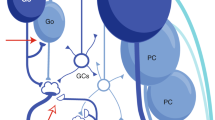
Abstract
The intricate neuronal circuitry of the cerebellum is thought to encode internal models that reproduce the dynamic properties of body parts. These models are essential for controlling the movement of these body parts: they allow the brain to precisely control the movement without the need for sensory feedback. It is thought that the cerebellum might also encode internal models that reproduce the essential properties of mental representations in the cerebral cortex. This hypothesis suggests a possible mechanism by which intuition and implicit thought might function and explains some of the symptoms that are exhibited by psychiatric patients. This article examines the conceptual bases and experimental evidence for this hypothesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Poon, C.-S. & Merfeld, D. M. Internal model: the state of art. J. Neural Eng. 2 (2005).
Ito, M. Neurophysiological basis of the cerebellar motor control system. Int. J. Neurol. 7, 162–176 (1970).
Ito, M. The Cerebellum and Neural Control (Raven, New York, 1984).
Kawato, M., Furukawa, K. & Suzuki, R. A hierarchical neural-network model for control and learning of voluntary movement. Biol. Cybern. 57, 169–185 (1987).
Miall, R. C., Weir, D. J., Wolpert, D. M. & Stein, J. F. Is the cerebellum a Smith predictor? J. Motor Behav. 25, 203–216 (1993).
Wolpert, D. M. & Kawato, M. Multiple paired forward and inverse models for motor control. Neural Netw. 11, 1317–1329 (1998).
Wolpert, D. M., Miall, R. C. & Kawato, M. Internal models in the cerebellum. Trends Cogn. Sci. 2, 338–347 (1998).
Kawato, M. Cerebellum: models. In New Encyclopedia of Neuroscience (ed. Squire, L.) (Elsevier, in the press 2008).
Schmahmann, J. D. Disorders of the cerebellum: ataxia, dysmetria of thought, and the cerebellar cognitive affective syndrome. J. Neuropsychiatry Clin. Neurosci. 16, 367–378 (2004).
Leiner, H. C., Leiner, A. L. & Dow, R. S. Does the cerebellum contribute to mental skills? Behav. Neurosci. 100, 443–454 (1986).
Leiner, H. C., Leiner, A. L. & Dow, R. S. Cognitive and language functions of the human cerebellum. Trends Neurosci. 16, 444–447 (1993).
Sasaki, K., Kawaguchi, S., Oka, H., Sakai, M. & Mizuno, N. Electrophysiological studies on the cerebellocerebral projections in monkeys. Exp. Brain Res. 24, 495–507 (1976).
Schmahmann, J. D. An emerging concept: the cerebellar contribution to higher function. Arch. Neurol. 48, 1178–1187 (1991).
Ito, M. A new physiological concept on cerebellum. Rev. Neurol. (Paris) 146, 564–569 (1990).
Ito, M. in The Principles of Design and Operation of the Brain (Experimental Brain Research) (eds Eccles, J. C. & Creutzfeldt, O.) 281–292 (Springer, 1990).
Ito, M. Movement and thought: identical control mechanisms by the cerebellum. Trends Neurosci. 16, 448–450 (1993).
Ito, M. in Cerebellum and Cognition (International Review of Neurobiology) (eds Schmahmann, J. D., Bradley, R. J., Adron Harris, R. & Jenner, P.) 475–487 (Academic, 1997).
Ito, M. Bases and implications of learning in the cerebellum – adaptive control and internal model mechanism. Prog. Brain Res. 148, 95–109 (2006).
Vandervert, L. R., Schlimp, P. H. & Liu, H. How working memory and the cerebellum collaborate to produce creativity and innovation. Creativity Res. J. 19, 1–18 (2007).
Wolpert, D. M., Doya, K. & Kawato, M. A unifying computational framework for motor control and social interaction. Philos. Trans. R. Soc. Lond. B Biol. Sci. 358, 593–602 (2003).
Kelly, R. M. & Strick, P. L. Cerebellar loops with motor cortex and prefrontal cortex of a nonhuman primate. J. Neurosci. 23, 8432–8444 (2003).
Dum, R. P. & Strick, P. L. An unfolded map of the cerebellar dentate nucleus and its projections to the cerebral cortex. J. Neurophysiol. 89, 634–639 (2003).
Clower, D. M., Dum, R. P. & Strick, P. L., Basal ganglia and cerebellar inputs to 'AIP'. Cereb. Cortex 15, 913–920 (2005).
Fang, P.-C., Stepniewska, I. & Kaas, J. H. Ispilateral cortical connections of motor, premotor, frontal eye, and posterior parietal fields in a prosimian primate, Otalemur garnetti. J. Comp. Neurol. 490, 305–333 (2005).
Ramnani, N. The primate cortico-cerebellar system: anatomy and function. Nature Rev. Neurosci. 7, 511–522, (2006).
Fiez, J. A., Raicle, M. E., Balota, D. A., Tallal, P. & Petersen, S. E. PET activation of posterior temporal regions during auditory word presentation and verb generation. Cereb. Cortex 6, 1–10 (1996).
Hanakawa, T., Honda, M., Okada, T., Fukuyama, H. & Shibasaki, H. Neural correlates underlying mental calculation in abacus experts: a functional magnetic resonance imaging study. Neuroimage 19, 296–307 (2003).
Baker, S. C. et al. Neural systems engaged by planning: a PET study of the Tower of London task. Neuropsychologia 34, 515–526 (1996).
Ploghaus, A. et al. Learning about pain: the neural substrate of the prediction error for aversive events. Proc. Natl Acad. Sci. USA 97, 9281–9286 (2000).
Allen, G. & Courchesne, E. Differential effects of developmental cerebellar abnormality on cognitive and motor functions in the cerebellum: an fMRI study of autism. Am. J. Psychiatry 160, 262–273 (2003).
Schlosser, R. et al. Functional magnetic resonance imaging of human brain activity in a verbal fluency task. J. Neurol. Neurosurg. Psychiatr. 64, 492–498 (1998).
de Zubicaray, G. I. et al. Prefrontal cortex involvement in selective letter generation: a functional magnetic resonance imaging study. Cortex 34, 389–401 (1998).
Desmond, J. E., Gabrieli, J. D. E., Wagner, A. D., Ginier, B. L. & Glove, G. H. Lobular patterns of cerebellar activation in verbal working memory and finger-tapping tasks as revealed by functional MRI. J. Neurosci. 17, 9675–9685 (1997).
Hayter, A. L., Lamgton, D. W. & Ramnani, N. Cerebellar contributions to working memory. Neuroimage 36, 943–954 (2007).
Nagahama, Y. et al. Cerebral activation during performance of a card sorting test. Brain 119, 1667–1675 (1996).
Narita, H., Odawara, T., Iseki, E., Kosaka, K. & Hirayasu, Y. Psychomotor retardation correlates with frontal hypoperfusion and the Modified Stroop Test in patients with major depression under 60-years-old. Psychiat. Clin. Neurosci. 58, 389–395 (2004).
Atherton, M., Zhung, J., Bart, W. M., Hu, X. & He, S. A functional MRI study of high-level cognition. I. The game of chess. Cogn. Brain Res. 16, 26–31 (2003).
Chen, X., Zhang, D., Zhang, X., Li, Z. & Meng, X. A functional MRI study of high-level cognition. II. The game of GO. Cogn. Brain Res. 16, 32–37 (2003).
Szpunar, K. K., Watson, J. M. & McDermott, K. B. Neural substrates of envisioning the future. Proc. Natl Acad. Sci. USA 2, 642–647 (2007).
Collinson, S. L., Anthonisz, B., Courtenay, D. & Winter, C. Frontal executive impairment associated with paraneoplastic cerebellar degeneration: a case study. Neurocase 12, 350–354 (2006).
Janssen, G. et al. Cerebellar mutism syndrome. Klin. Padiatr. 210, 243–247 (1998).
Fiez, J. A., Petersen, S. E., Cheney, M. K. & Raichle, M. E. Impaired non-motor learning and error detection associated with cerebellar damage. A single case study. Brain 115, 155–178 (1992).
Gebhart, A. L., Petersen, S. E & Thach, W. T. Role of the posterolateral cerebellum in language. Ann. NY Acad. Sci. 978, 318–333 (2002).
Leggio, M. G., Silveri, M. C., Petrosini, L. & Molinari, M. Phonological grouping is specifically affected in cerebellar patients; a verbal fluency study. J. Neurol. Neurosurg. Psychiatr. 69, 102–106 (2000).
Nicolson, R. I., Daum, I., Schugens, M. M., Fawcett, A. J. & Schulz, A. Eyeblink conditioning indicates cerebellar abnormality in dyslexia. Exp. Brain Res. 143, 42–50 (2002).
Stoodley, C. J., Fawcett, A. J., Nicolson, R. I. & Stein, J. F. Balancing and pointing tasks in dyslexic and control adults. Dyslexia 12, 276–288 (2006).
Bigelow, N. O. et al. Prism adaptation in schizophrenia. Brain Cogn. 61, 235–242 (2006).
Blakemore, S.-J., Smith, J., Steel, R., Johnstone, E. C. & Frith, C. D. The perception of self-produced sensory stimuli in patients with auditory hallucinations and passivity experiences: evidence for a breakdown in self-monitoring. Psychol. Med. 30, 1131–1139 (2000).
Miller, E. K. & Cohen, J. D. An integrative theory of prefrontal cortex function. Annu. Rev. Neurosci. 24, 167–202 (2001).
Happaney, K., Zelazo, P. D. & Stuss, D. T. Development of orbitofrontal function: current theme and future directions. Brain Cogn. 55, 1–10 (2004).
Baddeley, A. Working memory: looking back and looking forward. Nature Rev. Neurosci. 4, 829–839 (2003).
Baddeley, A. & Hitch, G. J. in Recent Advances in Learning and Motivation (ed. Bower, G. A.) 47–89 (Academic, New York, 1974).
Smith, E. E. & Jonides, J. Working memory: a view from neuroimaging. Cognit. Psychol. 33, 5–42 (1997).
Stuss, D. T. & Knight, R. T. Principles of Frontal Lobe Function (Oxford Univ. Press, New York, 2002).
Baddeley, A. D. & Logie, R. H. in Models of Working Memory (eds Miyake, A. & Shah, P.) 28–61 (Cambridge Univ. Press, Cambridge, UK, 1999).
Craik, K. J. W. The Nature of Explanation (Cambridge Univ. Press, Cambridge, UK, 1943).
Johnson-Raird, P. N. Mental Models (Cambridge Univ. Press, 1983).
Held, C., Knauff, M. & Vosgerau, G. Mental Models and the Mind: Current Developments in Cognitive Psychology, Neuroscience, and Philosophy of Mind (Advances in Psychology) (Elsevier, 2006).
Tanaka, K. Inferotemporal cortex and object vision. Ann. Rev. Neurosci. 19, 109–139 (1996).
Tsunoda, K., Yamane, Y., Nishizaka, M. & Tanifuji, M. Complex objects are represented in the macaque inferotemporal cortex by the combination of feature columns. Nature Neurosci. 4, 832–838 (2001).
Sheinberg., D. L. & Logothetis, N.K. The role of temporal areas in perceptual organization. Proc. Natl Acad. Sci. USA 94, 3408–3413 (1997).
Tarok, L. & Srinivasan, R. Transient MEG frequency-tagging response induced by switching stimulus salience during binocular rivalry. (Abstr.) 201.9/BBB20 (Society for Neuroscience Annual Meeting, San Diego, 2007).
Kosslyn, S. M., Ganis, G. & Thompson, W. L. Neural foundations of imagery. Nature Rev. Neurosci. 2, 635–642 (2001).
Pearson, D., De Beni, R. & Cornoldi, C. in Imagery, Language and Visuo-Spatial Thinking (eds Denis, M., Logie, R. H., Cornoldi, C., De Vega, M. & Engelkamp, J.) 1–27 (Psychological Press, 2001).
Hesslow, G. Conscious thought as simulation of behaviour and perception. Trends Cogn. Sci. 6, 242–247 (2002).
Le Bihan, D. et al. Activation of human primary visual cortex during visual recall: a magnetic resonance imaging study. Proc. Natl Acad. Sci. USA 90, 11802–11805 (1993).
Miyashita, Y. Cognitive memory: cellular and network machineries and their top-down control. Science 306, 435–440 (2004).
Hasegawa, I., Fukushima, T., Ihara, T. & Miyashita, Y. Callosal window between prefrontal cortices: cognitive interaction to retrieve long-term memory. Science 281, 814–818 (1998).
Penfield, W. & Perot, P. The brain's record of auditory and visual experience; a final summary and discussion. Brain 86, 595–696 (1963).
Fuster, J. M. The Prefrontal Cortex: Anatomy, Physiology, and Neuropsychology of the Frontal Lobe (Raven, New York, 1997).
Ito, M. Cerebellar long-term depression: characterization, signal transduction and functional roles. Physiol. Rev. 81, 1143–1195 (2001).
Ito, M. Cerebellar circuitry as a neuronal machine. Prog. Neurobiol. 78, 272–303 (2006).
Ralston, D. D. Corticorubral synaptic organization in Macaca fascicularis: a study utilizing degeneration, anterograde transport of WGA-HRP, and combined immuno-GABA-gold technique and computer-assisted reconstruction. J. Comp. Neurol. 350, 657–673 (1994).
Burman, K., Darian-Smith, C. & Darian-Smith, I. Macaque red nucleus: origins of spinal and olivary projections and terminations of cortical inputs. J. Comp. Neurol. 423, 178–196 (2000).
Burman, K., Darian-Smith, C. & Darian-Smith, I. Geometry of rubrospinal, rubroolivary, and local circuit neurons in the macaque red nucleus. J. Comp. Neurol. 423, 197–219, (2000).
Oka, H., Jinnai, K. & Yamamoto, T. The parieto-rubro-olivary pathway in the cat. Exp. Brain Res. 37, 115–125 (1979).
Yttri, E., Smith, A., Reid, E. & Thach, W. T. Inactivation of parvocellular red nucleus impairs prism adaptation to and learning of contralateral gaze-reach shift. (Abstr.) 440.15/K8 (Society for Neuroscience Annual Meeting, Atlanta, 2006).
Schmahmann, J. D. & Pandya, D. N. Anatomical investigation of projections to the basis pontis from posterior parietal association cortices in rhesus monkey. J. Comp. Neurol. 289, 53–73 (1989).
Shidara, M., Kawano, M., Gomi, H. & Kawato, M. Inverse-dynamics encoding of eye movements by Purkinje cells in the cerebellum. Nature 365, 50–52 (1993).
Miles, G. B., Cerminara, N. L. & Marple-Horvat, D. E. Purkinje cells in the lateral cerebellum of the cat encode visual events and target motion during visually guided reaching. J. Physiol. 571, 619–637 (2006).
Pasalar, S., Roitman, A. V., Durfee, W. K. & Ebner, T. J. Force field effects on cerebellar Purkinje cell discharge with implications for internal models. Nature Neurosci. 9, 1404–1411 (2006).
Yamamoto, K., Kawato, M., Kotosaka, S. & Kitazawa, S. Encoding of movement dynamics by Purkinje cell simple spike activity during fast arm movements under resistive and assistive force fields. J. Neurophysiol. 97, 1588–1599 (2007).
Schmajuk, N. A., Lam, Y.-W. & Gray, J. A. Latent inhibition: a neural network approach. J. Exp. Psychol. 22, 321–349 (1996).
Lisman, J. E. & Grace, A. A. The hippocampal-VTA loop: controlling the entry of information into long-term memory. Neuron 46, 703–713 (2005).
Miller, E. K., Erickson, C. A. & Desimone, R. Neural mechanisms of visual working memory in prefrontal cortex of the macaque. J. Neurosci. 16, 5154–5167 (1996).
Einstein, A. Letters a Maurice Solvince (Gauthier-Villars, Paris, 1956).
Spriner, S. P. & Deutsch, G. Left Brain, Right Brain: Perspectives on Cognitive Neuroscience 5th edn (Freeman, New York, 1998).
Diamond, A. Close interrelation of motor development and cognitive development and the cerebellum and prefrontal cortex. Child Develop. 71, 44–56 (2000).
Carper, R. A. & Courchesne, E. Inverse correlation between frontal lobe and cerebellum sizes in children with autism. Brain 123, 836–844 (2000).
Fetami, S. H. et al. Purkinje cell size is reduced in cerebellum of patients with autism. Cell. Mol. Neurosci. 22, 171–175 (2002).
Lee, M. et al. Nicotinic receptor abnormalities in the cerebellar cortex in autism. Brain 125, 1483–1495 (2002).
Sadakata, T. et al. Autistic-like phenotypes in Cadps 2-knockout mice and aberrant CADPS2 splicing in austistic patients. J. Clin. Invest. 117, 931–943 (2007).
Sadakata, T. et al. Impaired cerebellar development and function in mice lacking CAPS2, a protein involved in neurotrophin release. J. Neurosci. 27, 2472–2482 (2007).
Deuel, R. K. Autism: a cognitive developmental riddle. Pediatr. Neurol. 26, 349–357 (2002).
Ito, M. Nurturing the brain as an emerging research field involving child neurology. Brain Dev. 26, 429–433 (2004).
Sperling, A. J., Lu, Z. L., Manis, F. R. & Seidenberg, M. S. Deficits in perceptual noise exclusion in developmental dyslexia. Nature Neurosci. 8, 862–863 (2005).
Galaburda, A. M., Sherman, G. F., Rosen, G. D., Aboitiz, F. & Geschwind, N. Developmental dyslexia: four consecutive patients with cortical anomalies. Ann. Neurol. 18, 222–233 (1985).
Stein, J. & Walsh, V. To see but not to read: the magnocellular theory of dyslexia. Trends Neurosci. 20, 147–152 (1997).
Blakemore, S.-J., Wolpert, D. M. & Frith, C. D. Central cancellation of self-produced tickle sensation. Nature Neurosci. 1, 635–640 (1998).
Blakemore, S. J. & Sirigu, A. Action prediction in the cerebellum and in the parietal lobe. Exp. Brain Res. 153, 239–245 (2003).
Solms, M. & Turnbull, O. The Brain and the Inner World. (OTHER, 2002).
Wegner, D. M. The Illusion of Conscious Will (MIT Press, Cambridge, Massachusetts, 2002).
Crick, F. & Koch, C. Towards a neurobiological theory of consciousness. Semin. Neurosci. 2, 263–275 (1990).
Crick, F., Koch, C., Kreiman, G. & Fried, I. Consciousness and neurosurgery. Neurosurgery 55, 273–282 (2004).
Barlow, J. S. The Cerebellum and Adaptive Control (Cambridge Univ. Press, New York, 2002).
Marr, D. A theory of cerebellar cortex. J. Physiol. 202, 437–470 (1969).
Albus, J. S. A theory of cerebellar function. Math. Biosci. 10, 25–61 (1971).
Fujita, M. Adaptive filter model of the cerebellum. Biol. Cybern. 45, 195–206 (1982).
Mauk, M. D. & Donegan, N. H. A model of Pavlovian eyelid conditioning based on the synaptic organization of the cerebellum. Learn. Mem. 4, 130–158 (1997).
Yamazaki, T. & Tanaka, S. Neural modeling of an internal clock. Neural Comput. 17, 1–27 (2005).
Yamazaki, T. & Tanaka, S. The cerebellum as a liquid state machine. Neural Netw. 20, 290–297 (2007).
Schweighofer, N., Doya, K. & Kuroda, S. Cerebellar aminergic neuromodulation: towards a functional understanding. Brain Res. Rev. 44, 103–116 (2004).
Nagao, S., Ito, M. & Karachot, L. Eye field in the cerebellar flocculus of pigmented rabbits determined with local electrical stimulation. Neurosci. Res. 3, 39–51 (1985).
Christian, K. M. & Thompson, R. F. Neural substrates of eyeblink conditioning: acquisition and retention. Learn. Mem. 10, 427–455 (2003).
Apps, R. & Garwicz, M. Anatomical and physiological foundations of cerebellar information processing. Nature Rev. Neurosci. 6, 297–311 (2005).
Riklan, M., Cullinan, T., Shulman, M. & Cooper, I. S. A psychometric study of chronic cerebellar stimulation in man. Biol. Psychiatry 11, 543–574 (1976).
Koch, G. et al. Repetitive TMS of cerebellum interferes with millisecond time processing. Exp. Brain Res. 179, 291–299 (2007).
Acknowledgements
I would like to thank the RIKEN Brain Science Institute for their continuous support of my research on the cerebellum.
Author information
Authors and Affiliations
Related links
Glossary
- Body-part dynamics
-
The dynamic properties of a body part that are determined by physical factors such as weight, length, centre of gravity and viscosity. The dynamic properties in turn determine the movement of the body part in response to command signals.
- Column
-
A basic functional unit of the cerebral cortex. Each column is approximately 0.5 mm wide and 2 mm high and contains approximately 10,000 neurons. These units operate much like microcircuits in a computer. The human cerebral cortex is thought to have approximately a million columns.
- Control system
-
A term that was originally used to refer to a mechanical or chemical system equipped with a mechanism for manipulating an object or regulating a process. The term now broadly applies to an informational, biological, neural, psychological or social system.
- Controlled object
-
A key part of a control system, a controlled object converts a command into an output action. For example, a muscle converts signals in nerves into a contraction.
- Controller
-
A key part of a control system, a controller converts a given instruction into a command. For example, the brain converts an instructed spatial position of a target into a command, which consists of signals in the nerves that innervate muscles.
- Error signals
-
Signals representing errors in a system. The errors are discrepancies in the performance of a control system from either the instruction (consequence errors) or the prediction by an internal model (internal errors).
- Instructor
-
The part of a control system that supplies an instruction to the controller. The instructor gives a goal towards which a control system should work.
- Internal model
-
A functional dummy of a body part or of a mental representation in the cerebral cortex. Internal models are encoded in the neuronal circuitry of the cerebellum and mimic the essential properties of a body part or mental representation.
- Paced auditory serial-addition test
-
(PASAT). A test that is used to impose a high cognitive load on the working memory. Subjects receive a pseudo-random auditory presentation of a number between 1 and 9 every 3 seconds and are asked to add consecutive numbers and provide the answer to each addition verbally.
- Stroop Task
-
A task in which subjects are instructed to either read words or name the colour in which the words are written. Subjects must selectively attend to one attribute, particularly when naming the colour of a conflict stimulus (for example, the word 'green' displayed in red).
- Tower of London Task
-
A test of planning capability. Typically, starting from an initial condition in which three differently coloured rings are distributed to three poles, the subject is asked to gather all of the rings to one particular pole by moving one at a time and not making a total of more than five moves. A modified version is used in neuroimaging to avoid contamination of the results with activity relating to movements.
- Unit learning machine
-
The cerebellum contains numerous modular units, each of which consists of a uniform set of neuronal circuits that is capable of learning. Each unit learning machine is inserted into a neural control system and carries out the role of an internal model.
- Wisconsin Card-Sorting Task
-
(WCST). In this task, participants are given cards that can be sorted by colour, shape or name, and must deduce the correct sorting criterion. After several consecutive correct sorts, the correct sorting criterion is changed without warning.
Rights and permissions
About this article
Cite this article
Ito, M. Control of mental activities by internal models in the cerebellum. Nat Rev Neurosci 9, 304–313 (2008). https://doi.org/10.1038/nrn2332
Issue Date:
DOI: https://doi.org/10.1038/nrn2332
This article is cited by
-
Emergence of syntax and word prediction in an artificial neural circuit of the cerebellum
Nature Communications (2024)
-
Cerebellar state estimation enables resilient coupling across behavioural domains
Scientific Reports (2024)
-
Effects of glabellar botulinum toxin injections on resting-state functional connectivity in borderline personality disorder
European Archives of Psychiatry and Clinical Neuroscience (2024)
-
Cerebellar Neurostimulation for Boosting Social and Affective Functions: Implications for the Rehabilitation of Hereditary Ataxia Patients
The Cerebellum (2024)
-
Study protocol: effects of treatment expectation toward repetitive transcranial magnetic stimulation (rTMS) in major depressive disorder—a randomized controlled clinical trial
Trials (2023)