Key Points
-
Intelligence research is more advanced and less controversial than is generally realized. Definitive conclusions about the neural and genetic bases of intelligence are being drawn — these have ethical implications that need to be addressed.
-
General intelligence and volume of frontal grey matter (assessed by magnetic resonance imaging) are correlated.
-
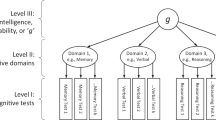
The lateral prefrontal cortex is consistently activated during intelligence testing. Frontal and parietal brain regions implicated in working memory are also activated under test conditions. These data contribute to the debate on whether intelligence has a unitary (activation of a single brain region/functional unit) or multiple basis.
-
The structure of brain regions that influence intelligence is under strong genetic control. Studies of intelligence using twins reared separately support the heritability of cognitive function.
-
Environmental factors — such as prenatal exposure to toxins, duration of breastfeeding and shared family environment — affect intellectual function. These non-genetic factors have a much greater effect on childhood intelligence in impoverished families.
-
Establishing a neurobiological basis for intelligence has important ethical implications. For example, is it ethical to assess racial differences in intelligence? Such questions need to be proactively addressed so that the field of intelligence research is not perceived as socially divisive.
Abstract
Human mental abilities, such as intelligence, are complex and profoundly important, both in a practical sense and for what they imply about the human condition. Understanding these abilities in mechanistic terms has the potential to facilitate their enhancement. There is strong evidence that the lateral prefrontal cortex, and possibly other areas, support intelligent behaviour. Variations in intelligence and brain structure are heritable, but are also influenced by factors such as education, family environment and environmental hazards. Cognitive, psychometric, genetic and neuroimaging studies are converging, and the emergence of mechanistic models of intelligence is inevitable. These exciting scientific advances encourage renewed responsiveness to the social and ethical implications of conducting such research.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Ross, L. The intuitive psychologist and his shortcomings: distortions in the attribution process. Adv. Exp. Soc. Psychol. 10, 173–220 (1977).
Aronson, J. & Steele, C. M. in The Handbook of Competence and Motivation (eds Elliot, A. & Dweck, C.) (Wiley, New York, in the press). Reviews the large body of literature on stereotype threat, illustrating how subtle situational factors can impact performance on tests of mental ability. The implications for standardized testing have been largely ignored by the testing industry.
Pinker, S. The Blank Slate (Viking, New York, 2002). A strong critique of the standard social science position that individual differences result only from differing environment (the 'blank slate' view of human nature). It emphasizes that such a view is not merely incorrect but also dangerous.
Kagan, J. Three Seductive Ideas (Harvard Univ. Press, Cambridge, Massachusetts, 1998).
Hunt, M. The New Know-nothings: The Political Foes of the Scientific Study of Human Nature (Transaction, New Brunswick, 1999).
Goode, E. Brain scans reflect problem-solving skill. The New York Times 17 Feb (2003).
Ordover, N. American Eugenics: Race, Queer Anatomy, and the Science of Nationalism (Univ. Minnesota Press, Minneapolis, 2003). A disturbing and instructive history of oversimplified genetic science misapplied to social policy in the United States.
Hume, D. A Treatise of Human Nature (Clarendon, Oxford, 1739/1964).
Loehlin, J. C. in Handbook of Intelligence (ed. Sternberg, R. J.) 176–193 (Cambridge Univ. Press, New York, 2000).
Gould, S. J. The Mismeasure of Man (Norton, New York, 1981/1996).
Cattell, R. B. Abilities: Their Structure, Growth, and Action (Houghton Mifflin, Boston, 1971).
Sternberg, R. J. Beyond IQ: A Triarchic Theory of Human Intelligence (Cambridge Univ. Press, Cambridge, UK, 1985). An empirically grounded argument against a unitary view of intelligence.
Jensen, A. R. Bias in Mental Testing (Free, New York, 1980).
Carroll, J. Human Cognitive Abilities: A Survey of Factor–Analytic Studies (Cambridge Univ. Press, Cambridge, UK, 1993).
Neisser, U. et al. Intelligence: knowns and unknowns. Am. Psychol. 51, 77–101 (1996). A consensus of experts on the state of intelligence research, outlining many points of agreement despite some contentious issues.
Lubinski, D. Introduction to the special section on cognitive abilities: 100 years after Spearman's (1904) 'general intelligence', objectively determined and measured. J. Pers. Soc. Psychol. 86, 96–111 (2004).
Deary, I. J. Human intelligence differences: a recent history. Trends Cogn. Sci. 5, 127–130 (2001).
Kane, M. J. & Engle, R. W. The role of prefrontal cortex in working-memory capacity, executive attention and general fluid intelligence: an individual-differences perspective. Psychon. Bull. Rev. 9, 637–671 (2002). An exhaustive review of the behavioural literature on intelligence and individual differences in working memory capacity, especially as related to the prefrontal cortex.
Vernon, P. A. et al. in Handbook of Intelligence (ed. Sternberg, R. J.) 245–264 (Cambridge Univ. Press, New York, 2000).
Deary, I. J. Looking Down on Human Intelligence (Oxford Univ. Press, New York, 2000).
Finger, S. Origins of Neuroscience (Oxford Univ. Press, New York, 1994).
Piercy, M. The effects of cerebral lesions on intellectual function: a review of current research trends. Brit. J. Psychiatry 110, 310–352 (1964).
Kolb, I. & Whishaw, B. Fundamentals of Human Neuropsychology (W. H. Freeman, New York, 1996).
Duncan, J., Emslie, H., Williams, P., Johnson, R. & Freer, C. Intelligence and the frontal lobe: the organization of goal-directed behavior. Cogn. Psychol. 30, 257–303 (1996).
Duncan, J., Burgess, P. & Emslie, H. Fluid intelligence after frontal lobe lesions. Neuropsychologia 33, 261–268 (1995).
Waltz, J. A. et al. A system for relational reasoning in human prefrontal cortex. Psychol. Sci. 10, 119–125 (1999).
Gray, J. R., Chabris, C. F. & Braver, T. S. Neural mechanisms of general fluid intelligence. Nature Neurosci. 6, 316–322 (2003). The first large-sample event-related fMRI study of intelligence, reporting correlations between Gf and activity in lateral prefrontal cortex and parietal cortex. It is notable for its individual-differences approach and emphasis on theoretically motivated trials.
Andreasen, N. C. et al. Intelligence and brain structure in normal individuals. Am. J. Psychiatry 150, 130–134 (1993).
Peters, M. Does brain size matter? A reply to Rushton and Ankney. Can. J. Exp. Psychol. 49 (1995).
McDaniel, M. A. & Nguyen, N. T. A meta-analysis of the relationship between MRI-assessed brain volume and intelligence. Proc. Int. Soc. Intell. Res. (2002).
Pennington, B. F. et al. A twin MRI study of size variations in human brain. J. Cogn. Neurosci. 12, 223–232 (2000).
Thompson, P. M. et al. Genetic influences on brain structure. Nature Neurosci. 4, 1253–1258 (2001). This twin study presents the first maps of genetic influences on human brain structure, revealing that the quantity of frontal grey matter is under strong genetic control and is also linked with intelligence.
Plomin, R. & Kosslyn, S. M. Genes, brain and cognition. Nature Neurosci. 4, 1153–1154 (2001).
Posthuma, D. et al. The association between brain volume and intelligence is of genetic origin. Nature Neurosci. 5, 83–84 (2002).
Draganski, B. et al. Changes in grey matter induced by training. Nature 247, 311–312 (2004).
Duncan, J. et al. A neural basis for general intelligence. Science 289, 457–460 (2000). A PET study of high- g versus low- g tasks. It is notable for using brain imaging data to inform the debate about the structure of intelligence.
Prabhakaran, V., Smith, J. A. L., Desmond, J. E., Glover, G. H. & Gabrieli, J. D. E. Neuronal substrates of fluid reasoning: an fMRI study of neocortical activation during performance of the Raven's Progressive Matrices Test. Cogn. Psychol. 33, 43–63 (1997).
Prabhakaran, V., Rypma, B. & Gabrieli, J. D. E. Neural substrates of mathematical reasoning: a functional magnetic resonance imaging study of neocortical activation during performance of the Necessary Arithmetic Operations Test. Neuropsychology 15, 115–127 (2001).
Esposito, G., Kirby, B. S., Van Horn, J. D., Ellmore, T. M. & Berman, K. F. Context-dependent, neural system-specific neurophysiological concomitants of ageing: mapping PET correlates during cognitive activation. Brain 122, 963–979 (1999).
Jensen, A. R. The g Factor: The Science of Mental Ability (Praeger, Westport, 1998). The modern argument for a unitary view of intelligence.
Smith, E. E. & Jonides, J. Storage and executive processes in the frontal lobes. Science 283, 1657–1661 (1999).
Cabeza, R. & Nyberg, L. Imaging cognition II: an empirical review of 275 PET and fMRI studies. J. Cogn. Neurosci. 12, 1–47 (2000).
Ruff, C. C., Knauff, M., Fangmeier, T. & Spreer, J. Reasoning and working memory: common and distinct neuronal processes. Neuropsychologia 41, 1241–1253 (2003).
Carpenter, P. A., Just, M. A. & Shell, P. What one intelligence test measures: a theoretical account of the processing in the Raven Progressive Matrices Test. Psychol. Rev. 97, 404–431 (1990).
Kyllonen, P. C. & Christal, R. E. Reasoning ability is (little more than) working memory capacity?! Intelligence 14, 389–433 (1990).
Conway, A. R. A., Kane, M. J. & Engle, R. W. Working memory capacity and its relation to general intelligence. Trends Cogn. Sci. 7, 547–552 (2003).
Kroger, J. K. et al. Recruitment of anterior dorsolateral prefrontal cortex in human reasoning: a parametric study of relational complexity. Cereb. Cortex 12, 477–485 (2002).
Goel, V. & Dolan, R. J. Functional neuroanatomy of three-term relational reasoning. Neuropsychologia 39, 901–909 (2001).
Christoff, K. et al. Rostrolateral prefrontal cortex involvement in relational integration during reasoning. Neuroimage 14, 1136–1149 (2001).
Braver, T. S. & Bongiolatti, S. R. The role of the frontopolar prefrontal cortex in subgoal processing during working memory. Neuroimage 15, 523–536 (2002).
Koechlin, E., Basso, G., Pietrini, P., Panzer, S. & Grafman, J. The role of the anterior prefrontal cortex in human cognition. Nature 399, 148–151 (1999).
Deary, I. J. Human intelligence differences: toward a combined experimental-differential approach. Trends Cogn. Sci. 5, 164–170 (2001).
Kosslyn, S. M. et al. Bridging psychology and biology: the analysis of individuals in groups. Am. Psychol. 57, 341–351 (2002).
Haier, R. J. et al. Cortical glucose metabolic-rate correlates of abstract reasoning and attention studied with positron emission tomography. Intelligence 12, 199–217 (1988).
Haier, R. J. et al. Regional glucose metabolic changes after learning a complex visuospatial/motor task: a positron emission tomography study. Brain Res. 570, 134–143 (1992).
Neubauer, A. C., Fink, A. & Schrausser, D. G. Intelligence and neural efficiency: the influence of task content and sex on the brain–IQ relationship. Intelligence 30, 515–536 (2002).
Haier, R. J., White, N. S. & Alkire, M. T. Individual differences in general intelligence correlate with brain function during nonreasoning tasks. Intelligence 31, 429–441 (2003).
Kane, M. J. & Engle, R. W. Working-memory capacity and the control of attention: the contributions of goal neglect, response competition, and task set to Stroop interference. J. Exp. Psychol. Gen. 132, 47–70 (2003).
Engle, R. W., Tuholski, S. W., Laughlin, J. E. & Conway, A. R. A. Working memory, short-term memory, and general fluid intelligence: a latent-variable approach. J. Exp. Psychol. Gen. 128, 309–331 (1999).
Deary, I. J. et al. The functional anatomy of inspection time: a pilot fMRI study. Intelligence 29, 497–510 (2001).
Stough, C., Thompson, J. C., Bates, T. C. & Nathan, P. J. Examining neurochemical determinants of inspection time: development of a biological model. Intelligence 29, 511–522 (2001).
Furey, M. L., Pietrini, P. & Haxby, J. V. Cholinergic enhancement and increased selectivity of perceptual processing during working memory. Science 290, 2315–2319 (2000).
Scarmeas, N. et al. Cognitive reserve modulates functional brain responses during memory tasks: a PET study in healthy young and elderly subjects. Neuroimage 19, 1215–1227 (2003).
Stern, Y. et al. Exploring the neural basis of cognitive reserve. J. Clin. Exp. Neuropsychol. 25, 691–701 (2003).
Seidman, L. J. et al. A functional magnetic resonance imaging study of auditory vigilance with low and high information processing demands. Neuropsychology 12, 505–518 (1998).
Miller, E. K. & Cohen, J. D. An integrative theory of prefrontal cortex function. Ann. Rev. Neurosci. 21, 167–202 (2001).
Jung, R. E. et al. Biochemical markers of intelligence: a proton MR spectroscopy study of normal human brain. Proc. R. Soc. Lond. B 266, 1375–1379 (1999).
Rae, C. et al. Brain bioenergetics and cognitive ability. Dev. Neurosci. 25, 324–331 (2003).
Anokhin, A. P., Lutzenberger, W. & Birbaumer, N. Spatiotemporal organization of brain dynamics and intelligence: an EEG study in adolescents. Int. J. Psychophysiol. 33, 259–273 (1999).
Gevins, A. & Smith, M. E. Neurophysiological measures of working memory and individual differences in cognitive ability and cognitive style. Cereb. Cortex 10, 829–839 (2000).
Haier, R. J., Siegel, B., Tang, C., Abel, L. & Buchsbaum, M. S. Intelligence and changes in regional cerebral glucose metabolic-rate following learning. Intelligence 16, 415–426 (1992).
Garlick, D. Understanding the nature of the general factor of intelligence: the role of individual differences in neural plasticity as an explanatory mechanism. Psychol. Rev. 109, 116–136 (2002).
Salovey, P., Mayer, J. D. & Caruso, D. in The Handbook of Positive Psychology (eds Snyder, C. R. & Lopez, S. J.) 159–171 (Oxford Univ. Press, New York, 2002).
Bar-On, R., Tranel, D., Denburg, N. L. & Bechara, A. Exploring the neurological substrate of emotional and social intelligence. Brain 126, 1790–1800 (2003).
Bouchard, T. J. Jr & McGue, M. Familial studies of intelligence: a review. Science 212, 1055–1059 (1981).
Grigorenko, E. L. in Handbook of Intelligence (ed. Sternberg, R. J.) 53–91 (Cambridge Univ. Press, New York, 2000).
Plomin, R. & Loehlin, J. C. Direct and indirect IQ heritability estimates: a puzzle. Behav. Genet. 19, 331–342 (1989).
Neale, M. C. & Cardon, L. R. Methodology for Genetic Studies of Twins and Families (Kluwer, Boston, 1992).
Scarr, S. & Weinberg, R. A. Educational and occupational achievements of brothers and sisters in adoptive and biologically related families. Behav. Genet. 24, 301–325 (1994).
Posthuma, D. & Boomsma, D. I. A note on the statistical power in extended twin designs. Behav. Genet. 30, 147–158 (2000).
Vogel, F. & Motulsky, A. G. Human Genetics (Springer, New York, 1997).
Lewontin, R., Rose, S. & Kamin, L. J. Not in Our Genes: Biology, Ideology and Human Nature (Random House, New York, 1984).
Bouchard, T. J. Jr, Lykken, D. T., McGue, M., Segal, N. L. & Tellegen, A. Sources of human psychological differences: the Minnesota study of twins reared apart. Science 250, 223–228 (1990).
Dorfman, D. D. Soft science with a neoconservative agenda (review of Richard J. Herrnstein and Charles Murray The Bell Curve: Intelligence and Class Structure in American Life). Contemp. Psychol. 40 (1995).
Spearman, C. The Nature of 'Intelligence' and the Principles of Cognition (Macmillan, London, 1927).
Kamin, L. J. Twin studies, heritability, and intelligence. Science 278, 1385 (1997).
McClearn, G. E. et al. Substantial genetic influence on cognitive abilities in twins 80 or more years old. Science 276, 1560–1563 (1997).
Feldman, M. W. & Otto, S. P. Twin studies, heritability, and intelligence. Science 278, 1383–1384 (1997).
Devlin, B., Daniels, M. & Roeder, K. The heritability of IQ. Nature 388, 468–471 (1997). A meta-analysis of IQ studies, showing that the prenatal environment might be more influential, and genetic factors less influential, in determining childhood IQ than was previously appreciated.
Finkel, D., Pedersen, N. L., Plomin, R. & McClearn, G. E. Longitudinal and cross-sectional twin data on cognitive abilities in adulthood: the Swedish Adoption/Twin Study of Aging. Dev. Psychol. 34, 1400–1413 (1998).
Loehlin, J. C. Partitioning environmental and genetic contributions to behavioral development. Am. Psychol. 44, 1285–1292 (1989).
Rowe, D. C. & Jacobson, K. C. in Behavioral Genetics (eds Carson, R. A. & Rothstein, M. A.) 12–34 (Johns Hopkins Univ. Press, Baltimore, 1999).
Boomsma, D. I., de Geus, E. J., van Baal, G. C. & Koopmans, J. R. A religious upbringing reduces the influence of genetic factors on disinhibition: evidence for interaction between genotype and environment on personality. Twin Research 2, 115–125 (1999).
Plomin, R. Genetics of childhood disorders. III. Genetics and intelligence. J. Am. Acad. Child Adolesc. Psychiatry 38, 786–788 (1999).
Dickens, W. T. & Flynn, J. R. Heritability estimates versus large environmental effects: the IQ paradox resolved. Psychol. Rev. 108, 346–369 (2001). A mathematical model that proposes powerful gene–environment interactions to reconcile the paradox that IQ is highly heritable even though average scores have increased significantly in recent decades.
Little, P. Genetic Destinies (Oxford Univ. Press, New York, 2002).
Ceci, S. J. & Williams, W. M. Schooling, intelligence, and income. Am. Psychol. 52, 1051–1058 (1997).
Neisser, U. (ed.) The Rising Curve: Long-term Gains in IQ and Related Measures (American Psychological Association, Washington DC, 1998).
Oddy, W. H. et al. Breast feeding and cognitive development in childhood: a prospective birth cohort study. Paediatr. Perinat. Epidemiol. 17, 81–90 (2003).
Drane, D. L. & Logemann, J. A. A critical evaluation of the evidence on the association between type of infant feeding and cognitive development. Paediatr. Perinat. Epidemiol. 14, 349–356 (2000).
McGue, M., Bouchard, T. J. Jr, Iacono, W. G. & Lykken, D. T. in Nature, Nurture and Psychology (eds Plomin, R. & McClearn, G. E.) 59–76 (American Psychological Association, Washington DC, 1993).
Turkheimer, E., Haley, A., Waldron, M., D'Onofrio, B. & Gottesman, I. I. Socioeconomic status modifies heritability of IQ in young children. Psychol. Sci. 14, 623–628 (2003). The heritability of intelligence depends on socioeconomic status, with low heritability at low socioeconomic status, and higher heritability for high socioeconomic status.
Sherry, S. T. et al. dbSNP: the NCBI database of genetic variation. Nucleic Acids Res. 29, 308–311 (2001).
Chorney, M. J. et al. A quantitative trait locus (QTL) associated with cognitive ability in children. Psychol. Sci. 9, 159–166 (1998).
Hill, L., Chorney, M. J., Lubinski, D., Thompson, L. A. & Plomin, R. A quantitative trait locus not associated with cognitive ability in children: a failure to replicate. Psychol. Sci. 13, 561–562 (2002).
Comings, D. E. et al. Role of the cholinergic muscarinic 2 receptor (CHRM2) gene in cognition. Mol. Psychiatry 8, 10–11 (2002).
Fisher, P. J. et al. DNA pooling identifies QTLs on chromosome 4 for general cognitive ability in children. Hum. Mol. Genet. 8, 915–922 (1999).
Payton, A. et al. Cathepsin D exon 2 polymorphism associated with general intelligence in a healthy older population. Mol. Psychiatry 8, 14–18 (2003).
Plomin, R. et al. A genome-wide scan of 1842 DNA markers for allelic associations with general cognitive ability: a five-stage design using DNA pooling and extreme selected groups. Behav. Genet. 31, 497–509 (2001). Describes a multistage molecular genetic approach to identify individual genetic variations that affect intelligence in healthy, normal individuals.
McGuffin, P., Riley, B. & Plomin, R. Genomics and behavior. Toward behavioral genomics. Science 291, 1232–1249 (2001).
Tamayo, P. et al. Interpreting patterns of gene expression with self-organizing maps: methods and application to hematopoietic differentiation. Proc. Natl Acad. Sci. USA 96, 2907–2912 (1999).
Mazziotta, J. C. et al. A probabilistic atlas and reference system for the human brain. J. R. Soc. 356, 1293–1322 (2001).
Posthuma, D. et al. Multivariate genetic analysis of brain structure in an extended twin design. Behav. Genet. 30, 311–319 (2000).
Pfefferbaum, A., Sullivan, E. V., Swan, G. E. & Carmelli, D. Brain structure in men remains highly heritable in the seventh and eighth decades of life. Neurobiol. Aging 21, 63–74 (2000).
Baare, W. F. et al. Quantitative genetic modeling of variation in human brain morphology. Cereb. Cortex 11, 816–824 (2001).
Geschwind, D. H., Miller, B. L., DeCarli, C. & Carmelli, D. Heritability of lobar brain volumes in twins supports genetic models of cerebral laterality and handedness. Proc. Natl Acad. Sci. USA 99, 3176–3181 (2002).
Wright, I. C., Sham, P., Murray, R. M., Weinberger, D. R. & Bullmore, E. T. Genetic contributions to regional human brain variability estimated in a twin study by path analysis of parcellated grey matter maps. Neuroimage 17, 256–271 (2002).
Carmelli, D., Swan, G. E., DeCarli, C. & Reed, T. Quantitative genetic modeling of regional brain volumes and cognitive performance in older male twins. Biol. Psychol. 61, 139–155 (2002).
Thompson, P. M., Cannon, T. D. & Toga, A. W. Mapping genetic influences on human brain structure. Ann. Med. 24, 523–536 (2002).
Bond, J. et al. ASPM is a major determinant of cerebral cortical size. Nature Genet. 32, 316–320 (2002).
Egan, M. F. et al. The BDNF val66met polymorphism affects activity-dependent secretion of BDNF and human memory and hippocampal function. Cell 112, 257–269 (2003).
Egan, M. F. et al. Effect of COMT Val 108/158 Met genotype on frontal lobe function and risk for schizophenia. Proc. Natl Acad. Sci. USA 98, 6917–6922 (2001).
Diamond, A., Briand, L., Fossella, J. & Gehlbach, L. Genetic and neurochemical modulation of prefrontal cognitive functions in children. Am. J. Psychiatry 161, 125–132 (2004).
Fan, J., Fossella, J., Sommer, T., Wu, Y. & Posner, M. I. Mapping the genetic variation of executive attention onto brain activity. Proc. Natl Acad. Sci. USA 100, 7406–7411 (2003).
Gottfredson, L. S. Why g matters: the complexity of everyday life. Intelligence 24, 79–132 (1997).
Lubinski, D. & Humphreys, L. G. Incorporating general intelligence into epidemiology and the social sciences. Intelligence 24, 159–201 (1997).
Duncan, J. in The Cognitive Neurosciences (ed. Gazzaniga, M.) 721–733 (MIT Press, Cambridge, Massachusetts, 1995).
Wolpert, L. Is science dangerous? Nature 398, 281–282 (1999). An essay on the relationship between science and society, emphasizing a distinction between knowledge and the application of knowledge, and the importance of not censoring research.
Farah, M. J. Emerging ethical issues in neuroscience. Nature Neurosci. 5, 1123–1129 (2002).
Kulynych, J. Legal and ethical issues in neuroimaging research: human subjects protection, medical privacy, and the public communication of research results. Brain Cogn. 50, 345–357 (2002).
Illes, J., Kirschen, M. P. & Gabrieli, J. D. From neuroimaging to neuroethics. Nature Neurosci. 6, 205 (2003).
Gottfredson, L. Egalitarian fiction and collective fraud. Society 31, 53–59 (1994).
Miele, F. Intelligence, Race, and Genetics: Conversations with Arthur R. Jensen (Westview, Boulder, 2002).
Feldman, M. W., Lewontin, R. C. & King, M. C. Race: a genetic melting-pot. Nature 424, 374 (2003).
Loehlin, J. C. Should we do research on race differences in intelligence? Intelligence 16, 1–4 (1992).
Steele, C. M. & Aronson, J. Stereotype threat and the intellectual test performance of African Americans. J. Pers. Soc. Psychol. 69, 797–811 (1995).
Schmader, T. & Johns, M. Converging evidence that stereotype threat reduces working memory capacity. J. Pers. Soc. Psychol. 85, 440–452 (2004).
Gray, J. R., Braver, T. S. & Raichle, M. E. Integration of emotion and cognition in the lateral prefrontal cortex. Proc. Natl Acad. Sci. USA 99, 4115–4120 (2002).
Bishop, S., Duncan, J., Brett, M. & Lawrence, A. D. Prefrontal cortical function and anxiety: controlling attention to threat-related stimuli. Nature Neurosci. 7, 184–188 (2004).
Mellers, B., Hertwig, R. & Kahneman, D. Do frequency representations eliminate conjunction effects? An exercise in adversarial collaboration. Psychol. Sci. 12, 269–275 (2001).
Thurstone, L. L. The Vectors of Mind (Univ. Chicago Press, Chicago, 1940).
Sternberg, R. J. The theory of successful intelligence. Rev. Gen. Psychol. 3, 292–316 (1999).
Baumeister, R. F., Twenge, J. & Nuss, C. K. Effects of social exclusion on cognitive processes: anticipated aloneness reduces intelligent thought. J. Pers. Soc. Psychol. 83, 817–827 (2002).
Snow, R. E., Kyllonen, P. C. & Marshalek, B. in Advances in the Psychology of Human Intelligence Vol. 2 (ed. Sternberg, R. J.) 47–103 (Erlbaum, Hillsdale, 1984).
Flynn, J. R. Massive IQ gains in 14 nations: what IQ tests really measure. Psychol. Bull. 101, 171–191 (1987).
Plomin, R., DeFries, J. C. & Loehlin, J. C. Genotype–environment interaction and correlation in the analysis of human behavior. Psychol. Bull. 84, 309–322 (1977).
Block, N. How heritability misleads about race. Cognition 56, 99–128 (1995).
Neale, M. C. Mx: Statistical Modeling (Virginia Commonwealth Univ., Richmond, 1997).
Sesardic, N. Heritability and indirect causation. Proc. Phil. Sci. Assoc. (2002). Available at; http://philsci-archive.pitt.edu/archive/00001097/index.html
Lewontin, R. Race and Intelligence. Bull. At. Sci. 2–8 Mar (1970). Reprinted in The IQ Controversy (eds Block, N. & Dworkin, G.) 78–92 (Pantheon, New York, 1976).
Mortensen, E. L., Michaelsen, K. F., Sanders, S. A. & Reinisch, J. M. The association between duration of breastfeeding and adult intelligence. JAMA 287, 2365–2371 (2002).
Brumback, C. R., Low, K. A., Gratton, G. & Fabiani, M. Sensory ERPs predict differences in working memory span and fluid intelligence. Neuroreport 15, 373–376 (2004).
Haier, R. J, Jung, R. E., Yeo, R. A., Head, K. & Alkire, M. T. Structural brain variation and general intelligence. Neuroimage (in the press).
Farah, M. J. et al. Neurocognitive enhancement: what can we do and what should we do? Nature Rev. Neurosci. 5, 421–426 (2004).
Acknowledgements
This work was supported in part by research grants from the National Institute of Mental Health to J.R.G., and from the National Institute for Biomedical Imaging and Bioengineering and the National Center for Research Resources to P.M.T. The authors thank R. J. Sternberg and W. R. Gray for their comments on a draft.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- FLUID INTELLIGENCE
-
(Gf). 'On the spot' reasoning and novel problem-solving ability.
- INTELLIGENCE QUOTIENT
-
(IQ). Intelligence scaled by age, standardized to have a population mean of 100 and SD of 15 (mental age divided by chronological age). It produces an intelligence ranking relative to other individuals of the same age, which tends to be stable across the lifespan of the individual.
- CRYSTALLIZED INTELLIGENCE
-
(Gc). Performance guided by overlearned skills or knowledge, such as vocabulary.
- g LOADING (OF A TASK)
-
A measure of how strongly individual differences in performance on a particular task predict differences in general intelligence (g). A task with a high g loading will better reveal differences between people of higher and lower intelligence.
- WORKING MEMORY
-
A cognitive/neural system for maintaining information actively in mind (storage) and manipulating it (executive processing), or holding it in mind despite potential distraction or interference (control of attention).
- PSYCHOMETRIC INTELLIGENCE
-
Intelligence as measured by an IQ-type test, typically assessing the accuracy of a response (and not the speed).
- INSPECTION TIME
-
The minimum duration of exposure to a visual stimulus that a study participant requires to respond accurately about that stimulus.
- GENETIC POLYMORPHISMS
-
Different variants of the same gene.
- HOMEOBOX GENE
-
A key regulator of embryonic development, highly conserved across species, which controls cell and organ differentiation. Homeobox genes can activate many other genes, producing entire body segments.
- QUANTITATIVE TRAIT LOCUS
-
(QTL). A genetic polymorphism that affects the expression of a continuously distributed phenotype. Typically, QTLs are statistically associated with trait variations that depend on multiple interacting loci.
- POLYGENIC
-
A complex trait is polygenic if it is determined by multiple genes that each have small effects and that can interact with each other to produce effects.
- ENDOPHENOTYPES
-
Gene products (phenotypes) that are not visible.
Rights and permissions
About this article
Cite this article
Gray, J., Thompson, P. Neurobiology of intelligence: science and ethics. Nat Rev Neurosci 5, 471–482 (2004). https://doi.org/10.1038/nrn1405
Issue Date:
DOI: https://doi.org/10.1038/nrn1405
This article is cited by
-
Neural activity and network analysis for understanding reasoning using the matrix reasoning task
Cognitive Processing (2023)
-
Aiding applicants: leveling the playing field within the immediate acceptance mechanism
Review of Economic Design (2023)
-
Test–retest reliability and convergent validity of the test of nonverbal intelligence-fourth edition in patients with schizophrenia
BMC Psychiatry (2021)
-
Participation in and provision of public goods: Does granularity matter?
Journal of Economic Interaction and Coordination (2021)
-
Probing the relationship between late endogenous ERP components with fluid intelligence in healthy older adults
Scientific Reports (2020)