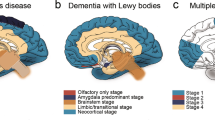
Key Points
-
Several classes of mammalian models of α-synucleinopathy are available for use in drug development. These include transgenic models and models based on delivery of α-synuclein protein (such as preformed fibrils (PFFs) or extracts of Lewy bodies from post-mortem human tissue) or α-synuclein-encoding genes (for example, using adeno-associated viruses (AAVs)).
-
These models are being applied to assess efficacy, inform dosing and define therapeutic windows of potential new treatments.
-
The AAV models stand out as drug development platforms as, to date, they are robust and amenable to medium-throughput evaluation and can be used in multiple species from mouse to rat to non-human primate (NHP). The transgenic and PFF models may prove important for longer-term evaluation of evolving symptoms and for examining pathology in multiple systems, whereas the Lewy extract models are not in a position for widespread use, as patient material will differ between experiments, and duration to reach end points, so far, exceeds a time frame for relatively rapid screening of candidates.
-
AAV NHP models are especially well suited as a final stage before initiating clinical development, not only for evaluating the efficacy of a treatment and the clinically relevant measures of target engagement, but also for defining the therapeutic index of a candidate in a species best able to predict effects anticipated in human.
-
Navigating the choice of different models available is often difficult. However, with the understanding of the mechanism of action of the proposed treatment, along with consideration of potential species interactions, one can now rationally define a development plan that uses available models most optimally to advance through the preclinical space.
Abstract
A major challenge in Parkinson disease (PD) will be to turn an emerging and expanding pipeline of novel disease-modifying candidate compounds into therapeutics. Novel targets need in vivo validation, and candidate therapeutics require appropriate preclinical platforms on which to define potential efficacy and target engagement before advancement to clinical development. We propose that α-synuclein (α-syn)-based mammalian models will be crucial for this process. Here, we review α-syn transgenic mouse models, viral vector models of α-syn overexpression and models of 'prion-like' spread of α-syn, and describe how each of these model types may contribute to PD drug discovery. We conclude by presenting our opinion on how to use a combination of these models through the late-stage preclinical, proof-of-principle investigation of novel therapeutics.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Pringsheim, T., Jette, N., Frolkis, A. & Steeves, T. D. The prevalence of Parkinson's disease: a systematic review and meta-analysis. Mov. Disord. 29, 1583–1590 (2014).
Kalia, L. V. & Lang, A. E. Parkinson's disease. Lancet 386, 896–912 (2015).
Spillantini, M. G. et al. α-Synuclein in Lewy bodies. Nature 388, 839–840 (1997).
Schneider, J. S. et al. GM1 ganglioside in Parkinson's disease: pilot study of effects on dopamine transporter binding. J. Neurol. Sci. 356, 118–123 (2015).
Schneider, J. S., Seyfried, T. N., Choi, H. S. & Kidd, S. K. Intraventricular sialidase administration enhances GM1 ganglioside expression and is partially neuroprotective in a mouse model of Parkinson's disease. PLoS ONE 10, e0143351 (2015). This study provides early preclinical support for the current clinical development of GM1 ganglioside for disease modification in PD.
Guo, J. F. et al. Involvement of Bcl-2-associated athanogene (BAG)-family proteins in the neuroprotection by rasagiline. Int. J. Clin. Exp. Med. 8, 18158–18164 (2015).
Mandel, S. A., Sagi, Y. & Amit, T. Rasagiline promotes regeneration of substantia nigra dopaminergic neurons in post-MPTP-induced Parkinsonism via activation of tyrosine kinase receptor signaling pathway. Neurochem. Res. 32, 1694–1699 (2007).
Eriksen, J. L., Dawson, T. M., Dickson, D. W. & Petrucelli, L. Caught in the act: α-synuclein is the culprit in Parkinson's disease. Neuron 40, 453–456 (2003).
Falkenburger, B. H., Saridaki, T. & Dinter, E. Cellular models for Parkinson's disease. J. Neurochem. 139 (Suppl. 1), 121–130 (2016).
Javed, H., Kamal, M. A. & Ojha, S. An overview on the role of α-synuclein in experimental models of Parkinson's disease from pathogenesis to therapeutics. CNS Neurol. Disord. Drug Targets 15, 1240–1252 (2016).
Peelaerts, W. & Baekelandt, V. α-Synuclein strains and the variable pathologies of synucleinopathies. J. Neurochem. 139 (Suppl. 1), 256–274 (2016).
Luth, E. S., Bartels, T., Dettmer, U., Kim, N. C. & Selkoe, D. J. Purification of α-synuclein from human brain reveals an instability of endogenous multimers as the protein approaches purity. Biochemistry 54, 279–292 (2015).
Logan, T., Bendor, J., Toupin, C., Thorn, K. & Edwards, R. H. α-Synuclein promotes dilation of the exocytotic fusion pore. Nat. Neurosci. 20, 681–689 (2017).
Abeliovich, A. et al. Mice lacking α-synuclein display functional deficits in the nigrostriatal dopamine system. Neuron 25, 239–252 (2000).
Chandra, S. et al. Double-knockout mice for α- and β-synucleins: effect on synaptic functions. Proc. Natl Acad. Sci. USA 101, 14966–14971 (2004).
Greten-Harrison, B. et al. αβγ-synuclein triple knockout mice reveal age-dependent neuronal dysfunction. Proc. Natl Acad. Sci. USA 107, 19573–19578 (2010).
Burre, J. et al. α-Synuclein promotes SNARE-complex assembly in vivo and in vitro. Science 329, 1663–1667 (2010).
Choi, B. K. et al. Large α-synuclein oligomers inhibit neuronal SNARE-mediated vesicle docking. Proc. Natl Acad. Sci. USA 110, 4087–4092 (2013).
Lundblad, M., Decressac, M., Mattsson, B. & Bjorklund, A. Impaired neurotransmission caused by overexpression of α-synuclein in nigral dopamine neurons. Proc. Natl Acad. Sci. USA 109, 3213–3219 (2012). This study uses in vivo amperometry to measure dopamine changes in the striatum and shows that α-syn is involved in synaptic dopamine release in the basal ganglia.
Taylor, T. N. et al. Region-specific deficits in dopamine, but not norepinephrine, signaling in a novel A30P α-synuclein BAC transgenic mouse. Neurobiol. Dis. 62, 193–207 (2014).
Platt, N. J., Gispert, S., Auburger, G. & Cragg, S. J. Striatal dopamine transmission is subtly modified in human A53Tα-synuclein overexpressing mice. PLoS ONE 7, e36397 (2012).
Nemani, V. M. et al. Increased expression of α-synuclein reduces neurotransmitter release by inhibiting synaptic vesicle reclustering after endocytosis. Neuron 65, 66–79 (2010).
Scott, D. & Roy, S. α-Synuclein inhibits intersynaptic vesicle mobility and maintains recycling-pool homeostasis. J. Neurosci. 32, 10129–10135 (2012).
Cooper, A. A. et al. α-Synuclein blocks ER–Golgi traffic and Rab1 rescues neuron loss in Parkinson's models. Science 313, 324–328 (2006).
Gitler, A. D. et al. The Parkinson's disease protein α-synuclein disrupts cellular Rab homeostasis. Proc. Natl Acad. Sci. USA 105, 145–150 (2008).
Chung, C. Y. et al. Identification and rescue of α-synuclein toxicity in Parkinson patient-derived neurons. Science 342, 983–987 (2013). This important paper shows how patient-derived neurons and yeast models of α-synucleinopathy can direct drug target discovery programmes.
Tardiff, D. F. et al. Yeast reveal a “druggable” Rsp5/Nedd4 network that ameliorates α-synuclein toxicity in neurons. Science 342, 979–983 (2013).
Tardiff, D. F., Khurana, V., Chung, C. Y. & Lindquist, S. From yeast to patient neurons and back again: powerful new discovery platform. Mov. Disord. 29, 1231–1240 (2014).
Lee, V. M. & Trojanowski, J. Q. Mechanisms of Parkinson's disease linked to pathological α-synuclein: new targets for drug discovery. Neuron 52, 33–38 (2006).
Maingay, M., Romero-Ramos, M., Carta, M. & Kirik, D. Ventral tegmental area dopamine neurons are resistant to human mutant α-synuclein overexpression. Neurobiol. Dis. 23, 522–532 (2006).
Guzman, J. N., Sanchez-Padilla, J., Chan, C. S. & Surmeier, D. J. Robust pacemaking in substantia nigra dopaminergic neurons. J. Neurosci. 29, 11011–11019 (2009).
Matsuda, W. et al. Single nigrostriatal dopaminergic neurons form widely spread and highly dense axonal arborizations in the neostriatum. J. Neurosci. 29, 444–453 (2009). This elegant study shows the extent of the burden of SN dopamine neurons and, in doing so, contributes to our understanding of vulnerabilities of these neurons in PD.
Braak, H. & Del Tredici, K. Invited article: nervous system pathology in sporadic Parkinson disease. Neurology 70, 1916–1925 (2008).
Fuchs, J. et al. Genetic variability in the SNCA gene influences α-synuclein levels in the blood and brain. FASEB J. 22, 1327–1334 (2008).
Greenbaum, E. A. et al. The E46K mutation in α-synuclein increases amyloid fibril formation. J. Biol. Chem. 280, 7800–7807 (2005).
Mata, I. F. et al. SNCA variant associated with Parkinson disease and plasma α-synuclein level. Arch. Neurol. 67, 1350–1356 (2010).
Hinault, M. P. et al. Stable α-synuclein oligomers strongly inhibit chaperone activity of the Hsp70 system by weak interactions with J-domain co-chaperones. J. Biol. Chem. 285, 38173–38182 (2010).
Xilouri, M., Brekk, O. R. & Stefanis, L. Autophagy and α-synuclein: relevance to Parkinson's disease and related synucleopathies. Mov. Disord. 31, 178–192 (2016).
Dryanovski, D. I. et al. Calcium entry and α-synuclein inclusions elevate dendritic mitochondrial oxidant stress in dopaminergic neurons. J. Neurosci. 33, 10154–10164 (2013).
Guardia-Laguarta, C. et al. α-Synuclein is localized to mitochondria-associated ER membranes. J. Neurosci. 34, 249–259 (2014).
Mosharov, E. V. et al. Interplay between cytosolic dopamine, calcium, and α-synuclein causes selective death of substantia nigra neurons. Neuron 62, 218–229 (2009).
Volpicelli-Daley, L. A. Effects of α-synuclein on axonal transport. Neurobiol. Dis. http://dx.doi.org/10.1016/j.nbd.2016.12.008 (2016).
Kim, C. et al. Antagonizing neuronal Toll-like receptor 2 prevents synucleinopathy by activating autophagy. Cell Rep. 13, 771–782 (2015).
Wang, S. et al. α-Synuclein, a chemoattractant, directs microglial migration via H2O2-dependent Lyn phosphorylation. Proc. Natl Acad. Sci. USA 112, E1926–E1935 (2015).
Wong, Y. C. & Krainc, D. α-Synuclein toxicity in neurodegeneration: mechanism and therapeutic strategies. Nat. Med. 23, 1–13 (2017).
Tofaris, G. K. et al. Pathological changes in dopaminergic nerve cells of the substantia nigra and olfactory bulb in mice transgenic for truncated human α-synuclein(1–120): implications for Lewy body disorders. J. Neurosci. 26, 3942–3950 (2006).
Wakamatsu, M. et al. Selective loss of nigral dopamine neurons induced by overexpression of truncated human α-synuclein in mice. Neurobiol. Aging 29, 574–585 (2008).
Daher, J. P. et al. Conditional transgenic mice expressing C-terminally truncated human α-synuclein (αSyn119) exhibit reduced striatal dopamine without loss of nigrostriatal pathway dopaminergic neurons. Mol. Neurodegener. 4, 34 (2009).
Rieker, C. et al. Neuropathology in mice expressing mouse α-synuclein. PLoS ONE 6, e24834 (2011).
Chandra, S., Gallardo, G., Fernandez-Chacon, R., Schluter, O. M. & Sudhof, T. C. α-Synuclein cooperates with CSPα in preventing neurodegeneration. Cell 123, 383–396 (2005).
Richfield, E. K. et al. Behavioral and neurochemical effects of wild-type and mutated human α-synuclein in transgenic mice. Exp. Neurol. 175, 35–48 (2002).
Manning-Bog, A. B., McCormack, A. L., Purisai, M. G., Bolin, L. M. & Di Monte, D. A. α-Synuclein overexpression protects against paraquat-induced neurodegeneration. J. Neurosci. 23, 3095–3099 (2003).
Matsuoka, Y. et al. Lack of nigral pathology in transgenic mice expressing human α-synuclein driven by the tyrosine hydroxylase promoter. Neurobiol. Dis. 8, 535–539 (2001).
Lin, X. et al. Leucine-rich repeat kinase 2 regulates the progression of neuropathology induced by Parkinson's-disease-related mutant α-synuclein. Neuron 64, 807–827 (2009).
Lim, Y., Kehm, V. M., Li, C., Trojanowski, J. Q. & Lee, V. M. Forebrain overexpression of α-synuclein leads to early postnatal hippocampal neuron loss and synaptic disruption. Exp. Neurol. 221, 86–97 (2010).
Lin, X. et al. Conditional expression of Parkinson's disease-related mutant α-synuclein in the midbrain dopaminergic neurons causes progressive neurodegeneration and degradation of transcription factor nuclear receptor related 1. J. Neurosci. 32, 9248–9264 (2012).
Nuber, S. et al. Neurodegeneration and motor dysfunction in a conditional model of Parkinson's disease. J. Neurosci. 28, 2471–2484 (2008).
Nuber, S. et al. Olfactory neuron-specific expression of A30P α-synuclein exacerbates dopamine deficiency and hyperactivity in a novel conditional model of early Parkinson's disease stages. Neurobiol. Dis. 44, 192–204 (2011).
Kuo, Y. M. et al. Extensive enteric nervous system abnormalities in mice transgenic for artificial chromosomes containing Parkinson disease-associated α-synuclein gene mutations precede central nervous system changes. Hum. Mol. Genet. 19, 1633–1650 (2010).
Janezic, S. et al. Deficits in dopaminergic transmission precede neuron loss and dysfunction in a new Parkinson model. Proc. Natl Acad. Sci. USA 110, E4016–E4025 (2013). This key paper describes early events leading up to neurodegeneration, enhancing our understanding of physiological responses to α-syn overexpression.
Petit, G. H. et al. Rasagiline ameliorates olfactory deficits in an α-synuclein mouse model of Parkinson's disease. PLoS ONE 8, e60691 (2013).
Rasmussen, N. B. et al. 5-HT2A receptor binding in the frontal cortex of Parkinson's disease patients and α-synuclein overexpressing mice: a postmortem study. Parkinsons Dis. 2016, 3682936 (2016).
Westerlund, M. et al. Lrrk2 and α-synuclein are co-regulated in rodent striatum. Mol. Cell. Neurosci. 39, 586–591 (2008).
Gispert, S. et al. Transgenic mice expressing mutant A53T human α-synuclein show neuronal dysfunction in the absence of aggregate formation. Mol. Cell. Neurosci. 24, 419–429 (2003).
Yavich, L. et al. Locomotor activity and evoked dopamine release are reduced in mice overexpressing A30P-mutated human α-synuclein. Neurobiol. Dis. 20, 303–313 (2005).
Fleming, S. M. et al. Olfactory deficits in mice overexpressing human wildtype α-synuclein. Eur. J. Neurosci. 28, 247–256 (2008).
Giasson, B. I. et al. Neuronal α-synucleinopathy with severe movement disorder in mice expressing A53T human α-synuclein. Neuron 34, 521–533 (2002).
Hallett, P. J., McLean, J. R., Kartunen, A., Langston, J. W. & Isacson, O. α-Synuclein overexpressing transgenic mice show internal organ pathology and autonomic deficits. Neurobiol. Dis. 47, 258–267 (2012).
Kahle, P. J. et al. Selective insolubility of α-synuclein in human Lewy body diseases is recapitulated in a transgenic mouse model. Am. J. Pathol. 159, 2215–2225 (2001).
Lee, H. J. et al. Transmission of synucleinopathies in the enteric nervous system of A53T α-synuclein transgenic mice. Exp. Neurobiol. 20, 181–188 (2011).
Neumann, M. et al. Misfolded proteinase K-resistant hyperphosphorylated α-synuclein in aged transgenic mice with locomotor deterioration and in human α-synucleinopathies. J. Clin. Invest. 110, 1429–1439 (2002).
Emmer, K. L., Waxman, E. A., Covy, J. P. & Giasson, B. I. E46K human α-synuclein transgenic mice develop Lewy-like and tau pathology associated with age-dependent, detrimental motor impairment. J. Biol. Chem. 286, 35104–35118 (2011).
Lee, M. K. et al. Human α-synuclein-harboring familial Parkinson's disease-linked Ala-53 → Thr mutation causes neurodegenerative disease with α-synuclein aggregation in transgenic mice. Proc. Natl Acad. Sci. USA 99, 8968–8973 (2002).
Cabin, D. E. et al. Exacerbated synucleinopathy in mice expressing A53T SNCA on a Snca null background. Neurobiol. Aging 26, 25–35 (2005).
Braak, H., Sastre, M., Bohl, J. R., de Vos, R. A. & Del Tredici, K. Parkinson's disease: lesions in dorsal horn layer I, involvement of parasympathetic and sympathetic pre- and postganglionic neurons. Acta Neuropathol. 113, 421–429 (2007).
Beach, T. G. et al. Multi-organ distribution of phosphorylated α-synuclein histopathology in subjects with Lewy body disorders. Acta Neuropathol. 119, 689–702 (2010).
Braak, H. et al. Staging of the intracerebral inclusion body pathology associated with idiopathic Parkinson's disease (preclinical and clinical stages). J. Neurol. 249 (Suppl. 3), iii1–iii5 (2002).
Braak, H., Ghebremedhin, E., Rub, U., Bratzke, H. & Del Tredici, K. Stages in the development of Parkinson's disease-related pathology. Cell Tissue Res. 318, 121–134 (2004).
Farrell, K. F. et al. Non-motor parkinsonian pathology in aging A53T α-synuclein mice is associated with progressive synucleinopathy and altered enzymatic function. J. Neurochem. 128, 536–546 (2014).
Noorian, A. R. et al. α-Synuclein transgenic mice display age-related slowing of gastrointestinal motility associated with transgene expression in the vagal system. Neurobiol. Dis. 48, 9–19 (2012). This important article demonstrates the link between α-syn expression in the vagal system and gastrointestinal motility.
Wang, L. et al. Mice overexpressing wild-type human α-synuclein display alterations in colonic myenteric ganglia and defecation. Neurogastroenterol. Motil. 24, e425–e436 (2012).
Bencsik, A., Muselli, L., Leboidre, M., Lakhdar, L. & Baron, T. Early and persistent expression of phosphorylated α-synuclein in the enteric nervous system of A53T mutant human α-synuclein transgenic mice. J. Neuropathol. Exp. Neurol. 73, 1144–1151 (2014).
Price, D. L. et al. Longitudinal live imaging of retinal α-synuclein::GFP deposits in a transgenic mouse model of Parkinson's disease/dementia with Lewy bodies. Sci. Rep. 6, 29523 (2016).
Gray, M. T., Munoz, D. G., Gray, D. A., Schlossmacher, M. G. & Woulfe, J. M. α-Synuclein in the appendiceal mucosa of neurologically intact subjects. Mov. Disord. 29, 991–998 (2014).
Visanji, N. P. et al. Colonic mucosal α-synuclein lacks specificity as a biomarker for Parkinson disease. Neurology 84, 609–616 (2015).
Antunes, L. et al. Similar α-synuclein staining in the colon mucosa in patients with Parkinson's disease and controls. Mov. Disord. 31, 1567–1570 (2016).
Koob, A. O. et al. Lovastatin ameliorates α-synuclein accumulation and oxidation in transgenic mouse models of α-synucleinopathies. Exp. Neurol. 221, 267–274 (2010).
Shaltiel-Karyo, R. et al. A blood–brain barrier (BBB) disrupter is also a potent α-synuclein (α-syn) aggregation inhibitor: a novel dual mechanism of mannitol for the treatment of Parkinson disease (PD). J. Biol. Chem. 288, 17579–17588 (2013).
Price, D. L. et al. Alterations in mGluR5 expression and signaling in Lewy body disease and in transgenic models of α-synucleinopathy — implications for excitotoxicity. PLoS ONE 5, e14020 (2010).
Martin, L. J. et al. Parkinson's disease α-synuclein transgenic mice develop neuronal mitochondrial degeneration and cell death. J. Neurosci. 26, 41–50 (2006).
Finkelstein, D. I. et al. Clioquinol improves cognitive, motor function, and microanatomy of the α-synuclein hA53T transgenic mice. ACS Chem. Neurosci. 7, 119–129 (2016).
Kachroo, A. & Schwarzschild, M. A. Adenosine A2A receptor gene disruption protects in an α-synuclein model of Parkinson's disease. Ann. Neurol. 71, 278–282 (2012).
Kurz, A. et al. A53T-α-synuclein overexpression impairs dopamine signaling and striatal synaptic plasticity in old mice. PLoS ONE 5, e11464 (2010).
Clark, J. et al. Oral N-acetyl-cysteine attenuates loss of dopaminergic terminals in α-synuclein overexpressing mice. PLoS ONE 5, e12333 (2010).
Hansen, C. et al. A novel α-synuclein–GFP mouse model displays progressive motor impairment, olfactory dysfunction and accumulation of α-synuclein–GFP. Neurobiol. Dis. 56, 145–155 (2013).
Gomez-Isla, T. et al. Motor dysfunction and gliosis with preserved dopaminergic markers in human α-synuclein A30P transgenic mice. Neurobiol. Aging 24, 245–258 (2003).
Freichel, C. et al. Age-dependent cognitive decline and amygdala pathology in α-synuclein transgenic mice. Neurobiol. Aging 28, 1421–1435 (2007).
Rothman, S. M. et al. Neuronal expression of familial Parkinson's disease A53T α-synuclein causes early motor impairment, reduced anxiety and potential sleep disturbances in mice. J. Parkinsons Dis. 3, 215–229 (2013).
Wakamatsu, M., Iwata, S., Funakoshi, T. & Yoshimoto, M. Dopamine receptor agonists reverse behavioral abnormalities of α-synuclein transgenic mouse, a new model of Parkinson's disease. J. Neurosci. Res. 86, 640–646 (2008).
Kalia, L. V., Kalia, S. K. & Lang, A. E. Disease-modifying strategies for Parkinson's disease. Mov. Disord. 30, 1442–1450 (2015).
Paumier, K. L. et al. Behavioral characterization of A53T mice reveals early and late stage deficits related to Parkinson's disease. PLoS ONE 8, e70274 (2013).
Games, D. et al. Reducing C-terminal-truncated α-synuclein by immunotherapy attenuates neurodegeneration and propagation in Parkinson's disease-like models. J. Neurosci. 34, 9441–9454 (2014).
George, S. et al. α-Synuclein transgenic mice exhibit reduced anxiety-like behaviour. Exp. Neurol. 210, 788–792 (2008).
Graham, D. R. & Sidhu, A. Mice expressing the A53T mutant form of human α-synuclein exhibit hyperactivity and reduced anxiety-like behavior. J. Neurosci. Res. 88, 1777–1783 (2010).
Yamakado, H. et al. α-Synuclein BAC transgenic mice as a model for Parkinson's disease manifested decreased anxiety-like behavior and hyperlocomotion. Neurosci. Res. 73, 173–177 (2012).
Kudo, T., Loh, D. H., Truong, D., Wu, Y. & Colwell, C. S. Circadian dysfunction in a mouse model of Parkinson's disease. Exp. Neurol. 232, 66–75 (2011).
Zhang, S., Xiao, Q. & Le, W. Olfactory dysfunction and neurotransmitter disturbance in olfactory bulb of transgenic mice expressing human A53T mutant α-synuclein. PLoS ONE 10, e0119928 (2015).
Wang, L., Fleming, S. M., Chesselet, M. F. & Tache, Y. Abnormal colonic motility in mice overexpressing human wild-type α-synuclein. Neuroreport 19, 873–876 (2008).
Rockenstein, E. et al. Differential neuropathological alterations in transgenic mice expressing α-synuclein from the platelet-derived growth factor and Thy-1 promoters. J. Neurosci. Res. 68, 568–578 (2002).
Hebron, M. L., Lonskaya, I. & Moussa, C. E. Nilotinib reverses loss of dopamine neurons and improves motor behavior via autophagic degradation of α-synuclein in Parkinson's disease models. Hum. Mol. Genet. 22, 3315–3328 (2013).
Kohl, Z. et al. Fluoxetine rescues impaired hippocampal neurogenesis in a transgenic A53T synuclein mouse model. Eur. J. Neurosci. 35, 10–19 (2012).
Mandler, M. et al. Next-generation active immunization approach for synucleinopathies: implications for Parkinson's disease clinical trials. Acta Neuropathol. 127, 861–879 (2014).
Masliah, E. et al. Effects of α-synuclein immunization in a mouse model of Parkinson's disease. Neuron 46, 857–868 (2005).
Masliah, E. et al. Passive immunization reduces behavioral and neuropathological deficits in an α-synuclein transgenic model of Lewy body disease. PLoS ONE 6, e19338 (2011). This is a preclinical study using a transgenic mouse model showing that administration of antibodies to α-syn ameliorates behavioural deficits and pathology.
Lindstrom, V. et al. Immunotherapy targeting α-synuclein protofibrils reduced pathology in (Thy-1)-h[A30P] α-synuclein mice. Neurobiol. Dis. 69, 134–143 (2014).
Roy, A., Rangasamy, S. B., Kundu, M. & Pahan, K. BPOZ-2 gene delivery ameliorates α-synucleinopathy in A53T transgenic mouse model of Parkinson's disease. Sci. Rep. 6, 22067 (2016).
Rocha, E. M. et al. Glucocerebrosidase gene therapy prevents α-synucleinopathy of midbrain dopamine neurons. Neurobiol. Dis. 82, 495–503 (2015).
Niu, Y. et al. Early Parkinson's disease symptoms in α-synuclein transgenic monkeys. Hum. Mol. Genet. 24, 2308–2317 (2015).
Baekelandt, V. et al. Characterization of lentiviral vector-mediated gene transfer in adult mouse brain. Hum. Gene Ther. 13, 841–853 (2002).
Kirik, D. et al. Parkinson-like neurodegeneration induced by targeted overexpression of α-synuclein in the nigrostriatal system. J. Neurosci. 22, 2780–2791 (2002).
Lo Bianco, C., Ridet, J.-L., Schneider, B. L., Deglon, N. & Aebischer, P. α-Synucleinopathy and selective dopaminergic neuron loss in a rat lentiviral-based model of Parkinson's disease. Proc. Natl Acad. Sci. USA 99, 10813–10818 (2002).
Lo Bianco, C. et al. Lentiviral vector delivery of parkin prevents dopaminergic degeneration in an α-synuclein rat model of Parkinson's disease. Proc. Natl Acad. Sci. USA 101, 17510–17515 (2004).
Klein, R. L., King, M. A., Hamby, M. E. & Meyer, E. M. Dopaminergic cell loss induced by human A30P α-synuclein gene transfer to the rat substantia nigra. Hum. Gene Ther. 13, 605–612 (2002).
St Martin, J. L. et al. Dopaminergic neuron loss and up-regulation of chaperone protein mRNA induced by targeted over-expression of α-synuclein in mouse substantia nigra. J. Neurochem. 100, 1449–1457 (2007).
Theodore, S., Cao, S., McLean, P. J. & Standaert, D. G. Targeted overexpression of human α-synuclein triggers microglial activation and an adaptive immune response in a mouse model of Parkinson disease. J. Neuropathol. Exp. Neurol. 67, 1149–1158 (2008).
Decressac, M., Mattsson, B., Lundblad, M., Weikop, P. & Bjorklund, A. Progressive neurodegenerative and behavioural changes induced by AAV-mediated overexpression of α-synuclein in midbrain dopamine neurons. Neurobiol. Dis. 45, 939–953 (2012).
Van der Perren, A. et al. Longitudinal follow-up and characterization of a robust rat model for Parkinson's disease based on overexpression of α-synuclein with adeno-associated viral vectors. Neurobiol. Aging 36, 1543–1558 (2015).
Kordower, J. H. et al. Disease duration and the integrity of the nigrostriatal system in Parkinson's disease. Brain 136, 2419–2431 (2013). This is a well-controlled study providing evidence of the degree of dopaminergic anatomy that remains at varying stages of PD — important for consideration of early-start clinical trials.
Fearnley, J. M. & Lees, A. J. Ageing and Parkinson's disease: substantia nigra regional selectivity. Brain 114, 2283–2301 (1991).
Greffard, S. et al. Motor score of the Unified Parkinson Disease Rating Scale as a good predictor of Lewy body-associated neuronal loss in the substantia nigra. Arch. Neurol. 63, 584–588 (2006).
Ma, S. Y., Roytta, M., Rinne, J. O., Collan, Y. & Rinne, U. K. Correlation between neuromorphometry in the substantia nigra and clinical features in Parkinson's disease using disector counts. J. Neurol. Sci. 151, 83–87 (1997).
Cheng, H. C., Ulane, C. M. & Burke, R. E. Clinical progression in Parkinson disease and the neurobiology of axons. Ann. Neurol. 67, 715–725 (2010).
Kirik, D., Rosenblad, C. & Bjorklund, A. Characterization of behavioral and neurodegenerative changes following partial lesions of the nigrostriatal dopamine system induced by intrastriatal 6-hydroxydopamine in the rat. Exp. Neurol. 152, 259–277 (1998).
Mayo, J. C. et al. Melatonin and Parkinson's disease. Endocrine 27, 169–178 (2005).
Koprich, J., Johnston, T., Reyes, M., Sun, X. & Brotchie, J. Expression of human A53T α-synuclein in the rat substantia nigra using a novel AAV1/2 vector produces a rapidly evolving pathology with protein aggregation, dystrophic neurite architecture and nigrostriatal degeneration with potential to model the pathology of Parkinson's disease. Mol. Neurodegener. 5, 43 (2010).
Koprich, J. B. et al. Progressive neurodegeneration or endogenous compensation in an animal model of Parkinson's disease produced by decreasing doses of α-synuclein. PLoS ONE 6, e17698 (2011).
Azeredo da Silveira, S. et al. Phosphorylation does not prompt, nor prevent, the formation of α-synuclein toxic species in a rat model of Parkinson's disease. Hum. Mol. Genet. 18, 872–887 (2009).
Oliveras-Salva, M. et al. rAAV2/7 vector-mediated overexpression of α-synuclein in mouse substantia nigra induces protein aggregation and progressive dose-dependent neurodegeneration. Mol. Neurodegener. 8, 44 (2013).
Yamada, M., Iwatsubo, T., Mizuno, Y. & Mochizuki, H. Overexpression of α-synuclein in rat substantia nigra results in loss of dopaminergic neurons, phosphorylation of α-synuclein and activation of caspase-9: resemblance to pathogenetic changes in Parkinson's disease. J. Neurochem. 91, 451–461 (2004).
Lauwers, E. et al. Non-invasive imaging of neuropathology in a rat model of α-synuclein overexpression. Neurobiol. Aging 28, 248–257 (2007).
McFarland, N. R., Lee, J. S., Hyman, B. T. & McLean, P. J. Comparison of transduction efficiency of recombinant AAV serotypes 1, 2, 5, and 8 in the rat nigrostriatal system. J. Neurochem. 109, 838–845 (2009).
Chung, C. Y., Koprich, J. B., Siddiqi, H. & Isacson, O. Dynamic changes in presynaptic and axonal transport proteins combined with striatal neuroinflammation precede dopaminergic neuronal loss in a rat model of AAV α-synucleinopathy. J. Neurosci. 29, 3365–3373 (2009).
Chu, Y. et al. Alterations in axonal transport motor proteins in sporadic and experimental Parkinson's disease. Brain 135, 2058–2073 (2012).
He, Q. et al. Treatment with trehalose prevents behavioral and neurochemical deficits produced in an AAV α-synuclein rat model of Parkinson's disease. Mol. Neurobiol. 53, 2258–2268 (2016).
Qin, H. et al. Inhibition of the JAK/STAT pathway protects against α-synuclein-induced neuroinflammation and dopaminergic neurodegeneration. J. Neurosci. 36, 5144–5159 (2016).
Van der Perren, A. et al. FK506 reduces neuroinflammation and dopaminergic neurodegeneration in an α-synuclein-based rat model for Parkinson's disease. Neurobiol. Aging 36, 1559–1568 (2015).
Shahaduzzaman, M. et al. Anti-human α-synuclein N-terminal peptide antibody protects against dopaminergic cell death and ameliorates behavioral deficits in an AAV-α-synuclein rat model of Parkinson's disease. PLoS ONE 10, e0116841 (2015).
Aldrin-Kirk, P., Davidsson, M., Holmqvist, S., Li, J. Y. & Bjorklund, T. Novel AAV-based rat model of forebrain synucleinopathy shows extensive pathologies and progressive loss of cholinergic interneurons. PLoS ONE 9, e100869 (2014).
Ulusoy, A. et al. Caudo-rostral brain spreading of α-synuclein through vagal connections. EMBO Mol. Med. 5, 1119–1127 (2013).
Vermilyea, S. C. & Emborg, M. E. α-Synuclein and nonhuman primate models of Parkinson's disease. J. Neurosci. Methods 255, 38–51 (2015).
Eslamboli, A. et al. Long-term consequences of human α-synuclein overexpression in the primate ventral midbrain. Brain 130, 799–815 (2007). This is a follow-up study to an earlier proof-of-concept report further characterizing the effects of AAV delivery of α-syn to the primate brain.
Kirik, D. et al. Nigrostriatal α-synucleinopathy induced by viral vector-mediated overexpression of human α-synuclein: a new primate model of Parkinson's disease. Proc. Natl Acad. Sci. USA 100, 2884–2889 (2003).
Yang, W. et al. Mutant α-synuclein causes age-dependent neuropathology in monkey brain. J. Neurosci. 35, 8345–8358 (2015).
Koprich, J. B., Johnston, T. H., Reyes, G., Omana, V. & Brotchie, J. M. Towards a non-human primate model of α-synucleinopathy for development of therapeutics for Parkinson's disease: optimization of AAV1/2 delivery parameters to drive sustained expression of alpha synuclein and dopaminergic degeneration in macaque. PLoS ONE 30, e0167235 (2016).
Rey, N. L. et al. Widespread transneuronal propagation of α-synucleinopathy triggered in olfactory bulb mimics prodromal Parkinson's disease. J. Exp. Med. 213, 1759–1778 (2016).
Holmqvist, S. et al. Direct evidence of Parkinson pathology spread from the gastrointestinal tract to the brain in rats. Acta Neuropathol. 128, 805–820 (2014).
Desplats, P. et al. Inclusion formation and neuronal cell death through neuron-to-neuron transmission of α-synuclein. Proc. Natl Acad. Sci. USA 106, 13010–13015 (2009).
Hansen, C. et al. α-Synuclein propagates from mouse brain to grafted dopaminergic neurons and seeds aggregation in cultured human cells. J. Clin. Invest. 121, 715–725 (2011).
Volpicelli-Daley, L. A. et al. Exogenous α-synuclein fibrils induce Lewy body pathology leading to synaptic dysfunction and neuron death. Neuron 72, 57–71 (2011).
Kordower, J. H. et al. Transfer of host-derived α synuclein to grafted dopaminergic neurons in rat. Neurobiol. Dis. 43, 552–557 (2011).
Reyes, J. F. et al. α-Synuclein transfers from neurons to oligodendrocytes. Glia 62, 387–398 (2014).
Luk, K. C. et al. Pathological α-synuclein transmission initiates Parkinson-like neurodegeneration in nontransgenic mice. Science 338, 949–953 (2012). This study highlights how spreading of α-syn can occur in the normal mammalian brain, with implications for mechanisms of multisystem involvement in human PD.
Mougenot, A. L. et al. Transmission of prion strains in a transgenic mouse model overexpressing human A53T mutated α-synuclein. J. Neuropathol. Exp. Neurol. 70, 377–385 (2011).
Luk, K. C. et al. Intracerebral inoculation of pathological α-synuclein initiates a rapidly progressive neurodegenerative α-synucleinopathy in mice. J. Exp. Med. 209, 975–986 (2012).
Masuda-Suzukake, M. et al. Prion-like spreading of pathological α-synuclein in brain. Brain 136, 1128–1138 (2013).
Paumier, K. L. et al. Intrastriatal injection of pre-formed mouse α-synuclein fibrils into rats triggers α-synuclein pathology and bilateral nigrostriatal degeneration. Neurobiol. Dis. 82, 185–199 (2015).
Tran, H. T. et al. α-Synuclein immunotherapy blocks uptake and templated propagation of misfolded α-synuclein and neurodegeneration. Cell Rep. 7, 2054–2065 (2014).
Recasens, A. et al. Lewy body extracts from Parkinson disease brains trigger α-synuclein pathology and neurodegeneration in mice and monkeys. Ann. Neurol. 75, 351–362 (2014).
Kordower, J. H. et al. Intrastriatal alpha synuclein preformed fibrils in macaque monkeys: neuronal transport, longterm imaging and neuropathologic changes [abstract]. Society for Neuroscience Meeting 2014, Washington D.C. 409.07/I11 (2014).
Shimozawa, A. et al. Propagation of pathological α-synuclein in marmoset brain. Acta Neuropathol. Commun. 5, 12 (2017).
Ip, C. W. et al. AAV1/2-induced overexpression of A53T-α-synuclein in the substantia nigra results in degeneration of the nigrostriatal system with Lewy-like pathology and motor impairment: a new mouse model for Parkinson's disease. Acta Neuropathol. Commun. 5, 11 (2017).
Marras, C. et al. Nomenclature of genetic movement disorders: recommendations of the International Parkinson and Movement Disorder Society task force. Mov. Disord. 31, 436–457 (2016).
Moore, D. J., West, A. B., Dawson, V. L. & Dawson, T. M. Molecular pathophysiology of Parkinson's disease. Annu. Rev. Neurosci. 28, 57–87 (2005).
Trojanowski, J. Q. & Lee, V. M. Aggregation of neurofilament and α-synuclein proteins in Lewy bodies: implications for the pathogenesis of Parkinson disease and Lewy body dementia. Arch. Neurol. 55, 151–152 (1998).
Martin, I., Kim, J. W., Dawson, V. L. & Dawson, T. M. LRRK2 pathobiology in Parkinson's disease. J. Neurochem. 131, 554–565 (2014).
Nichols, R. J. et al. 14-3-3 binding to LRRK2 is disrupted by multiple Parkinson's disease-associated mutations and regulates cytoplasmic localization. Biochem. J. 430, 393–404 (2010).
Dodson, M. W. & Guo, M. Pink1, Parkin, DJ-1 and mitochondrial dysfunction in Parkinson's disease. Curr. Opin. Neurobiol. 17, 331–337 (2007).
Whitworth, A. J. & Pallanck, L. J. The PINK1/Parkin pathway: a mitochondrial quality control system? J. Bioenerg. Biomembr. 41, 499–503 (2009).
Dawson, T. M. & Dawson, V. L. The role of parkin in familial and sporadic Parkinson's disease. Mov. Disord. 25 (Suppl. 1), S32–S39 (2010).
Ko, H. S. et al. Phosphorylation by the c-Abl protein tyrosine kinase inhibits parkin's ubiquitination and protective function. Proc. Natl Acad. Sci. USA 107, 16691–16696 (2010).
Shin, J. H. et al. PARIS (ZNF746) repression of PGC-1α contributes to neurodegeneration in Parkinson's disease. Cell 144, 689–702 (2011).
Bose, A. & Beal, M. F. Mitochondrial dysfunction in Parkinson's disease. J. Neurochem. 139, 216–231 (2016).
Rousseaux, M. W. et al. Progressive dopaminergic cell loss with unilateral-to-bilateral progression in a genetic model of Parkinson disease. Proc. Natl Acad. Sci. USA 109, 15918–15923 (2012).
Kalia, L. V. et al. Clinical correlations with Lewy body pathology in LRRK2-related Parkinson disease. JAMA Neurol. 72, 100–105 (2015).
Poulopoulos, M., Levy, O. A. & Alcalay, R. N. The neuropathology of genetic Parkinson's disease. Mov. Disord. 27, 831–842 (2012).
Taipa, R. et al. DJ-1 linked parkinsonism (PARK7) is associated with Lewy body pathology. Brain 139, 1680–1687 (2016).
Polymeropoulos, M. H. et al. Mutation in the α-synuclein gene identified in families with Parkinson's disease. Science 276, 2045–2047 (1997).
Kruger, R. et al. Ala30Pro mutation in the gene encoding α-synuclein in Parkinson's disease. Nat. Genet. 18, 106–108 (1998).
Zarranz, J. J. et al. The new mutation, E46K, of α-synuclein causes Parkinson and Lewy body dementia. Ann. Neurol. 55, 164–173 (2004).
Appel-Cresswell, S. et al. α-Synuclein p.H50Q, a novel pathogenic mutation for Parkinson's disease. Mov. Disord. 28, 811–813 (2013).
Proukakis, C. et al. A novel α-synuclein missense mutation in Parkinson disease. Neurology 80, 1062–1064 (2013).
Lesage, S. et al. G51D α-synuclein mutation causes a novel parkinsonian-pyramidal syndrome. Ann. Neurol. 73, 459–471 (2013).
Martikainen, M. H., Paivarinta, M., Hietala, M. & Kaasinen, V. Clinical and imaging findings in Parkinson disease associated with the A53E SNCA mutation. Neurol. Genet. 1, e27 (2015).
Pasanen, P. et al. Novel α-synuclein mutation A53E associated with atypical multiple system atrophy and Parkinson's disease-type pathology. Neurobiol. Aging 35, 2180.e1–2180.e5 (2014).
Farrer, M. et al. Comparison of kindreds with parkinsonism and α-synuclein genomic multiplications. Ann. Neurol. 55, 174–179 (2004).
Nishioka, K. et al. Clinical heterogeneity of α-synuclein gene duplication in Parkinson's disease. Ann. Neurol. 59, 298–309 (2006).
Buell, A. K. et al. Solution conditions determine the relative importance of nucleation and growth processes in α-synuclein aggregation. Proc. Natl Acad. Sci. USA 111, 7671–7676 (2014).
Galvagnion, C. et al. Lipid vesicles trigger α-synuclein aggregation by stimulating primary nucleation. Nat. Chem. Biol. 11, 229–234 (2015).
Ibanez, P. et al. Causal relation between α-synuclein gene duplication and familial Parkinson's disease. Lancet 364, 1169–1171 (2004).
Nalls, M. A. et al. Large-scale meta-analysis of genome-wide association data identifies six new risk loci for Parkinson's disease. Nat. Genet. 46, 989–993 (2014).
Singleton, A. B. et al. α-Synuclein locus triplication causes Parkinson's disease. Science 302, 841 (2003).
Walsh, D. M. & Selkoe, D. J. A critical appraisal of the pathogenic protein spread hypothesis of neurodegeneration. Nat. Rev. Neurosci. 17, 251–260 (2016).
Engelender, S. & Isacson, O. The threshold theory for Parkinson's disease. Trends Neurosci. 40, 4–14 (2017).
Surmeier, D. J., Obeso, J. A. & Halliday, G. M. Selective neuronal vulnerability in Parkinson disease. Nat. Rev. Neurosci. 18, 101–113 (2017).
Fleming, S. M. et al. Early and progressive sensorimotor anomalies in mice overexpressing wild-type human α-synuclein. J. Neurosci. 24, 9434–9440 (2004).
Fleming, S. M. et al. A pilot trial of the microtubule-interacting peptide (NAP) in mice overexpressing alpha-synuclein shows improvement in motor function and reduction of α-synuclein inclusions. Mol. Cell. Neurosci. 46, 597–606 (2011).
Kim, C. et al. Hypoestoxide reduces neuroinflammation and α-synuclein accumulation in a mouse model of Parkinson's disease. J. Neuroinflamm. 12, 236 (2015).
Kahle, P. J. et al. Subcellular localization of wild-type and Parkinson's disease-associated mutant α-synuclein in human and transgenic mouse brain. J. Neurosci. 20, 6365–6373 (2000).
Masliah, E. et al. Dopaminergic loss and inclusion body formation in α-synuclein mice: implications for neurodegenerative disorders. Science 287, 1265–1269 (2000).
Amschl, D. et al. Time course and progression of wild type α-synuclein accumulation in a transgenic mouse model. BMC Neurosci. 14, 6 (2013).
Acknowledgements
L.V.K. holds a Canadian Institutes of Health Research (CIHR) Clinician-Scientist Award.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
J.B.K. and J.M.B. have equity stakes in, and have received consultancy fees from, Atuka Inc., Toronto, Canada, a contract research organization that provides services using some of the animal models discussed in this Review.
Glossary
- Bradykinesia
-
Slowness of movement and decrement in amplitude or speed (or progressive hesitations or halts) as movements are continued.
- 1-Methyl-4-phenyl-1,2,3,6-tetrahydropyridine
-
(MPTP). A neurotoxin that, when injected into most animals, will produce a selective lesion of the dopamine system that can be used to model nigral degeneration.
- Ganglioside
-
Sialic acid-containing glycosphingolipid differentiated by the structure of its carbohydrate chains. Gangliosides are primarily localized in plasma membranes and have prominent roles in various cell functions.
- Protofibrils, fibrils and oligomers
-
Different α-synuclein conformers associated with the pathogenesis of Lewy body diseases, including Parkinson disease. Fibrils in particular are found in abundance in Lewy bodies.
- Rapid eye movement (REM) sleep behaviour disorder
-
(RBD). A parasomnia characterized by abnormal or disruptive behaviours (such as shouting, gesturing or kicking) that occur during REM sleep and are often related to dream enactment.
- Tetracycline-controlled transcriptional activation
-
Inducible gene expression in which transcription of a target transgene is reversibly turned on or off in the presence of tetracycline or a derivative (such as doxycycline).
- pSer129
-
Phosphorylation site associated with toxic forms of α-synuclein.
- 'Core and halo' morphology
-
The classical morphology of a nigral Lewy body: a spherical cytoplasmic inclusion with a hyaline eosinophilic core and a narrow, pale-stained halo.
- Morris water maze
-
A commonly used behavioural test for mouse or rat that assesses spatial learning and memory.
- Construct validity
-
The ability of a model to measure what it is intended to measure.
- Face validity
-
The ability of a model to reproduce the clinical and pathological features of the human disease.
- Passive immunotherapies
-
Exogenous antibodies specific to an antigen (such as α-synuclein) that are delivered by intravenous, subcutaneous or intraperitoneal injection.
- Active immunotherapies
-
Vaccinations that activate the immune system of the body to produce endogenous antibodies specific to an antigen (such as α-synuclein).
- Hybrid serotype adeno-associated viruses
-
Adeno-associated viruses produced to express two viral serotypes on their particle surface.
Rights and permissions
About this article
Cite this article
Koprich, J., Kalia, L. & Brotchie, J. Animal models of α-synucleinopathy for Parkinson disease drug development. Nat Rev Neurosci 18, 515–529 (2017). https://doi.org/10.1038/nrn.2017.75
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrn.2017.75
This article is cited by
-
The Interplay of Mitochondrial Bioenergetics and Dopamine Agonists as an Effective Disease-Modifying Therapy for Parkinson’s Disease
Molecular Neurobiology (2024)
-
LRP10 and α-synuclein transmission in Lewy body diseases
Cellular and Molecular Life Sciences (2024)
-
Cortical Lewy body injections induce long-distance pathogenic alterations in the non-human primate brain
npj Parkinson's Disease (2023)
-
Impaired neuronal activity as a potential factor contributing to the underdeveloped cerebrovasculature in a young Parkinson’s disease mouse model
Scientific Reports (2023)
-
A small molecule 20C from Gastrodia elata inhibits α-synuclein aggregation and prevents progression of Parkinson’s disease
Cell Death & Disease (2023)