Key Points
-
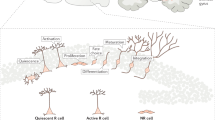
The subventricular zone (SVZ) maintains its ability to produce new neurons and glial cells throughout life.
-
In response to Huntington's disease, the number of proliferating progenitor cells in the SVZ increases approximately threefold and correlates with the advancing pathology in the striatum.
-
In the SVZ in Huntington's disease, the levels of mitogenic compounds such as neuropeptide Y and cannabinoid CB1 receptors are increased, indicating that SVZ cells are preserved despite the nearby pathology.
-
In Parkinson's disease, the nigrostriatal pathway degenerates and, as a result of lowered dopaminergic inputs to the type C cells, there is a reduction in progenitor cell proliferation in the SVZ. Experimentally this can be restored by administering dopaminergic agonists to the SVZ.
-
In the SVZ in Alzheimer's disease, the amyloid plaques appear to be toxic to the progenitor cells and, thus, progenitor cells cannot survive in the SVZ in the presence of this pathology. It is not clear whether the SVZ attempts to increase progenitor cell proliferation, but the net result is decreased SVZ proliferation due to b-amyloid toxicity.
-
The introduction of mitogenic factors to the SVZ progenitors leads to increased proliferation and migration towards sites of injury in the brain. The delivery of a combination of mitogenic factors or transcription factors to the SVZ by viral vectors may lead to improved progenitor cell replacement at the injury site.
Abstract
During brain development, one of the most important structures is the subventricular zone (SVZ), from which most neurons are generated. In adulthood the SVZ maintains a pool of progenitor cells that continuously replace neurons in the olfactory bulb. Neurodegenerative diseases induce a substantial upregulation or downregulation of SVZ progenitor cell proliferation, depending on the type of disorder. Far from being a dormant layer, the SVZ responds to neurodegenerative disease in a way that makes it a potential target for therapeutic intervention.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Deacon, T. W., Pakzaban, P. & Isacson, O. The lateral ganglionic eminence is the origin of cells committed to striatal phenotypes: neural transplantation and developmental evidence. Brain Res. 668, 211–219 (1994).
Freeman, T. B., Sanberg, P. R. & Isacson, O. Development of the human striatum: implications for fetal striatal transplantation in the treatment of Huntington's disease. Cell Transplant. 4, 539–545 (1995).
Ohtani, N., Goto, T., Waeber, C. & Bhide, P. G. Dopamine modulates cell cycle in the lateral ganglionic eminence. J. Neurosci. 23, 2840–2850 (2003).
Olsson, M., Bjorklund, A. & Campbell, K. Early specification of striatal projection neurons and interneuronal subtypes in the lateral and medial ganglionic eminence. Neuroscience. 84, 867–876 (1998).
Smart, I. H. A pilot study of cell production by the ganglionic eminences of the developing mouse brain. J. Anat. 121, 71–84 (1976).
Wichterle, H., Turnbull, D. H., Nery, S., Fishell, G. & Alvarez-Buylla, A. In utero fate mapping reveals distinct migratory pathways and fates of neurons born in the mammalian basal forebrain. Development 128, 3759–3771 (2001).
Arvidsson, A., Collin, T., Kirik, D., Kokaia, Z. & Lindvall, O. Neuronal replacement from endogenous precursors in the adult brain after stroke. Nature Med. 8, 963–970 (2002). Shows the first experimental evidence that the SVZ can substantially upregulate the production of progenitor cells in response to stroke and that the progenitors migrate and develop into specific neuron types.
Bedard, A., Levesque, M., Bernier, P. J. & Parent, A. The rostral migratory stream in adult squirrel monkeys: contribution of new neurons to the olfactory tubercle and involvement of the antiapoptotic protein Bcl-2. Eur. J. Neurosci. 16, 1917–1924 (2002).
Luskin, M. B. Restricted proliferation and migration of postnatally generated neurons derived from the forebrain subventricular zone. Neuron 11, 173–189 (1993).
Jin, K. et al. Evidence for stroke-induced neurogenesis in the human brain. Proc. Natl Acad. Sci. USA 103, 13198–13202 (2006).
Parent, J. M., Valentin, V. V. & Lowenstein, D. H. Prolonged seizures increase proliferating neuroblasts in the adult rat subventricular zone–olfactory bulb pathway. J. Neurosci. 22, 3174–3188 (2002).
Parent, J. M., Vexler, Z. S., Gong, C., Derugin, N. & Ferriero, D. M. Rat forebrain neurogenesis and striatal neuron replacement after focal stroke. Ann. Neurol. 52, 802–813 (2002).
Eriksson, P. S. et al. Neurogenesis in the adult human hippocampus. Nature Med. 4, 1313–1317 (1998). The first demonstration of adult human neurogenesis.
Haydar, T. F., Wang, F., Schwartz, M. L. & Rakic, P. Differential modulation of proliferation in the neocortical ventricular and subventricular zones. J. Neurosci. 20, 5764–5774 (2000).
Privat, A. & Leblond, C. P. The subependymal layer and neighboring region in the brain of the young rat. J. Comp. Neurol. 146, 277–302 (1972).
Smart, I. H. Proliferative characteristics of the ependymal layer during the early development of the mouse diencephalon, as revealed by recording the number, location, and plane of cleavage of mitotic figures. J. Anat. 113, 109–129 (1972).
Curtis, M. A., Waldvogel, H. J., Synek, B. & Faull, R. L. M. A histochemical and immunohistochemical analysis of the subependymal layer in the normal and Huntington's disease brain. J. Comp. Neurol. 30, 55–66 (2005)
Doetsch, F., Garcia-Verdugo, J. M. & Alvarez-Buylla, A. Cellular composition and three-dimensional organization of the subventricular germinal zone in the adult mammalian brain. J. Neurosci. 17, 5046–5061 (1997). Detailed account of the anatomy and cellular composition of the rodent SVZ.
Quinones-Hinojosa, A. et al. Cellular composition and cytoarchitecture of the adult human subventricular zone: a niche of neural stem cells. J. Comp. Neurol. 494, 415–434 (2006).
Bernier, P. J., Vinet, J., Cossette, M. & Parent, A. Characterization of the subventricular zone of the adult human brain: evidence for the involvement of Bcl-2. Neurosci. Res. 37, 67–78 (2000).
Lois, C., Garcia-Verdugo, J. M. & Alvarez-Buylla, A. Chain migration of neuronal precursors. Science 271, 978–981 (1996).
Doetsch, F., Caillé, I., Lim, D. A., García-Verdugo, J. M. & Alvarez-Buylla, A. Subventricular zone astrocytes are neural stem cells in the adult mammalian brain. Cell 97, 703–716 (1999).
Imura, T., Kornblum, H. I. & Sofroniew, M. V. The predominant neural stem cell isolated from postnatal and adult forebrain but not early embryonic forebrain expresses GFAP. J. Neurosci. 23, 2824–2832 (2003).
Gusella, J. F. et al. A polymorphic DNA marker genetically linked to Huntington's disease. Nature 306, 234–238 (1983).
Snell, R. G. et al. Relationship between trinucleotide repeat expansion and phenotypic variation in Huntington's disease. Nature Genet. 4, 393–397 (1993).
The Huntington's Disease Collaborative Research Group. A novel gene containing a trinucleotide repeat that is expanded and unstable on Huntington's disease chromosomes. Cell 72, 971–983 (1993).
Vonsattel, J. P. et al. Neuropathological classification of Huntington's disease. J. Neuropathol. Exp. Neurol. 44, 559–577 (1985).
van Dellen, A., Blakemore, C., Deacon, R., York, D. & Hannan, A. J. Delaying the onset of Huntington's in mice. Nature 404, 721–722 (2000).
Sathasivam, K. et al. Transgenic models of Huntington's disease. Philos. Trans. R. Soc. Lond. B Biol. Sci. 354, 963–969 (1999).
Slow, E. J. et al. Absence of behavioral abnormalities and neurodegeneration in vivo despite widespread neuronal huntingtin inclusions. Proc. Natl Acad. Sci. USA 102, 11402–11407 (2005).
Phillips, W., Morton, A. J. & Barker, R. A. Abnormalities of neurogenesis in the R6/2 mouse model of Huntington's disease are attributable to the in vivo microenvironment. J. Neurosci. 25, 11564–11576 (2005).
Tattersfield, A. S. et al. Neurogenesis in the striatum of the quinolinic acid lesion model of Huntington's disease. Neuroscience 127, 319–332 (2004).
Hockly, E. et al. Environmental enrichment slows disease progression in R6/2 Huntington's disease mice. Ann. Neurol. 51, 235–242 (2002).
Lazic, S. E. et al. Decreased hippocampal cell proliferation in R6/1 Huntington's mice. Neuroreport 15, 811–813 (2004).
Lazic, S. E. et al. Neurogenesis in the R6/1 transgenic mouse model of Huntington's disease: effects of environmental enrichment. Eur. J. Neurosci. 23, 1829–1838 (2006).
Batista, C. M. et al. A progressive and cell non-autonomous increase in striatal neural stem cells in the Huntington's disease R6/2 mouse. J. Neurosci. 26, 10452–10460 (2006).
Chiasson, B. J., Tropepe, V., Morshead, C. M. & van der Kooy, D. Adult mammalian forebrain ependymal and subependymal cells demonstrate proliferative potential, but only subependymal cells have neural stem cell characteristics. J. Neurosci. 19, 4462–4471 (1999).
Tropepe, V. et al. Distinct neural stem cells proliferate in response to EGF and FGF in the developing mouse telencephalon. Dev. Biol. 208, 166–188 (1999).
Curtis, M. A. et al. Increased cell proliferation and neurogenesis in the adult human Huntington's disease brain. Proc. Natl Acad. Sci. USA 100, 9023–9027 (2003). The first demonstration of neurogenesis and progenitor cell proliferation in the human SVZ in response to neuropathology.
Hansel, D. E., Eipper, B. A. & Ronnett, G. V. Neuropeptide Y functions as a neuroproliferative factor. Nature 410, 940–944 (2001). Shows the extremely potent effect of factors such as NPY on progenitor proliferation in the brain.
Chen, J. et al. Endothelial nitric oxide synthase regulates brain-derived neurotrophic factor expression and neurogenesis after stroke in mice. J. Neurosci. 25, 2366–2375 (2005).
Reif, A. et al. Differential effect of endothelial nitric oxide synthase (NOS-III) on the regulation of adult neurogenesis and behaviour. Eur. J. Neurosci. 20, 885–895 (2004).
Saxena, N. C. & Macdonald, R. L. Assembly of GABAA receptor subunits: role of the δ subunit. J. Neurosci. 14, 7077–7086 (1994).
LoTurco, J. J., Owens, D. F., Heath, M. J., Davis, M. B. & Kriegstein, A. R. GABA and glutamate depolarize cortical progenitor cells and inhibit DNA synthesis. Neuron 15, 1287–1298 (1995).
Curtis, M. A., Faull, R. L. & Glass, M. A novel population of progenitor cells expressing cannabinoid receptors in the subependymal layer of the adult normal and Huntington's disease human brain. J. Chem. Neuroanat. 31, 210–215 (2006).
MartÌn-Aparicio, E., Avila, J. & Lucas, J. J. Nuclear localization of N-terminal mutant huntingtin is cell cycle dependent. Eur. J. Neurosci. 16, 355–359 (2002).
Yoshizawa, T. et al. Cell cycle arrest enhances the in vitro cellular toxicity of the truncated Machado–Joseph disease gene product with an expanded polyglutamine stretch. Hum. Mol. Genet. 9, 69–78 (2000).
Lledo, P. M., Alonso, M. & Grubb, M. S. Adult neurogenesis and functional plasticity in neuronal circuits. Nature Rev. Neurosci. 7, 179–193 (2006).
Höglinger, G. U. et al. Dopamine depletion impairs precursor cell proliferation in Parkinson disease. Nature Neurosci. 7, 726–735 (2004). Demonstrates how dopaminergic inputs to the SVZ enhance progenitor cell proliferation. Conversely, dopaminergic denervation causes a reduction in progenitor cell proliferation in PD.
Winner, B. et al. Striatal deafferentation increases dopaminergic neurogenesis in the adult olfactory bulb. Exp. Neurol. 197, 113–121 (2006).
Huisman, E., Uylings, H. B. & Hoogland, P. V. A 100% increase of dopaminergic cells in the olfactory bulb may explain hyposmia in Parkinson's disease. Mov. Disord. 19, 687–692 (2004).
Baker, S. A., Baker, K. A. & Hagg, T. Dopaminergic nigrostriatal projections regulate neural precursor proliferation in the adult mouse subventricular zone. Eur. J. Neurosci. 20, 575–579 (2004).
Diaz, J. et al. Dopamine D3 receptors expressed by all mesencephalic dopamine neurons. J. Neurosci. 20, 8677–8684 (2000).
Baker, S. A., Baker, K. A. & Hagg, T. D3 dopamine receptors do not regulate neurogenesis in the subventricular zone of adult mice. Neurobiol. Dis. 18, 523–527 (2005).
Fallon, J. et al. In vivo induction of massive proliferation, directed migration, and differentiation of neural cells in the adult mammalian brain. Proc. Natl Acad. Sci. USA 97, 14686–14691 (2000).
Cooper, O. & Isacson, O. Intrastriatal transforming growth factor α delivery to a model of Parkinson's disease induces proliferation and migration of endogenous adult neural progenitor cells without differentiation into dopaminergic neurons. J. Neurosci. 24, 8924–8931 (2004). Demonstrates how a mitogenic factor can cause remarkable proliferation and migration of progenitor cells bilaterally despite a unilateral administration.
Grundke-Iqbal, I. et al. Abnormal phosphorylation of the microtubule-associated protein tau (τ) in Alzheimer cytoskeletal pathology. Proc. Natl Acad. Sci. USA 83, 4913–4917 (1986).
Mattson, M. P. Apoptosis in neurodegenerative disorders. Nature Rev. Mol. Cell Biol. 1, 120–129 (2000).
Ge, S., Yang, C. H., Hsu, K. S., Ming, G. L. & Song, H. A critical period for enhanced synaptic plasticity in newly generated neurons of the adult brain. Neuron 54, 559–566 (2007).
Denovan-Wright, E. M. & Robertson, H. A. Cannabinoid receptor messenger RNA levels decrease in a subset of neurons of the lateral striatum, cortex and hippocampus of transgenic Huntington's disease mice. Neuroscience 98, 705–713 (2000).
Jin, K. et al. Increased hippocampal neurogenesis in Alzheimer's disease. Proc. Natl Acad. Sci. USA 101, 343–347 (2004).
Boekhoorn, K., Joels, M. & Lucassen, P. J. Increased proliferation reflects glial and vascular-associated changes, but not neurogenesis in the presenile Alzheimer hippocampus. Neurobiol. Dis. 24, 1–14 (2006).
Donovan, M. H. et al. Decreased adult hippocampal neurogenesis in the PDAPP mouse model of Alzheimer's disease. J. Comp. Neurol. 495, 70–83 (2006).
Haughey, N. J., Liu, D., Nath, A., Borchard, A. C. & Mattson, M. P. Disruption of neurogenesis in the subventricular zone of adult mice, and in human cortical neuronal precursor cells in culture, by amyloid-β peptide: implications for the pathogenesis of Alzheimer's disease. Neuromolecular Med. 1, 125–135 (2002). Shows how the amyloid-β peptide is toxic to the progenitor cells in the SVZ.
Haughey, N. J. et al. Disruption of neurogenesis by amyloid-β peptide, and perturbed neural progenitor cell homeostasis, in models of Alzheimer's disease. J. Neurochem. 83, 1509–1524 (2002).
Wang, R., Dineley, K. T., Sweatt, J. D. & Zheng, H. Presenilin 1 familial Alzheimer's disease mutation leads to defective associative learning and impaired adult neurogenesis. Neuroscience 126, 305–312 (2004).
Wen, P. H. et al. The presenilin-1 familial Alzheimer disease mutant P117L impairs neurogenesis in the hippocampus of adult mice. Exp. Neurol. 188, 224–237 (2004).
Devanand, D. P. et al. Olfactory deficits in patients with mild cognitive impairment predict Alzheimer's disease at follow-up. Am. J. Psychiatry 157, 1399–1405 (2000).
Murphy, C., Gilmore, M. M., Seery, C. S., Salmon, D. P. & Lasker, B. R. Olfactory thresholds are associated with degree of dementia in Alzheimer's disease. Neurobiol. Aging 11, 465–469 (1990).
Nordin, S., Almkvist, O., Berglund, B. & Wahlund, L. O. Olfactory dysfunction for pyridine and dementia progression in Alzheimer disease. Arch. Neurol. 54, 993–998 (1997).
Serby, M., Larson, P. & Kalkstein, D. The nature and course of olfactory deficits in Alzheimer's disease. Am. J. Psychiatry 148, 357–360 (1991).
Ziabreva, I. et al. Altered neurogenesis in Alzheimer's disease. J. Psychosom. Res. 61, 311–316 (2006).
Yasuoka, K., Hirata, K., Kuraoka, A., He, J. W. & Kawabuchi, M. Expression of amyloid precursor protein-like molecule in astroglial cells of the subventricular zone and rostral migratory stream of the adult rat forebrain. J. Anat. 205, 135–146 (2004).
Hayashi, Y. et al. Alzheimer amyloid protein precursor enhances proliferation of neural stem cells from fetal rat brain. Biochem. Biophys. Res. Commun. 205, 936–943 (1994).
Ohsawa, I., Takamura, C., Morimoto, T., Ishiguro, M. & Kohsaka, S. Amino-terminal region of secreted form of amyloid precursor protein stimulates proliferation of neural stem cells. Eur. J. Neurosci. 11, 1907–1913 (1999).
Caillé, I. et al. Soluble form of amyloid precursor protein regulates proliferation of progenitors in the adult subventricular zone. Development 131, 2173–2181 (2004).
Yang, P., Baker, K. A. & Hagg, T. A disintegrin and metalloprotease 21 (ADAM21) is associated with neurogenesis and axonal growth in developing and adult rodent CNS. J. Comp. Neurol. 490, 163–179 (2005).
Becker, M., Lavie, V. & Solomon, B. Stimulation of endogenous neurogenesis by anti-EFRH immunization in a transgenic mouse model of Alzheimer's disease. Proc. Natl Acad. Sci. USA 104, 1691–1696 (2007).
Kempermann, G., Kuhn, H. G. & Gage, F. H. More hippocampal neurons in adult mice living in an enriched environment. Nature 386, 493–495 (1997).
Komitova, M., Mattsson, B., Johansson, B. B. & Eriksson, P. S. Enriched environment increases neural stem/progenitor cell proliferation and neurogenesis in the subventricular zone of stroke-lesioned adult rats. Stroke 36, 1278–1282 (2005). Shows that altering the living environment of the mammal has a neurogenic effect on its neurological outcome after a middle-cerebral-artery-occlusion-induced stroke.
Nilsson, M., Perfilieva, E., Johansson, U., Orwar, O. & Eriksson, P. S. Enriched environment increases neurogenesis in the adult rat dentate gyrus and improves spatial memory. J. Neurobiol. 39, 569–578 (1999).
Lazarov, O. et al. Environmental enrichment reduces Aβ levels and amyloid deposition in transgenic mice. Cell 120, 701–713 (2005).
Craig, C. G. et al. In vivo growth factor expansion of endogenous subependymal neural precursor cell populations in the adult mouse brain. J. Neurosci. 16, 2649–2658 (1996).
Fallon, J. H. et al. Epidermal growth factor immunoreactive material in the central nervous system: location and development. Science 224, 1107–1109 (1984).
Kuhn, H. G., Winkler, J., Kempermann, G., Thal, L. J. & Gage, F. H. Epidermal growth factor and fibroblast growth factor-2 have different effects on neural progenitors in the adult rat brain. J. Neurosci. 17, 5820–5829 (1997).
Reynolds, B. A., Tetzlaff, W. & Weiss, S. A multipotent EGF-responsive striatal embryonic progenitor cell produces neurons and astrocytes. J. Neurosci. 12, 4565–4574 (1992).
Vescovi, A. L., Reynolds, B. A., Fraser, D. D. & Weiss, S. bFGF regulates the proliferative fate of unipotent (neuronal) and bipotent (neuronal/astroglial) EGF-generated CNS progenitor cells. Neuron 11, 951–966 (1993).
Zigova, T., Pencea, V., Wiegand, S. J. & Luskin, M. B. Intraventricular administration of BDNF increases the number of newly generated neurons in the adult olfactory bulb. Mol. Cell. Neurosci. 11, 234–245 (1998).
Benraiss, A., Chmielnicki, E., Lerner, K., Roh, D. & Goldman, S. A. Adenoviral brain-derived neurotrophic factor induces both neostriatal and olfactory neuronal recruitment from endogenous progenitor cells in the adult forebrain. J. Neurosci. 21, 6718–6731 (2001). Shows how a viral vector method of mitogen delivery can cause recruitment of progenitor cells to an injury site in the brain. BDNF affected multiple brain regions when it was administered by an adenovirus.
Duman, R. S., Nakagawa, S. & Malberg, J. Regulation of adult neurogenesis by antidepressant treatment. Neuropsychopharmacology 25, 836–844 (2001).
Malberg, J. E., Eisch, A. J., Nestler, E. J. & Duman, R. S. Chronic antidepressant treatment increases neurogenesis in adult rat hippocampus. J. Neurosci. 20, 9104–9110 (2000).
Chen, G., Rajkowska, G., Du, F., Seraji-Bozorgzad, N. & Manji, H. K. Enhancement of hippocampal neurogenesis by lithium. J. Neurochem. 75, 1729–1734 (2000).
Senatorov, V. V., Ren, M., Kanai, H., Wei, H. & Chuang, D. M. Short-term lithium treatment promotes neuronal survival and proliferation in rat striatum infused with quinolinic acid, an excitotoxic model of Huntington's disease. Mol. Psychiatry 9, 371–385 (2004).
Manji, H. K., Moore, G. J. & Chen, G. Clinical and preclinical evidence for the neurotrophic effects of mood stabilizers: implications for the pathophysiology and treatment of manic-depressive illness. Biol. Psychol. 48, 740–754 (2000). Shows how antidepressant drugs are involved in essential progenitor cell proliferation, maturation and neurite outgrowth.
Manji, H. K., Moore, G. J. & Chen, G. Lithium at 50: have the neuroprotective effects of this unique cation been overlooked? Biol. Psychol. 46, 929–940 (1999).
Malberg, J. E. & Duman, R. S. Cell proliferation in adult hippocampus is decreased by inescapable stress: reversal by fluoxetine treatment. Neuropsychopharmacology 28, 1562–1571 (2003).
Clarke, D. L. et al. Generalized potential of adult neural stem cells. Science 288, 1660–1663 (2000).
Flax, J. D. et al. Engraftable human neural stem cells respond to developmental cues, replace neurons, and express foreign genes. Nature Biotechnol. 16, 1033–1039 (1998).
Herrera, D. G., García-Verdugo, J. M. & Alvarez-Buylla, A. Adult-derived neural precursors transplanted into multiple regions in the adult brain. Ann. Neurol. 46, 867–877 (1999).
Vazey, E. M., Chen, K., Hughes, S. M. & Connor, B. Transplanted adult neural progenitor cells survive, differentiate and reduce motor function impairment in a rodent model of Huntington's disease. Exp. Neurol. 199, 384–396 (2006).
Nakao, N., Ogura, M., Nakai, K. & Itakura, T. Embryonic striatal grafts restore neuronal activity of the globus pallidus in a rodent model of Huntingtons disease. Neuroscience 88, 469–477 (1999).
Visnyei, K. et al. Neural progenitor implantation restores metabolic deficits in the brain following striatal quinolinic acid lesion. Exp. Neurol. 197, 465–474 (2006).
Shim, J. W. et al. Generation of functional dopamine neurons from neural precursor cells isolated from the subventricular zone and white matter of the adult rat brain using Nurr1 overexpression. Stem Cells 25, 1252–1262 (2007).
Dunnett, S. B. & Rosser, A. E. Stem cell transplantation for Huntington's disease. Exp. Neurol. 203, 279–292 (2007).
Bjorklund, A., Dunnett, S. B., Stenevi, U., Lewis, M. E. & Iversen, S. D. Reinnervation of the denervated striatum by substantia nigra transplants: functional consequences as revealed by pharmacological and sensorimotor testing. Brain Res. 199, 307–333 (1980).
Faull, R. L. M., Waldvogel, H. J., Nicholson, L. F. B., Williams, M. N. & Dragunow, M. in Neurotransmitters in the Human Brain (ed. Tracey, D. J.) 173–197 (Plenum, New York, 1995).
Freed, C. R. et al. Transplantation of embryonic dopamine neurons for severe Parkinson's disease. N. Engl. J. Med. 344, 710–719 (2001).
Kopyov, O. V., Jacques, D. S., Lieberman, A., Duma, C. M. & Rogers, R. L. Outcome following intrastriatal fetal mesencephalic grafts for Parkinson's patients is directly related to the volume of grafted tissue. Exp. Neurol. 146, 536–545 (1997).
Kopyov, O. V., Jacques, S., Lieberman, A., Duma, C. M. & Eagle, K. S. Safety of intrastriatal neurotransplantation for Huntington's disease patients. Exp. Neurol. 149, 97–108 (1998).
Bylund, M., Andersson, E., Novitch, B. G. & Muhr, J. Vertebrate neurogenesis is counteracted by Sox1–3 activity. Nature Neurosci. 6, 1162–1168 (2003).
Graham, V., Khudyakov, J., Ellis, P. & Pevny, L. SOX2 functions to maintain neural progenitor identity. Neuron 39, 749–765 (2003).
Baer, K., Eriksson, P. S., Faull, R. L., Rees, M. I. & Curtis, M. A. Sox-2 is expressed by glial and progenitor cells and Pax-6 is expressed by neuroblasts in the human subventricular zone. Exp. Neurol. 204, 828–831 (2007).
Hack, M. A. et al. Neuronal fate determinants of adult olfactory bulb neurogenesis. Nature Neurosci. 8, 865–872 (2005).
Kohwi, M., Osumi, N., Rubenstein, J. L. & Alvarez-Buylla, A. Pax6 is required for making specific subpopulations of granule and periglomerular neurons in the olfactory bulb. J. Neurosci. 25, 6997–7003 (2005).
Reimold, A. M. et al. Chondrodysplasia and neurological abnormalities in ATF-2-deficient mice. Nature 379, 262–265 (1996).
Pearson, J., Curtis, M. A., Waldvogel, H. J., Faull, R. L. M. & Dragunow, M. Activating transcription factor 2 expression in the adult human brain: association with both neurodegeneration and neurogenesis. Neuroscience 133, 437–451 (2005).
Matsuoka, N. et al. Adenovirus-mediated gene transfer of fibroblast growth factor-2 increases BrdU-positive cells after forebrain ischemia in gerbils. Stroke 34, 1519–1525 (2003).
Vicario, I. & Schimmang, T. Transfer of FGF-2 via HSV-1-based amplicon vectors promotes efficient formation of neurons from embryonic stem cells. J. Neurosci. Methods 123, 55–60 (2003).
Dolcetta, D. et al. Design and optimization of lentiviral vectors for transfer of GALC expression in Twitcher brain. J. Gene Med. 8, 962–971 (2006).
Geraerts, M. et al. Lentiviral vectors mediate efficient and stable gene transfer in adult neural stem cells in vivo. Hum. Gene Ther. 17, 635–650 (2006).
Sugiura, S. et al. Adenovirus-mediated gene transfer of heparin-binding epidermal growth factor-like growth factor enhances neurogenesis and angiogenesis after focal cerebral ischemia in rats. Stroke 36, 859–864 (2005).
Tabata, H. & Nakajima, K. Multipolar migration: the third mode of radial neuronal migration in the developing cerebral cortex. J. Neurosci. 23, 9996–10001 (2003).
Nakatomi, H. et al. Regeneration of hippocampal pyramidal neurons after ischemic brain injury by recruitment of endogenous neural progenitors. Cell 110, 429–441 (2002).
Luskin, M. B. & Boone, M. S. Rate and pattern of migration of lineally-related olfactory bulb interneurons generated postnatally in the subventricular zone of the rat. Chem. Senses 19, 695–714 (1994).
Lois, C. & Alvarez-Buylla, A. Long-distance neuronal migration in the adult mammalian brain. Science 264, 1145–1148 (1994).
Curtis, M. A. et al. Human neuroblasts migrate to the olfactory bulb via a lateral ventricular extension. Science 315, 1243–1249 (2007). Demonstrates the ongoing usefulness of the SVZ in the normal brain for producing new neurons for cell replacement in the olfactory bulb. Details the only long-distance progenitor migratory system in the adult human brain.
Jankovski, A., Garcia, C., Soriano, E. & Sotelo, C. Proliferation, migration and differentiation of neuronal progenitor cells in the adult mouse subventricular zone surgically separated from its olfactory bulb. Eur. J. Neurosci. 10, 3853–3868 (1998).
Kirschenbaum, B., Doetsch, F., Lois, C. & Alvarez-Buylla, A. Adult subventricular zone neuronal precursors continue to proliferate and migrate in the absence of the olfactory bulb. J. Neurosci. 19, 2171–2180 (1999).
Wiltrout, C., Lang, B., Yan, Y., Dempsey, R. J. & Vemuganti, R. Repairing brain after stroke: a review on post-ischemic neurogenesis. Neurochem. Int. 50, 1028–1041 (2007).
Leker, R. R. et al. Long-lasting regeneration after ischemia in the cerebral cortex. Stroke 38, 153–161 (2007).
Minger, S. L. et al. Endogenous neurogenesis in the human brain following cerebral infarction. Regen. Med. 2, 69–74 (2007).
Dempsey, R. J. & Kalluri, H. S. Ischemia-induced neurogenesis: role of growth factors. Neurosurg. Clin. N. Am. 18, 183–190, xi (2007).
Schabitz, W. R. et al. Intravenous brain-derived neurotrophic factor enhances poststroke sensorimotor recovery and stimulates neurogenesis. Stroke 38, 2165–2172 (2007).
Schneider, A. et al. An extended window of opportunity for G-CSF treatment in cerebral ischemia. BMC Biol. 4, 36 (2006).
Altman, J. & Das, G. D. Autoradiographic and histological evidence of postnatal hippocampal neurogenesis in rats. J. Comp. Neurol. 124, 319–335 (1965). One of the earliest studies on adult neurogenesis; important technique advances are displayed in which tritiated thymidine, together with histological techniques, reveal adult neurogenesis in the adult rodent brain.
Aimone, J. B., Wiles, J. & Gage, F. H. Potential role for adult neurogenesis in the encoding of time in new memories. Nature Neurosci. 9, 723–727 (2006).
Black, A. H., Nadel, L. & O'Keefe, J. Hippocampal function in avoidance learning and punishment. Psychol. Bull. 84, 1107–1129 (1977).
Eichenbaum, H. Hippocampus: mapping or memory? Curr. Biol. 10, R785–R787 (2000).
Kim, J. J. & Diamond, D. M. The stressed hippocampus, synaptic plasticity and lost memories. Nature Rev. Neurosci. 3, 453–462 (2002).
van Praag, H. et al. Functional neurogenesis in the adult hippocampus. Nature. 415, 1030–1034 (2002).
Houser, C. R. Granule cell dispersion in the dentate gyrus of humans with temporal lobe epilepsy. Brain Res. 535, 195–204 (1990).
Luskin, M. B., Zigova, T., Soteres, B. J. & Stewart, R. R. Neuronal progenitor cells derived from the anterior subventricular zone of the neonatal rat forebrain continue to proliferate in vitro and express a neuronal phenotype. Mol. Cell. Neurosci. 8, 351–366 (1997). Demonstration that the SVZ progenitors can be removed and encouraged to adopt a neuronal phenotype in culture.
Parent, J. M., Elliott, R. C., Pleasure, S. J., Barbaro, N. M. & Lowenstein, D. H. Aberrant seizure-induced neurogenesis in experimental temporal lobe epilepsy. Ann. Neurol. 59, 81–91 (2006).
Parent, J. M., Janumpalli, S., McNamara, J. O. & Lowenstein, D. H. Increased dentate granule cell neurogenesis following amygdala kindling in the adult rat. Neurosci. Lett. 247, 9–12 (1998).
Parent, J. M., von dem Bussche, N. & Lowenstein, D. H. Prolonged seizures recruit caudal subventricular zone glial progenitors into the injured hippocampus. Hippocampus 16, 321–328 (2006).
Raedt, R. et al. Radiation of the rat brain suppresses seizure-induced neurogenesis and transiently enhances excitability during kindling acquisition. Epilepsia 6 Jun 2007 (doi: 10.1111/j.1528–1167.2007.01146.x).
Dashtipour, K., Tran, P. H., Okazaki, M. M., Nadler, J. V. & Ribak, C. E. Ultrastructural features and synaptic connections of hilar ectopic granule cells in the rat dentate gyrus are different from those of granule cells in the granule cell layer. Brain Res. 890, 261–271 (2001).
Parent, J. M. et al. Dentate granule cell neurogenesis is increased by seizures and contributes to aberrant network reorganization in the adult rat hippocampus. J. Neurosci. 17, 3727–3738 (1997).
Scharfman, H. E., Sollas, A. L. & Goodman, J. H. Spontaneous recurrent seizures after pilocarpine-induced status epilepticus activate calbindin-immunoreactive hilar cells of the rat dentate gyrus. Neuroscience 111, 71–81 (2002).
Bonde, S., Ekdahl, C. T. & Lindvall, O. Long-term neuronal replacement in adult rat hippocampus after status epilepticus despite chronic inflammation. Eur. J. Neurosci. 23, 965–974 (2006).
Mangiarini, L. et al. Exon 1 of the HD gene with an expanded CAG repeat is sufficient to cause a progressive neurological phenotype in transgenic mice. Cell 87, 493–506 (1996). Demonstration of the effect of inserting the human exon 1 of the huntingtin gene into rodent brains.
Davies, S. W. et al. Formation of neuronal intranuclear inclusions underlies the neurological dysfunction in mice transgenic for the HD mutation. Cell 90, 537–548 (1997).
Beal, M. F. et al. Replication of the neurochemical characteristics of Huntington's disease by quinolinic acid. Nature 321, 168–171 (1986).
Beal, M. F., Kowall, N. W., Swartz, K. J., Ferrante, R. J. & Martin, J. B. Differential sparing of somatostatin-neuropeptide Y and cholinergic neurons following striatal excitotoxin lesions. Synapse 3, 38–47 (1989).
Ferrante, R. J., Kowall, N. W., Cipolloni, P. B., Storey, E. & Beal, M. F. Excitotoxin lesions in primates as a model for Huntington's disease: histopathologic and neurochemical characterisation. Exp. Neurol. 119, 46–71 (1993).
Waldvogel, H. J., Faull, R. L. M., Williams, M. N. & Dragunow, M. Differential sensitivity of calbindin and parvalbumin immunoreactive cells in the striatum to excitotoxins. Brain Res. 546, 329–335 (1991).
Collin, T., Arvidsson, A., Kokaia, Z. & Lindvall, O. Quantitative analysis of the generation of different striatal neuronal subtypes in the adult brain following excitotoxic injury. Exp. Neurol. 195, 71–80 (2005).
Parent, J. M. Injury-induced neurogenesis in the adult mammalian brain. Neuroscientist 9, 261–272 (2003). Review of stroke and epilepsy studies on proliferation in the germinal zones in rodent brains.
Acknowledgements
We wish to thank the Swedish Research Council, the Health Research Council of New Zealand and the Neurological Foundation of New Zealand. In particular, M.A.C. is supported as a Wrightson Postdoctoral Fellow. We thank J. Westin for his expertise in making the original illustrations.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Glossary
- Ganglionic eminence
-
An embryonic cluster of nerve cells near the ventricle, where neurons are generated before they migrate to different parts of the brain.
- Progenitor cell
-
A dividing cell that has the capacity to differentiate.
- Basal ganglion
-
A type of specialized nerve cell cluster that is found deep within each cerebral hemisphere and the upper brainstem. Basal ganglia consist of the caudate nucleus, the putamen, the globus pallidus, the sub-thalamic nucleus and the substantia nigra. The basal ganglia assist in initiating movement and in motor control.
- Neuroblast
-
A dividing neuronal precursor cell.
- Striatum
-
The area of the brain that controls movement and balance. It consists of the caudate nucleus and the putamen, and it is connected to and receives signals from the substantia nigra and the cortex.
- Rota-rod performance
-
A motor test that determines the ability of rodents to keep their balance on a rotating cylinder.
- Neurosphere culture
-
A free-floating multidimensional cell structure that is derived from neural stem cells in vitro. Neurosphere assays enable the investigation of the differentiation and proliferative potential of neural stem cells and progenitor cells.
- Differentiation
-
The process whereby an unspecialized cell develops the specialized functions of a mature cell, for example, a neuron, or a liver or muscle cell.
- Acute treatment
-
A remedial action that is administered within the first few hours of a neurodegenerative or necrotic event such as a stroke.
- Sub-acute treatment
-
A remedial action that is administered after the initial neurodegenerative and/or necrotic events of a stroke are over.
- Proliferating cell nuclear antigen
-
(PCNA). A cell-cycle protein that is expressed during, and shortly after, G1 and S-phase. It is used as a marker of recent cell division.
- Substantia nigra pars compacta
-
(SNpc). The area of the brain where dopamine is produced. It is also one of the basal ganglia nuclei.
- Levodopa
-
The most commonly used drug for the treatment of Parkinson's disease. After administration, it is converted into dopamine, such that it raises the levels of dopamine in the brain. Levodopa treatment is an effective control of Parkinson's disease symptoms, however, its long-term use is associated with complications.
- Transit-amplifying cell
-
A type of proliferative stem cell that is able to divide only 3–5 times before all of its daughter cells terminally differentiate.
- β-amyloid (Aβ) peptide
-
A peptide that is derived from amyloid precursor protein. These peptides are the main protein component of amyloid plaques.
- Amyloid precursor protein
-
(APP). A membrane glycoprotein component of fast axonal transport that is cleaved by secretases to give rise to Aβ.
- Neural stem cell
-
A stem cell that is found in adult neural tissue and can give rise to neurons and glial cells.
- Radial glial cell
-
A connective tissue cell that guides the migration of neurons from the SVZ to the cortex. Radial glia are also a major source of cortical neurons during development.
- Autologous transplant
-
A tissue transplant in which a patient's own progenitor cells are removed, expanded in culture and then transplanted back into the disease-affected region.
Rights and permissions
About this article
Cite this article
Curtis, M., Faull, R. & Eriksson, P. The effect of neurodegenerative diseases on the subventricular zone. Nat Rev Neurosci 8, 712–723 (2007). https://doi.org/10.1038/nrn2216
Issue Date:
DOI: https://doi.org/10.1038/nrn2216
This article is cited by
-
α-synuclein inclusions are abundant in non-neuronal cells in the anterior olfactory nucleus of the Parkinson’s disease olfactory bulb
Scientific Reports (2020)
-
Intranasal insulin administration may be highly effective in improving cognitive function in mice with cognitive dysfunction by reversing brain insulin resistance
Cognitive Neurodynamics (2020)
-
PRDX6 Inhibits Neurogenesis through Downregulation of WDFY1-Mediated TLR4 Signal
Molecular Neurobiology (2019)
-
Layer-specific lipid signatures in the human subventricular zone demonstrated by imaging mass spectrometry
Scientific Reports (2018)
-
Drug-induced Parkinson’s disease modulates protein kinase A and Olfactory Marker Protein in the mouse olfactory bulb
Behavioral and Brain Functions (2017)