Abstract
Innate immunity is a highly effective set of conserved mechanisms used by multicellular organisms to recognize and counter the constant threat of microbial infections. There is evidence to indicate that innate responses are key to controlling most infections, as well as contributing to inflammatory responses that are central components of disease. In addition to Toll-like-receptor-mediated effects, many other mechanisms are used to recognize and respond to microbial threats. Natural molecules such as CpG DNA and small cationic peptides trigger innate responses that help to control infection. This indicates there is potential to utilize such compounds to activate or enhance innate responses as antimicrobials. Harnessing this activity, without associated harmful inflammatory responses, is the main challenge.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Beutler, B. Innate immunity: an overview. Mol. Immunol. 40, 845–859 (2004).
Janeway, C. A., Jr & Medzhitov, R. Innate immune recognition. Annu. Rev. Immunol. 20, 197–216 (2002).
Athman, R. & Philpott, D. Innate immunity via Toll-like receptors and Nod proteins. Curr. Opin. Microbiol. 7, 25–32 (2004).
Hoebe, K. et al. Identification of Lps2 as a key transducer of MyD88-independent TIR signalling. Nature 424, 743–748 (2003).
Serbina, N. V. et al. Sequential MyD88-independent and -dependent activation of innate immune responses to intracellular bacterial infection. Immunity 19, 891–901 (2003).
Sugawara, I., Yamada, H., Mizuno, S., Takeda, K. & Akira, S. Mycobacterial infection in MyD88-deficient mice. Microbiol. Immunol. 47, 841–847 (2003).
Shi, S. et al. MyD88 primes macrophages for full-scale activation by interferon-γ yet mediates few responses to Mycobacterium tuberculosis. J. Exp. Med. 198, 987–997 (2003).
Tauszig-Delamasure, S., Bilak, H., Capovilla, M., Hoffmann, J. A. & Imler, J. L. Drosophila MyD88 is required for the response to fungal and Gram-positive bacterial infections. Nature Immunol. 3, 91–97 (2002).
Wells, C. A. et al. Genetic control of the innate immune response. BMC Immunol. 4, 5 (2003).
He, Y. W. et al. The extracellular matrix protein mindin is a pattern-recognition molecule for microbial pathogens. Nature Immunol. 5, 88–97 (2004).
Picard, C. et al. Pyogenic bacterial infections in humans with IRAK-4 deficiency. Science 299, 2076–2079 (2003).
Medvedev, A. E. et al. Distinct mutations in IRAK-4 confer hyporesponsiveness to lipopolysaccharide and interleukin-1 in a patient with recurrent bacterial infections. J. Exp. Med. 198, 521–531 (2003).
Kobayashi, K. S. & Flavell, R. A. Shielding the double-edged sword: negative regulation of the innate immune system. J. Leukoc. Biol. 75, 428–433 (2004).
Holland, S. M. et al. Treatment of refractory disseminated nontuberculous mycobacterial infection with interferon-γ. A preliminary report. N. Engl. J. Med. 330, 1348–1355 (1994).
Hengge, U. R. & Cusini, M. Topical immunomodulators for the treatment of external genital warts, cutaneous warts and molluscum contagiosum. Br. J. Dermatol. 149, S15–S19 (2003).
Persing, D. H. et al. Taking toll: lipid A mimetics as adjuvants and immunomodulators. Trends Microbiol. 10, S32–S37 (2002).
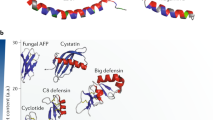
Ganz, T. Defensins: antimicrobial peptides of innate immunity. Nature Rev. Immunol. 3, 710–720 (2003).
Hancock, R. E. W. Cationic peptides: effectors in innate immunity and novel antimicrobials. Lancet Infect. Dis. 1, 156–164 (2001).
Zasloff. M. Antimicrobial peptides of multicellular organisms. Nature 415, 389–395 (2002).
Hancock, R. E. W. & Devine, D. (eds) in Mammalian Host Defence Peptides. (Cambridge Univ. Press, New York, in the press).
Maxwell, A. I., Morrison, G. M. & Dorin, J. R. Rapid sequence divergence in mammalian β-defensins by adaptive evolution. Mol. Immunol. 40, 413–421 (2003).
Lynn, D. J., Lloyd, A. T., Fares, M. A. & O'Farrelly, C. Evidence of positively selected sites in mammalian α-defensins. Mol. Biol. Evol. 21, 819–827 (2004).
Emes, R. D., Goodstadt, L., Winter, E. E. & Ponting, C. P. Comparison of the genomes of human and mouse lays the foundation of genome zoology. Hum. Mol. Genetics 12, 701–709 (2003).
Nizet, V. et al. Innate antimicrobial peptide protects the skin from invasive bacterial infection. Nature 414, 454–457 (2001).
Salzman, N. H., Ghosh, D., Huttner, K. M., Paterson, Y. & Bevins, C. L. Protection against enteric salmonellosis in transgenic mice expressing a human intestinal defensin. Nature 422, 522–526 (2003).
Moser, C. et al. β-defensin 1 contributes to pulmonary innate immunity in mice. Infect. Immun. 70, 3068–3072 (2002).
Rosenberger, C. M., Gallo, R. L. & Finlay, B. B. Interplay between antibacterial effectors: a macrophage antimicrobial peptide impairs intracellular Salmonella replication. Proc. Natl Acad. Sci. USA 101, 2422–2427 (2004).
Zhang, L. & Falla, T. J. Cationic antimicrobial peptides — an update. Expert Opin. Investig. Drugs 13, 1–9 (2004).
Scott, M. G., Davidson, D. J., Gold, M. R., Bowdish, D. & Hancock, R. E. W. The human antimicrobial peptide LL-37 is a multifunctional modulator of innate immune responses. J. Immunol. 169, 3883–3891 (2002).
Rustici, A. et al. Molecular mapping and detoxification of the lipid A binding site by synthetic peptides. Science 259, 361–365 (1993).
Gray, B. H. & Haseman, J. R. Bactericidal activity of synthetic peptides based on the structure of the 55-kilodalton bactericidal protein from human neutrophils. Infect. Immun. 62, 2732–2739 (1994).
Hirata, M., Shimomura, Y., Yoshida, M., Wright, S. C. & Larrick, J. W. Endotoxin-binding synthetic peptides with endotoxin-neutralizing, antibacterial and anticoagulant activities. Progress Clin. Biol. Res. 388, 147–159 (1994).
Battafaraono, R. J. et al. Peptide derivatives of three distinct lipopolysaccharide binding proteins inhibit lipopolysaccharide-induced tumor necrosis factor-α secretion in vitro. Surgery 118, 318–324 (1995).
Gough, M., Hancock, R. E. W. & Kelly, N. M. Anti-endotoxic potential of cationic peptide antimicrobials. Infect. Immun. 64, 4922–4927 (1996).
Chan, C. C., Tuaillon, N., Li, Q. & Shen, D. F. Therapeutic applications of antiflammin peptides in experimental ocular inflammation. Ann. NY Acad. Sci. 923, 141–146 (2000).
Tjabringa, G. S. et al. The antimicrobial peptide LL-37 activates innate immunity at the airway epithelial surface by transactivation of the epidermal growth factor receptor. J. Immunol. 171, 6690–6696 (2003).
Scott, M. G., Rosenberger, C. M., Gold, M. R., Finlay, B. B. & Hancock, R. E. W. An α-helical cationic antimicrobial peptide selectively modulates macrophage responses to lipopolysaccharide and directly alters macrophage gene expression. J. Immunol. 165, 3358–3365 (2000).
Hancock, R. E. W. & Diamond, G. The role of cationic antimicrobial peptides in innate host defences. Trends Microbiol. 8, 402–410 (2000).
Gudmundsson, G. H. & Agerberth, B. Neutrophil antibacterial peptides, multifunctional effector molecules in the mammalian immune system. J. Immunol. Methods 232, 45–54 (1999).
Otte, J. M., Kiehne, K. & Herzig, K. H. Antimicrobial peptides in innate immunity of the human intestine. J. Gastroenterol. 38, 717–726 (2003).
Gallo, R. L., Murakami, M., Ohtake, T. & Zaiou, M. Biology and clinical relevance of naturally occurring antimicrobial peptides. J. Allergy Clin. Immunol. 110, 823–831 (2002).
Biragyn, A. et al. Toll-like receptor 4-dependent activation of dendritic cells by β-defensin 2. Science 298, 1025–1029 (2002).
Davidson, D. J. et al. The cationic antimicrobial peptide LL-37 modulates dendritic cell differentiation and dendritic cell-induced T cell polarization. J. Immunol. 172, 1146–1156 (2004).
Bowdish, D. M. E., Davidson, D. J., Speert, D. P. & Hancock, R. E. W. The human cationic peptide LL-37 induces activation of the extracellular signal regulated kinase and p38 kinase pathways in primary human monocytes. J. Immunol. 172, 3758–3765 (2004).
Dong, C., Davis, R. J. & Flavell, R. A. MAP kinases in the immune response. Annu. Rev. Immunol. 20, 55–72 (2002).
De, Y. et al. LL-37, the neutrophil granule- and epithelial cell-derived cathelicidin, utilizes formyl peptide receptor-like 1 (FPRL1) as a receptor to chemoattract human peripheral blood neutrophils, monocytes, and T cells. J. Exp. Med. 192, 1069–1074 (2000).
Niyonsaba, F. et al. A cathelicidin family of human antibacterial peptide LL-37 induces mast cell chemotaxis. Immunology 106, 20–26 (2002).
Oppenheim, J. J., Biragyn, A., Kwak, L. W. & Yang, D. Roles of antimicrobial peptides such as defensins in innate and adaptive immunity. Ann. Rheum. Dis. 62, S17–S21 (2003).
van Wetering, S., Mannesse-Lazeroms, S. P., van Sterkenburg, M. A. & Hiemstra, P. S. Neutrophil defensins stimulate the release of cytokines by airway epithelial cells: modulation by dexamethasone. Inflamm. Res. 51, 8–15 (2002).
Yang, D. et al. Many chemokines including CCL20/MIP-3α display antimicrobial activity. J. Leukoc. Biol. 74, 448–455 (2003).
Dittmer, U. & Olbrich, A. R. Treatment of infectious diseases with immunostimulatory oligodeoxynucleotides containing CpG motifs. Curr. Opin. Microbiol. 6, 472–477 (2003).
Hemmi, H. et al. A Toll-like receptor recognizes bacterial DNA. Nature 408, 740–745 (2000).
Liu, L., Zhou, X., Shi, J., Xie, X. & Yuan, Z. Toll-like receptor-9 induced by physical trauma mediates release of cytokines following exposure to CpG motif in mouse skin. Immunology 110, 341–347 (2003).
Raghavan, S., Nystrom, J., Fredriksson, M., Holmgren, J. & Harandi, A. M. Orally administered CpG oligodeoxynucleotide induces production of CXC and CC chemokines in the gastric mucosa and suppresses bacterial colonization in a mouse model of Helicobacter pylori infection. Infect. Immun. 71, 7014–7022 (2003).
Verthelyi, D. & Klinman, D. M. Immunoregulatory activity of CpG oligonucleotides in humans and nonhuman primates. Clin. Immunol. 109, 64–71 (2003).
Zimmermann, S. et al. CpG oligodeoxynucleotides trigger protective and curative TH1 responses in lethal murine leishmaniasis. J. Immunol. 160, 3627–3630 (1998).
Dalpke, A., Zimmermann, S. & Heeg, K. CpG DNA in the prevention and treatment of infections. BioDrugs 16, 419–431 (2002).
Elkins, K. L., Rhinehart-Jones, T. R., Stibitz, S., Conover, J. S. & Klinman, D. M. Bacterial DNA containing CpG motifs stimulates lymphocyte-dependent protection of mice against lethal infection with intracellular bacteria. J. Immunol. 162, 2291–2298 (1999).
Lee, C. H., Jeong, H. D., Chung, J. K., Lee, H. H. & Kim, K. H. CpG motif in synthetic ODN primes respiratory burst of olive flounder Paralichthys olivaceus phagocytes and enhances protection against Edwardsiella tarda. Dis. Aquat. Organ. 56, 43–48 (2003).
Gomis, S. et al. Protection of chickens against Escherichia coli infections by DNA containing CpG motifs. Infect. Immun. 71, 857–863 (2003).
Rosenberger, C. M. & Finlay, B. B. Phagocyte sabotage: disruption of macrophage signalling by bacterial pathogens. Nature Rev. Mol. Cell Biol. 4, 385–396 (2003).
Hornef, M. W., Wick, M. J., Rhen, M. & Normark, S. Bacterial strategies for overcoming host innate and adaptive immune responses. Nature Immunol. 3, 1033–1040 (2002).
Hackett, C. J. Innate immune activation as a broad-spectrum biodefense strategy: prospects and research challenges. J. Allergy Clin. Immunol. 112, 686–694 (2003).
Acknowledgements
B.B.F. is a Howard Hughes Medical Institute (HHMI) International Research Scholar, a Canadian Institutes for Health Research (CIHR) Distinguished Investigator and the University of British Columbia Peter Wall Distinguished Professor. R.E.W.H. holds a Canada Research Chair. Operating grants from Genome Canada, with matching money from Inimex Pharmaceuticals, HHMI, CIHR, the Canadian Bacterial Diseases Network and the Canadian Cystic Fibrosis Foundation support work in their laboratories.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors are co-founders of Inimex Pharmaceuticals Inc.
Related links
Rights and permissions
About this article
Cite this article
Finlay, B., Hancock, R. Can innate immunity be enhanced to treat microbial infections?. Nat Rev Microbiol 2, 497–504 (2004). https://doi.org/10.1038/nrmicro908
Issue Date:
DOI: https://doi.org/10.1038/nrmicro908
This article is cited by
-
The gut–liver axis in sepsis: interaction mechanisms and therapeutic potential
Critical Care (2022)
-
CpG-ODN induced antimicrobial immunity in neonatal chicks involves a substantial shift in serum metabolic profiles
Scientific Reports (2021)
-
Machine learning-enabled predictive modeling to precisely identify the antimicrobial peptides
Medical & Biological Engineering & Computing (2021)
-
Evidence of multi-functional peptide activity: potential role of KT2 and RT2 for anti-inflammatory, anti-oxidative stress, and anti-apoptosis properties
Applied Biological Chemistry (2020)
-
Probiotics Modulate a Novel Amphibian Skin Defense Peptide That Is Antifungal and Facilitates Growth of Antifungal Bacteria
Microbial Ecology (2020)