2007).
Kitamura, T. et al. Adult neurogenesis modulates the hippocampus-dependent period of associative fear memory. Cell 139, 814–827 (2009). The first study to show a role for adult hippocampal neurogenesis in system consolidation.
Kesner, R. P. A behavioral analysis of dentate gyrus function. Prog. Brain Res. 163, 567–576 (2007).
Manganas, L. N. et al. Magnetic resonance spectroscopy identifies neural progenitor cells in the live human brain. Science 318, 980–985 (2007).
Pereira, A. C. et al. An in vivo correlate of exercise-induced neurogenesis in the adult dentate gyrus. Proc. Natl Acad. Sci. USA 104, 5638–5643 (2007).
Amaral, D. G., Scharfman, H. E. & Lavenex, P. The dentate gyrus: fundamental neuroanatomical organization (dentate gyrus for dummies). Prog. Brain Res. 163, 3–22 (2007).
Baker, J. L. Is there a support vector machine hiding in the dentate gyrus? Neurocomputing 52–54, 199–207 (2003).
Houser, C. R. Interneurons of the dentate gyrus: an overview of cell types, terminal fields and neurochemical identity. Prog. Brain Res. 163, 217–232 (2007).
Jung, M. W. & McNaughton, B. L. Spatial selectivity of unit activity in the hippocampal granular layer. Hippocampus 3, 165–182 (1993).
Myers, C. E. & Scharfman, H. E. A role for hilar cells in pattern separation in the dentate gyrus: a computational approach. Hippocampus 19, 321–337 (2009).
Henze, D. A., Wittner, L. & Buzsaki, G. Single granule cells reliably discharge targets in the hippocampal CA3 network in vivo. Nature Neurosci. 5, 790–795 (2002).
Rolls, E. T. & Kesner, R. P. A computational theory of hippocampal function, and empirical tests of the theory. Prog. Neurobiol. 79, 1–48 (2006).
Hopfield, J. J. Neural networks and physical systems with emergent collective computational abilities. Proc. Natl Acad. Sci. USA 79, 2554–2558 (1982).
Dupret, D. et al. Methylazoxymethanol acetate does not fully block cell genesis in the young and aged dentate gyrus. Eur. J. Neurosci. 22, 778–783 (2005).
Monje, M. L., Toda, H. & Palmer, T. D. Inflammatory blockade restores adult hippocampal neurogenesis. Science 302, 1760–1765 (2003).
Garcia, A. D., Doan, N. B., Imura, T., Bush, T. G. & Sofroniew, M. V. GFAP-expressing progenitors are the principal source of constitutive neurogenesis in adult mouse forebrain. Nature Neurosci. 7, 1233–1241 (2004).
Farioli-Vecchioli, S. et al. The timing of differentiation of adult hippocampal neurons is crucial for spatial memory. PLoS Biol. 6, e246 (2008).
Acknowledgements
We thank M. L. Gage for editorial comments. This work is funded by the James S. McDonnell Foundation, the Lookout Fund, the Kavli Institute for Brain and Mind, the NSF Temporal Dynamics of Learning Center, the US National Institutes of Health (NS-050217) and National Institute on Aging (AG-020938).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
F.H.G. is on the scientific advisory boards of Stem Cell Inc., Ceregene Therapeutics and Brain Cells Inc.
Supplementary information
Supplementary Information S1 (Table)
In format provided by Deng et al. (MAY 2010) (XLS 44 kb)
Related links
Glossary
- Filopodia
-
Thin, long and highly motile protrusions that are the predecessors of spines in an early stage of spine formation.
- Thorny excrescences
-
The complex spines on the dendrites of CA3 pyramidal neurons in the stratum lucidum. These spines form multiple synapses with mossy fibres of dentate granule cells.
- Morris water maze (MWM)
-
A spatial learning paradigm in which an animal must learn a fixed location of a platform using distal spatial cues. Animals are released from a variable start point in each trial to encourage them to use a spatial strategy to solve the task.
- BrdU birth-dating
-
The thymidine analogue bromodeoxyuridine (BrdU) is injected into adult animals and incorporated into cells synthesizing DNA in preparation for division, which are visualized using immunocytochemistry. Because the in vivo half-life of BrdU is ∼2 hours, it only labels dividing cells in a short time window.
- Sparse coding
-
A type of neural code in which each event is encoded by the strong activation of a small set of neurons.
- Attractor
-
A stable point in a dynamic system. Attractors are typically found in neural networks with strong feedback connections and are determined by the weights of the recurrent connections between units (neurons) in the network. Depending on the initial conditions and external inputs, the network will evolve towards one of these stable states.
- Pattern integration
-
The ability of immature dentate granule cells to provide an association between events owing to their indiscriminate responses to inputs.
- Pattern completion
-
A process by which a stored neural representation is reactivated by a cue that consists of a subset of that representation.
- Trace conditioning
-
A form of classical conditioning in which the conditioned stimulus occurs before the unconditioned stimulus with a stimulus-free period (the 'trace interval' or 'conditioning interval') between the two.
- Delay conditioning
-
A form of classical conditioning in which the onset of the conditioned stimulus precedes the onset of the unconditioned stimulus, with an overlap between the presentation of the conditioned stimulus and the presentation of the unconditioned stimulus.
- Recognition memory
-
The ability to correctly remember something that has been previously encountered. It is a subcategory of declarative memory.
- Contextual fear conditioning
-
A form of conditioning in which animals associate the conditioning context (the 'neutral' conditioned stimulus) with an aversive stimulus — for example, a foot shock.
- Spatial discrimination
-
The ability to discriminate separate locations in space.
- Conjunctive encoding
-
A form of information encoding in which a neuron requires the concurrent activity of multiple input neurons. In the hippocampus, dentate granule cells can associate spatial information from the medial entorhinal cortex with non-spatial information from the lateral entorhinal cortex to form a multi-dimensional representation of an event.
Rights and permissions
About this article
Cite this article
Deng, W., Aimone, J. & Gage, F. New neurons and new memories: how does adult hippocampal neurogenesis affect learning and memory?. Nat Rev Neurosci 11, 339–350 (2010). https://doi.org/10.1038/nrn2822
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrn2822
This article is cited by
-
Neurotrophin-3 from the dentate gyrus supports postsynaptic sites of mossy fiber-CA3 synapses and hippocampus-dependent cognitive functions
Molecular Psychiatry (2024)
-
The functional and structural changes in the hippocampus of COVID-19 patients
Acta Neurologica Belgica (2023)
-
Application of P7C3 Compounds to Investigating and Treating Acute and Chronic Traumatic Brain Injury
Neurotherapeutics (2023)
-
Effect of Heat Stress on Hippocampal Neurogenesis: Insights into the Cellular and Molecular Basis of Neuroinflammation-Induced Deficits
Cellular and Molecular Neurobiology (2023)
-
Experiences Shape Hippocampal Neuron Morphology and the Local Levels of CRHR1 and OTR
Cellular and Molecular Neurobiology (2023)