Abstract
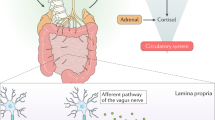
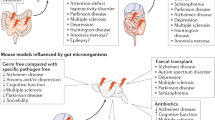
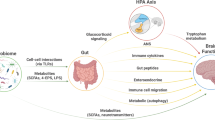
The intestinal microbiota consists of a vast bacterial community that resides primarily in the lower gut and lives in a symbiotic relationship with the host. A bidirectional neurohumoral communication system, known as the gut–brain axis, integrates the host gut and brain activities. Here, we describe the recent advances in our understanding of how the intestinal microbiota communicates with the brain via this axis to influence brain development and behaviour. We also review how this extended communication system might influence a broad spectrum of diseases, including irritable bowel syndrome, psychiatric disorders and demyelinating conditions such as multiple sclerosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Qin, J. et al. A human gut microbial gene catalogue established by metagenomic sequencing. Nature 464, 59–65 (2010).
Schiano, T. D. Treatment options for hepatic encephalopathy. Pharmacotherapy 30, S16–S21 (2010).
Wu, J. C. Psychological co-morbidity in functional gastrointestinal disorders: epidemiology, mechanisms and management. J. Neurogastroenterol. Motil. 18, 13–18 (2012).
Mayer, E. A. Gut feelings: the emerging biology of gut–brain communication. Nature Rev. Neurosci. 12, 453–466 (2011).
Tracey, K. J. Reflex control of immunity. Nature Rev. Immunol. 9, 418–428 (2009).
Mawdsley, J. E. & Rampton, D. S. Psychological stress in IBD: new insights into pathogenic and therapeutic implications. Gut 54, 1481–1491 (2005).
DuPont, A. W. & DuPont, H. L. The intestinal microbiota and chronic disorders of the gut. Nature Rev. Gastroenterol. Hepatol. 8, 523–531 (2011).
Collins, S. M. & Bercik, P. The relationship between intestinal microbiota and the central nervous system in normal gastrointestinal function and disease. Gastroenterology 136, 2003–2014 (2009).
Freestone, P. P., Sandrini, S. M., Haigh, R. D. & Lyte, M. Microbial endocrinology: how stress influences susceptibility to infection. Trends Microbiol. 16, 55–64 (2008).
Kaper, J. B. & Sperandio, V. Bacterial cell-to-cell signaling in the gastrointestinal tract. Infect. Immun. 73, 3197–3209 (2005).
Crawley, J. N. Behavioral phenotyping strategies for mutant mice. Neuron 57, 809–818 (2008).
Sudo, N. et al. Postnatal microbial colonization programs the hypothalamic–pituitary–adrenal system for stress response in mice. J. Physiol. 558, 263–275 (2004).
Neufeld, K. M., Kang, N., Bienenstock, J. & Foster, J. A. Reduced anxiety-like behavior and central neurochemical change in germ-free mice. Neurogastroenterol. Motil. 23, 255–264 (2011).
Heijtz, R. D. et al. Normal gut microbiota modulates brain development and behavior. Proc. Natl Acad. Sci. USA 108, 3047–3052 (2011).
Gareau, M. G. et al. Bacterial infection causes stress-induced memory dysfunction in mice. Gut 60, 307–317 (2011).
Costello, E. K. et al. Bacterial community variation in human body habitats across space and time. Science 326, 1694–1697 (2009).
Jalanka-Tuovinen, J. et al. Intestinal microbiota in healthy adults: temporal analysis reveals individual and common core and relation to intestinal symptoms. PLoS ONE 6, e23035 (2011).
Li, W., Dowd, S. E., Scurlock, B., Acosta-Martinez, V. & Lyte, M. Memory and learning behavior in mice is temporally associated with diet-induced alterations in gut bacteria. Physiol. Behav. 96, 557–567 (2009).
Bercik, P. et al. The intestinal microbiota affect central levels of brain-derived neurotropic factor and behavior in mice. Gastroenterology 141, 599–609 (2011).
Salonen, A., de Vos, W. M. & Palva, A. Gastrointestinal microbiota in irritable bowel syndrome: present state and perspectives. Microbiology 156, 3205–3215 (2010).
Abouesh, A., Stone, C. & Hobbs, W. R. Antimicrobial-induced mania (antibiomania): a review of spontaneous reports. J. Clin. Psychopharmacol. 22, 71–81 (2002).
Desbonnet, L., Garrett, L., Clarke, G., Bienenstock, J. & Dinan, T. G. The probiotic Bifidobacteria infantis: an assessment of potential antidepressant properties in the rat. J. Psychiatr. Res. 2, 164–174 (2008).
Desbonnet, L. et al. Effects of the probiotic Bifidobacterium infantis in the maternal separation model of depression. Neuroscience 170, 1179–1188 (2010).
Bercik, P. et al. Chronic gastrointestinal inflammation induces anxiety-like behavior and alters central nervous system biochemistry in mice. Gastroenterology 139, 2102–2112 (2010).
Bravo, J. A. et al. Ingestion of Lactobacillus strain regulates emotional behavior and central GABA receptor expression in a mouse via the vagus nerve. Proc. Natl Acad. Sci. USA 108, 10650–16055 (2011).
Bercik, P. et al. The anxiolytic effect of Bifidobacterium longum NCC3001 involves vagal pathways for gut–brain communication. Neurogastroenterol. Motil. 23, 1132–1139 (2011).
Kunze, W. A. et al. Lactobacillus reuteri enhances excitability of colonic AH neurons by inhibiting calcium-dependent potassium channel opening. J. Cell. Mol. Med. 13, 2261–2270 (2009).
Wikoff, W. R. et al. Metabolomics analysis reveals large effects of gut microflora on mammalian blood metabolites. Proc. Natl Acad. Sci. USA 106, 3698–3703 (2009).
Hanstock, T. L., Mallet, P. E. & Clayton, E. H. Increased plasma D-lactic acid associated with impaired memory in rats. Physiol. Behav. 101, 653–659 (2010).
Tana, C. et al. Altered profiles of intestinal microbiota and organic acids may be the origin of symptoms in irritable bowel syndrome. Neurogastroenterol. Motil. 22, 512–519 (2010).
Ledochowski, M. et al. Carbohydrate malabsorption syndromes and early signs of mental depression in females. Dig. Dis. Sci. 45, 1255–1259 (2000).
Myint, A. M. Kynurenines: from the perspective of major psychiatric disorders. FEBS J. 279, 1375–1385 (2012).
Barrett, E. et al. γ-Aminobuyric acid production by culturable bacteria from the human intestine. J. Appl. Microbiol. 113, 411–417 (2012).
Lyte, M. Probiotics function mechanistically as delivery vehicles for neuroactive compounds: microbial endocrinology in the design and use of probiotics. Bioessays 33, 574–581 (2011).
Macpherson, A. J. & Harris, N. L. Interactions between commensal intestinal bacteria and the immune system. Nature Rev. Immunol. 4, 478–485 (2004).
Salzman, N. H. Microbiota–immune system interaction: an uneasy alliance. Curr. Opin. Microbiol. 14, 99–105 (2011).
Derecki, N. C. et al. Regulation of learning and memory by meningeal immunity: a key role for IL-4. J. Exp. Med. 207, 1067–1080 (2010).
Lotrich, F. E., El-Gabalawy, H., Guenther, L. C. & Ware, C. F. The role of inflammation in the pathophysiology of depression: different treatments and their effects. J. Rheumatol. 88, 48–54 (2011).
O'Mahony, L. et al. Lactobacillus and bifidobacterium in irritable bowel syndrome: symptom responses and relationship to cytokine profiles. Gastroenterology 128, 541–551 (2005).
Honda, K. & Takeda, K. Regulatory mechanisms of immune responses to intestinal bacteria. Mucosal Immunol. 2, 187–196 (2009).
Uribe, A., Alam, M., Johansson, O., Midtvedt, T. & Theodorsson, E. Microflora modulates endocrine cells in the gastrointestinal mucosa of the rat. Gastroenterology 107, 1259–1269 (1994).
Bailey, M. T. et al. Exposure to a social stressor alters the structure of the intestinal microbiota: implications for stressor-induced immunomodulation. Brain Behav. Immun. 25, 397–407 (2011).
Lyte, M., Vulchanova, L. & Brown, D. R. Stress at the intestinal surface: catecholamines and mucosa–bacteria interactions. Cell Tissue Res. 2431, 23–32 (2011).
Bajaj, J. S. et al. Linkage of gut microbiome with cognition in hepatic encephalopathy. Am. J. Physiol. Gastrointest. Liver Physiol. 302, G168–G175 (2012).
Lee, Y. K., Menezes, J. S., Umesaki, Y. & Mazmanian, S. K. Proinflammatory T-cell responses to gut microbiota promote experimental autoimmune encephalomyelitis. Proc. Natl Acad. Sci. USA 108, 4615–4622 (2011).
Berer, K. et al. Commensal microbiota and myelin autoantigen cooperate to trigger autoimmune demyelination. Nature 479, 538–541 (2011).
Finegold, S. M., Downes, J. & Summanen, P. H. Microbiology of regressive autism. Anaerobe 18, 260–262 (2012).
Wang, L. et al. Low relative abundances of the mucolytic bacterium Akkermansia muciniphila and Bifidobacterium spp. in feces of children with autism. Appl. Environ. Microbiol. 77, 6718–6721 (2011).
Williams, B. L., Hornig, M., Parekh, T. & Lipkin, W. I. Application of novel PCR-based methods for detection, quantitation, and phylogenetic characterization of Sutterella species in intestinal biopsy samples from children with autism and gastrointestinal disturbances. MBio 3, e00261–e00211 (2012).
Williams, B. L. et al. Impaired carbohydrate digestion and transport and mucosal dysbiosis in the intestines of children with autism and gastrointestinal disturbances. PLoS ONE 6, e24585 (2011).
Sandler, R. H. et al. Short-term benefit from oral vancomycin treatment of regressive-onset autism. J. Child Neurol. 15, 429–435 (2000).
MacFabe, D. F. et al. Neurobiological effects of intraventricular propionic acid in rats: possible role of short chain fatty acids on the pathogenesis and characteristics of autism spectrum disorders. Behav. Brain. Res. 176, 149–169 (2007).
Rendeiro, C. P. et al. Flavonoids as modulators of memory and learning: molecular interactions resulting in behavioural effects. Proc. Nutr. Soc. 71, 246–262 (2012).
Juárez, I., Gratton, A. & Flores, G. Ontogeny of altered dendritic morphology in the rat prefrontal cortex, hippocampus, and nucleus accumbens following cesarean delivery and birth anoxia. J. Comp. Neurol. 507, 1734–1747 (2008).
Kim, H. R. et al. Delivery modes and neonatal EEG: spatial pattern analysis. Early Hum. Dev. 75, 35–53 (2003).
Acknowledgements
The authors' work cited in this paper was supported by the Canadian Institutes of Health Research (CIHR) and the Crohn's and Colitis Foundation of Canada (CCFC). Support to S.M.C. and P.B. was also received from the Nestle Research Center.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Stephen M. Collins and Premysl Bercik are the recipients of a grant from the Nestle Research Centre. Michael Surette declares no competing financial interests.
Related links
Related links
FURTHER INFORMATION
Farmcombe Family Digestive Health Research Institute, McMaster University
Glossary
- 5-hydroxytryptamine
-
(5-HT). A neurotransmitter that is produced mainly by the enteroendocrine cells of the gut, where it is an important regulator of gut physiology (particularly motility); however, 10% of 5-HT is found in the central nervous system, where it contributes to mood. 5-HT, also called serotonin, is derived from tryptophan via the formation of 5-hydroxytryptophan.
- Brain-derived neurotrophic factor
-
A protein that is widely distributed in the nervous system. In the brain, it is found in the hippocampus, amygdala and cortex. It contributes to a range of functions, including memory, mood and learning.
- Core microbiome
-
The 50–100 bacterial species that are common to the microbiomes of many individuals. Outside of these core species, there is considerable diversity in the microbiome components among healthy subjects.
- Dysbiosis
-
A compositional change in the microbiota and/or an abnormality in the interactions between the host and the commensal microbiota.
- Encephalopathy
-
An impairment of brain function, ranging in severity from mild confusion to deep coma. Severe liver disease is a common cause.
- GABA
-
(γ-aminobutyric acid). An inhibitory neurotransmitter that is found throughout the nervous system, including in the central and enteric nervous systems.
- Postsynaptic density protein 95
-
A component of the postsynaptic density, a lattice-like array of proteins that is crucial for synaptic function.
- Probiotic
-
According to the WHO: “Live microorganisms which when administered in adequate amounts confer a health benefit on the host.”
- Specific-pathogen-free mice
-
Laboratory mice that are free of defined pathogens.
- Synaptophysin
-
An integral membrane protein of small synaptic vesicles. Initially considered a synaptic marker, it is now thought to have several roles in synaptic function throughout the nervous system.
- Toll-like receptors
-
Transmembrane proteins that recognize highly conserved molecules of microbial origin and subsequently trigger activation of the innate immune system.
Rights and permissions
About this article
Cite this article
Collins, S., Surette, M. & Bercik, P. The interplay between the intestinal microbiota and the brain. Nat Rev Microbiol 10, 735–742 (2012). https://doi.org/10.1038/nrmicro2876
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrmicro2876
This article is cited by
-
Migraine and the microbiota. Can probiotics be beneficial in its prevention? - a narrative review
Pharmacological Reports (2024)
-
Altered gut microbiota in individuals with episodic and chronic migraine
Scientific Reports (2023)
-
The microbiome of the marine flatworm Macrostomum lignano provides fitness advantages and exhibits circadian rhythmicity
Communications Biology (2023)
-
Altered gut microbiota in patients with idiopathic Parkinson’s disease: an age–sex matched case–control study
Acta Neurologica Belgica (2023)
-
The critical role of gut-brain axis microbiome in mental disorders
Metabolic Brain Disease (2023)