Key Points
-
The field of viral-vector vaccines has expanded rapidly in recent years, following a much greater understanding of viral biology and the immune system. A wide range of vectors are now in advanced preclinical and clinical development for multiple human and veterinary applications, with targets ranging from certain types of cancer to a vast array of infectious diseases.
-
Vaccine vector development is now focusing on methods of rational attenuation and modification to enhance safety, immunogenicity and vaccine yield. A greater understanding of viral biology and immune evasion strategies, innate signalling pathways, antigen presentation and T cell and B cell biology is driving this process.
-
Pre-existing immunity in the human population to common viruses is driving the development of new or modified vectors for clinical use in humans, such as adenoviruses of rare human or simian serotypes. These vectors are being combined in varied heterologous prime–boost protocols to maximize cellular and humoral immunogenicity against target antigens for many difficult diseases.
-
The most successful applications of viral-vector vaccines have been in the veterinary field, with at least 12 viral-vector vaccines currently licensed for veterinary use and many more under development. Researchers in the fields of veterinary and human vaccine development are now working together to develop vaccines for closely related pathogens of animals and humans, such as tuberculosis and influenza.
-
Highly immunogenic vaccine candidates, mainly based on adenovirus and poxvirus vectors, are now in Phase I and Phase II clinical trials for malaria, tuberculosis and influenza. Similar approaches are being adopted in the field of HIV and cancer vaccines.
-
The development of such vaccines for use in humans faces substantial challenges, in both the scientific and the regulatory arenas. However, the licensure of replication-deficient viral-vector vaccines is wholly feasible, and the first chimeric flavivirus vaccine against Japanese encephalitis virus is currently pending regulatory approval.
Abstract
Recent developments in the use of viruses as vaccine vectors have been facilitated by a better understanding of viral biology. Advances occur as we gain greater insight into the interrelationship of viruses and the immune system. Viral-vector vaccines remain the best means to induce cellular immunity and are now showing promise for the induction of strong humoral responses. The potential benefits for global health that are offered by this field reflect the scope and utility of viruses as vaccine vectors for human and veterinary applications, with targets ranging from certain types of cancer to a vast array of infectious diseases.
Similar content being viewed by others
Main
Twenty-five years have elapsed since the first published use of a recombinant virus to deliver antigens from another infectious agent for the purposes of vaccination. Vaccinia virus (VACV) recombinant for the hepatitis B surface antigen (HBsAg) was shown to express HBsAg and, subsequently, to induce immunity in chimpanzees that was sufficient to protect against hepatitis B infection1,2. Despite this promising early start and substantial further development, the list of licensed viral-vector vaccines for human use is short, in part owing to the long, hard road to human licensure (for a review, see Ref. 3). Issues concerning the large-scale production and stability of such vaccines often require substantial scientific investment, and stringent safety requirements must be met for viruses that, in their natural state, have the potential to be human pathogens. This is especially true for those viral vectors that remain replication competent or that, by their nature, persist in the host beyond the inflammatory period. Indeed, in the simplest sense there remains a range of safety that increases with attenuation, which historically had to be balanced with the belief that reduced replicative capacity would result in reduced immunogenicity. However, despite these challenges, arguably the most important factor driving the continued development of viral-vector vaccines is their promising immunogenicity, especially with some of nature's most difficult antigens belonging to some of the world's toughest pathogens and cancers (Table 1).
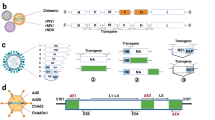
In most cases, a replicating viral infection is very effective at eliciting robust immune responses in a host that may last for several years. This is in contrast to many recombinant antigens that are delivered either as subunit DNA plasmids or proteins; although these are considered to be reasonably safe (dependent on the adjuvant), they have frequently suffered from poor immunogenicity. Conversely, viral vaccine vectors that replicate in vivo with a vigour that is similar to their wild-type parental viruses are often highly immunogenic, but also carry the risk of recombination, reactogenicity or reversion to virulence. The search for an optimal medium has driven renewed effort in the development of attenuated replication-defective viral vectors, a strategy aimed at maximizing immunogenicity and safety (Fig. 1).
Attenuated replication-defective viral vectors have been developed as part of a strategy to maximize immunogenicity and safety. They lie in the middle of a range of complexity and replication potential, between vaccine technologies at both extremes that have been licensed for human use. Although clinical vaccine development requires a long-term programme of work, the licensure of such vectored vaccines (if proved to be safe and effective) remains a wholly attainable and realistic goal. AAV, adeno-associated virus; AdV, adenovirus; AS01, adjuvant system 01 from GlaxoSmithKline; HPV, human papilloma virus; MF59, oil-in-water emulsion adjuvant from Novartis; MVA, modified vaccinia virus Ankara; VEE, Venezuelan equine encephalitis virus; VLP, virus like particle.
Attenuating viruses as vaccine vectors
One of the breakthroughs underpinning the viral vaccine field over the past decade has been the realization that replication competence and viral virulence do not always correlate with immunological robustness. For example, VACV and its safer, highly attenuated (replication-defective) cousins — modified vaccinia virus Ankara (MVA)4,5,6 and New York attenuated vaccinia virus (NYVAC)7 — have shown comparable and sometimes even better immunogenicity in animal studies8,9,10,11. Currently, a wide range of virus families are under intensive development as vaccine vectors for either human or veterinary use, including some that are replication competent but also many that are specifically attenuated. Table 1 outlines the main virus families that are under development as vectors and their associated diseases.
Immune evasion: implications for attenuation. Historically, the attenuation of viruses has been empirical, through continuous passage in transformed cell lines or in atypical hosts, and through delivery by unnatural routes. Many viruses are complex pathogens, and large DNA viruses such as poxviruses, adenoviruses (AdVs) and herpesviruses have devoted much of their genomes to genes that allow them to evade host immune responses. Many such viral genes encode proteins that affect specific immune defences, including those that act on early innate pathways such as pathways involving interferons12, pattern recognition receptors (Toll-like receptors; TLRs)13,14, chemokines15,16 and cytokines17, as well as those that act on subsequent adaptive responses by altering major histocompatibility complex processing of viral peptides18,19,20. To date, most processes of attenuation — for instance, through continuous in vitro passage in cell lines — are non-selective of the types of viral genes that are lost or become altered. Ultimately, viral variants have been selected for their lack of deleterious effects in small animals, such that they are no longer pathogenic and do the host little or no harm. However, viral genes that become lost or altered during the nonspecific attenuation process may not necessarily be the most optimal in terms of making a virus better for the immune presentation of recombinant inserts with a more potent adjuvant effect. Given that many viral genomes are now well characterized, future efforts to improve the adjuvant properties of viral vectors and to engineer them to induce different types of responses will be based on targeting specific viral genes that differentially affect the host's immune system. For example, knocking out viral genes that have evolved to reduce host antiviral immune responses could reduce immune evasion properties and improve adjuvant-specific characteristics of certain viral vectors; however, anti-vector responses may preclude the subsequent re-use of that vector in an individual.
Empirical versus rational attenuation. Attenuated poxviruses were first used during the smallpox eradication campaign and have since been further developed and exploited as vaccine vectors. One of these, MVA, was passaged more than 570 times in chick embryo fibroblasts, lost approximately 15% of its genome and was ultimately replication defective in mammalian cells4,21,22. Conversely, NYVAC was derived from the Copenhagen isolate of VACV by the deletion of 18 different ORFs through in vitro recombination, which caused replication deficiency7. Several recent studies have indicated that MVA maintains a strong immunostimulatory capacity, perhaps even greater than that of NYVAC23,24,25,26, whereas a recent primate study of HIV-1 vaccine candidates showed that these two vaccinia-derived vectors induced qualitatively different types of immune responses to the encoded Gag–Pol–Nef (group-specific antigen–polymerase–HIV-1 negative factor) and envelope (Env) antigens — NYVAC induced a CD4+ T cell-dominant response, whereas MVA induced a stronger CD8+ T cell response and accompanying CD4+ T cell responses27. The combined action of many viral proteins, some of which act on TLR signalling pathways and other antigen-presenting cell functions, probably accounts for these differences. Until there are better predictive assays for in vivo outcome, the process will remain largely empirical and dependant on the best in vivo models to assess the immunostimulatory properties of each vector. Nonetheless, in the case of empirically attenuated MVA, the rational deletion of genes can further enhance immunogenicity, with two reports demonstrating that the deletion of B15R, which encodes a soluble interleukin-1β (IL-1β) receptor, can increase the virus-specific memory CD8+ T cell response28,29. Similarly, by reducing vector-specific gene expression of a recombinant VACV, the CD8+ T cell response to the vector itself can be decreased and that against the transgene consequently enhanced30. Future efforts are set to continue along these lines, aiming to delete more genes, alone and in combination, in an attempt to rationally enhance the immunogenicity of these viral vectors.
Enhancing immunogenicity
Prime–boost regimes. In the 1990s, excitement surrounded the advent of DNA plasmid vaccines. However, it soon became apparent that DNA vaccines by themselves were insufficiently immunogenic to generate T cell responses of a magnitude sufficient to protect against difficult diseases in humans31,32. With advances in the quantitative T cell assays that are used in clinical trials, including the ex vivo interferon-γ ELISPOT (enzyme-linked immunosorbent spot) and cultured interferon-γ ELISPOT assays as well as multiparameter flow cytometry33,34, viral vectors resumed centre stage following the demonstration of their enhanced potency for cellular immune responses compared with other vaccine types35,36. However, unlike DNA vaccines, after one or two administrations in vivo, host responses to the structural proteins of the viral vector itself become self-limiting, leading to diminishing responses against the vaccine insert when the vector is used for multiple immunizations. The development of prime–boost protocols has been required to overcome these anti-vector responses while maximizing the host response to the vaccine insert.
Initially, heterologous prime–boost protocols with common vaccine inserts often used a DNA plasmid to prime the immune system; however, more recently interest has grown in the use of combinations of different recombinant viral vectors and in how their sequence of administration can influence the nature of the induced immune response. Multiple approaches have now been tested in both animal models and humans, including DNA–MVA, DNA–NYVAC, fowlpox virus (FPV)–MVA, influenza–MVA, AdV–MVA, heterologous AdV–AdV, and DNA–Sendai virus, targeting a wide range of diseases from malaria, HIV-1, tuberculosis (TB) and hepatitis C through to cancers (Table 2). A consistent observation throughout all of these studies is the differential ability of certain vectors to prime or boost responses. DNA vaccines, FPV and influenza virus are good priming vectors, whereas poxviruses (MVA and NYVAC) are consistently able to boost T cell responses that are primed by other means. A large body of data now indicates that, in general, recombinant AdV can prime and boost T cell and B cell responses remarkably well10,31,37,38,39,40,41,42,43,44,45,46. More recently, a combination of three unrelated vector modalities expressing the same vaccine insert has been used47,48. The assessment of these vectors has been aided by a greater in-depth analysis, in particular using multi-parameter flow cytometry34, which allows the dissection of the different T cell populations — both phenotypically and in terms of cytokine production and effector function — that are induced by such different vectors and regimes27,37,49,50,51,52,53. Correlating the protective outcomes of 'quality versus quantity' of the T cell responses that are induced by these different immunization regimes and vector strategies remains a challenge to the field.
Enhancing humoral immunity. More recent work has established the use of prime–boost immunization regimes to induce B cell as well as T cell responses, in particular AdV–MVA or heterologous AdV–AdV regimes39,40. The induction of both arms of the adaptive immune response is likely to be beneficial for protection against pathogens such as malaria parasites and many viruses, in contrast to mycobacteria, for which cellular immunity is probably the dominant protective mechanism. AdV vectors seem to be able to prime and boost B cell responses, in contrast to MVA vectors, which are far better able to boost the response than they are to prime it. This may be a result of prolonged high-level antigen expression following AdV immunization that is more beneficial for B cell priming, compared with a short burst of antigen from MVA that is more suited for B cell boosting54. Alternatively, studies on plasmacytoid dendritic cells have shown that a wave of cytokines, specifically type I interferon (IFN) and then IL-6, is essential for driving B cell differentiation into effector plasma cells in response to influenza virus infection55. Conversely, the priming of transgene-specific antibody responses by AdV vectors seems to negatively correlate with type I IFN production56,57, which is also associated with reduced transgene expression58. In the case of MVA, E3L confers resistance to type I IFN and is essential for sustained growth in chick embryo fibroblasts, and an E3L-knockout virus induces enhanced levels of chicken type I IFN and IL-6 in cell culture59,60. If B cell priming by MVA occurs in a similar manner to that induced by the influenza virus, the E3L-mediated immune-evasion mechanism could explain the weak B cell priming that is observed with this vector. Ultimately, a much better understanding of how cytokines can affect the induction of B cell responses by different virus families is essential to improve the rational design of vectors and prime–boost strategies that can be tailored to induce optimal antibody responses.
Circumventing pre-existing immunity. Viruses that commonly infect the human population bring with them the problem of pre-existing immunity. For certain viral vectors, such as those based on human AdVs and herpesviruses, this can be a major obstacle. For example, a candidate HIV-1 vaccine based on the human AdV serotype 5 (HAdV-5) vector was recently assessed in a Phase II trial that was conducted in individuals who were at high risk of HIV-1 infection. The trial was halted owing to lack of efficacy, but at the time a nonsignificant trend was observed towards increased acquisition of HIV-1 infection in those individuals with higher titres of pre-existing HAdV-5-neutralizing antibodies at the time of enrollment52,61. This issue provoked intense debate about the use of this commonly occurring human virus as a vaccine vector, but despite this setback for the HAdV-5 vector platform other strategies are actively being pursued to provide viable alternatives that can overcome pre-existing vector immunity62. These include modifying the vector to express heterologous adenovirus hexons63 or using human adenoviruses of rarer serotypes40,44,64,65 or those from other species such as chimpanzees66,67,68,69,70. Importantly, all such new vectors, especially those exogenous to a species, will need to clear the regulatory, safety and manufacturing hurdles.
Empirical versus rational design of vectored vaccines. With our expanding knowledge of immunology and viral biology, a new era of science in vaccinology has been established that has provided a future basis for the rational development of viral-vector vaccines. Optimizing subunit vaccine antigen design and understanding vector tropism, including target antigen processing and presentation in vivo, is crucial to this approach. In the case of VACV, it has been shown that the route of administration71, as well as the poxviral promoter used to drive transgene expression72, can influence the extent to which antigens are directly or cross-presented to T cells. Similarly, new data indicate that orthopoxvirus structural antigens, which are expressed late in infection and normally prevented from entering the direct presentation pathway owing to an abortive infection in antigen-presenting cells, are also specifically hidden in viral factories, thereby escaping the cross-presentation pathway and hampering the induction of poxvirus-specific CD8+ T cell responses73. This is not the case for foreign antigens driven by poxvirus early or late promoters, because these antigens are not usually targeted to such poxvirus factories. It is important that the consequences of such observations for vaccine-induced T cell responses against the vector and the transgene are better understood, and these data may begin to explain why poxvirus vectors such as MVA have been shown to be effective at re-boosting immune responses in humans 12 months after a previous immunization with the same vector74.
'Engineering' immunity. In contrast to the formidable complexities of the anti-vector immunity that is associated with persistent DNA viruses such as adenoviruses, engineered lentiviruses devoid of much of their structural antigens have been developed as vectors to deliver genes to specific cells in vivo for intracellular immunization75. This is made possible in part by the minimal expression of lentivirus-vector genes and in part because repeated in vivo administration (as in traditional immunization) is not the aim. Indeed, lentiviruses are currently being used to 'engineer immunity' for cancer and HIV-1 (Refs 76, 77). Transduction of haematopoietic stem cells with lentiviruses containing T cell receptor genes has now entered clinical testing for cancer immunotherapy. Substantial preclinical progress has also been made using these vectors to transduce human CD34+ cells with B cell receptor genes encoding neutralizing monoclonal antibodies against HIV-1 (Ref. 76). Strategies for targeting lentiviral vectors to specific cell types, such as skin dendritic cell (DC) subsets, have also been developed; for example, a mutated version of the Sindbis virus fusogenic protein that retains the ability to bind DC-SIGN (dendritic cell-specific ICAM3-grabbing non-integrin) proteins has been developed78. The development of further specific targeting technologies could greatly enhance the rational design of viral-vector vaccines that induce tailored and effective immune responses for the prevention of a wide range of infectious diseases and cancers.
Advances in veterinary vaccines
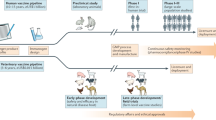
The most successful applications of viral-vector vaccines have undoubtedly been in the veterinary field. This is in contrast to the development of vaccines for human use, which entails more stringent regulatory requirements, step-wise clinical trials in human volunteers and a longer time to licensure and return on investment. At least 12 viral-vector vaccines are currently licensed for veterinary use (Table 3), and many more are under development (for a review, see Refs 79, 80). Nonetheless, these technologies still face an uphill struggle against competition from classical inactivated vaccines, which are often easier to produce. The challenges of controlling avian influenza in poultry are driven by economics, and require mass vaccination strategies such as administration in drinking water, by aerosol spray or by immunization in ovo81 (Fig. 2). Keeping the costs of these routes of administration low while maintaining vaccine efficacy poses considerable challenges for recombinant viral-vector technologies. One successful approach has been to use the new recombinant Newcastle disease virus (NDV), which provides a bivalent method for the control of two different avian pathogens at once by in ovo immunization82. New vaccine strategies for another important and highly contagious veterinary pathogen, foot-and-mouth disease virus (FMDV)83, are being explored using replication-defective HAdV-5. Protective immunity has been elicited in cattle and swine 7 days after a single immunization with recombinant HAdV-5 expressing the FMDV capsid and protease antigens84,85. Unlike for inactivated, whole-virus vaccines, the ability to distinguish infected from vaccinated animals (DIVA; see Box 1) using this vector vaccine platform, coupled with greatly increased biosafety, would make this effective technology highly desirable for FMDV-free countries. Similarly, with respect to important respiratory pathogens of livestock such as bovine tuberculosis, recombinant HAdV-5 or MVA vaccine vectors are being explored. These vectors, expressing the mycobacterial mycolyl transferase 85A antigen, have both been described as effective boosting agents for the Mycobacterium bovis bacille Calmette–Guérin (BCG) vaccine in cattle86,87,88. Challenge experiments are now required to assess protection with these BCG prime–vector boost regimes in herds of immunized cattle following direct exposure and also in a natural transmission setting. If more effective than the BCG vaccine alone, the use of vectored vaccines to boost BCG-primed responses could hold promise for the control of bovine TB, which is estimated to cost the world approximately US$3 billion per annum. Importantly, the cattle TB setting continues to provide useful immunological insight for the design and testing of candidate human TB vaccines89.
Potential advantages (+) and disadvantages (−) are shown for various possible routes of vaccination. a | Intramuscular immunization forms the mainstay approach to human vaccination, with minor exceptions such as Mycobacterium bovis bacille Calmette–Guérin (BCG) vaccine that are routinely given intradermally. Work is now progressing to develop needle-free transcutaneous devices such as patches coated with vaccine that can be self-applied in the absence of a trained individual — an obvious advantage for mass and emergency vaccination. Certain cancer vaccines may also be administered intravenously, but in general the safety concerns remain high for this route. Mucosal immunity may be enhanced in the gastrointestinal (GI) and respiratory tracts through the use of oral or aerosolized vaccines. This may enhance protective immunity to pathogens, for example against tuberculosis infection in the lungs or HIV infection in mucosal tissue. Recombinant adenovirus human serotype 41 (HAdV-41), a natural enteric pathogen, may prove to be a more suitable vector for the oral route, given that it has been reported to stimulate immune responses under acidic conditions127. However, there is still much work to be done in terms of demonstrating and maintaining vaccine safety, efficacy, stability and dosing before these routes of vaccination can be widely used in humans. Keeping the costs of using any of these less standard routes of administration in humans low while maintaining vaccine efficacy and, more importantly, safety poses considerable challenges for recombinant viral-vector technologies, and much proof-of-concept research is still required. b | Veterinary vaccines also make use of standard parenteral immunization routes (intradermal and intramuscular), but the use of alternative routes such as administration in drinking water, by aerosol spray or by immunization in ovo are driven by economics, less stringent safety requirements in animals and the frequent need for mass vaccination strategies.
Vaccines for human infectious disease
Viral-vector vaccines were originally developed to induce protective T cell responses against intracellular pathogens. However, to date neither a viral-vector vaccine nor a vaccine that acts directly by T cell-mediated immunity (with the possible exception of BCG) has been licensed for human use. This situation is compounded by the fact that these new approaches have been chosen for some of the most complex human pathogens for which conventional vaccine approaches have not been effective. These include Plasmodium falciparum malaria, Mycobacterium tuberculosis and HIV-1. Nonetheless, substantial progress has been made with recombinant viral-vaccine technologies. A list of vectors that are currently in development for human use is presented in Table 2. Impressive levels of T cell immunogenicity are now being reported for the first time in human clinical trials — a goal that, despite extensive efforts, has yet to be realized by DNA plasmid vaccine technologies alone. Key to this advance has been the development of AdV-vectored vaccines — AdV has a remarkable ability to prime immune responses against a transgene that can be boosted to high levels with a second vector that is recombinant for the same antigen, typically MVA or a second AdV of heterologous serotype. Similarly, both MVA and AdV can substantially boost immune responses primed by other means, for example the BCG vaccination. A comparison of these two widely used vectors is provided in Table 4.
Malaria vaccines. Recent progress has been made using a mixture of two HAdV-5 vectors expressing the pre-erythrocytic-stage malaria antigen circumsporozoite protein (CSP) or blood-stage malaria antigen apical membrane antigen 1 (AMA1)90. Following a single immunization in human volunteers, an impressive mean response to each antigen of 500–1,000 spot-forming units (SFU) per million peripheral blood mononuclear cells (PBMCs) was measured by ex vivo IFN-γ ELISPOT. Depletion studies defined the response as a mixed CD8+ and CD4+ T cell response, with the CD8+ phenotype three to five times as frequent as the CD4+ phenotype among IFN-γ-secreting responder cells, as measured by flow cytometry (M. Sedegah and T. Richie, personal communication). In an effort to circumvent the problem of pre-existing host responses to HAdV-5, recombinant adenoviruses of rare human serotypes or from non-human primates are also under development as malaria vaccine candidates. Priming and boosting with heterologous AdVs, such as HAdV-35 and HAdV-11, is being explored preclinically44 and, encouragingly, a recombinant chimpanzee adenovirus serotype 63 (AdCh63)70 expressing the pre-erythrocytic-stage antigen thrombospondin-related adhesion protein fused to a multi-epitope string (ME-TRAP) followed by a boost with MVA elicits mean levels of >1,000 SFU per million PBMCs against the malaria antigen in human volunteers (A. Hill, personal communication). Further preclinical studies have also reported the use of an AdV–MVA regime in mice to induce high-titre antibody responses and potent T cell responses. This specific HAdV-5–MVA regime, using vectors that are recombinant for the blood-stage malaria antigen merozoite surface protein 1 (MSP1), induced protective antibody responses against blood-stage infection39, as well as T cell responses that were partially effective against the preceding liver-stage infection91.
Heterologous poxvirus and DNA–poxvirus immunization regimes established the value of prime–boost approaches in humans, achieving much stronger T cell responses in clinical trials than single-vector approaches31,38,92. Nonetheless, efficacy was limited, and so Phase IIa malaria challenge studies of these new AdV-vector vaccines, scheduled for 2009–2010, will be important. Even if successful, these promising vaccine candidates will face further challenges, including the potential problems of high-level pre-existing immunity to the HAdV-5 vector in the human (especially African) population93. Furthermore, it remains to be seen how immunogenic AdV vectors will be in the target population of young African children. In children between 1 and 6 years of age, living in areas of high malaria transmission, reduced vaccine immunogenicity has been reported in comparison with naturally immune African adults and malaria-naive volunteers in developed countries94.
Tuberculosis vaccines. Similar to the situation for malaria, highly promising vectored-vaccine programmes are under way for human TB. These remain somewhat hindered in the absence of an experimental human challenge model, although efforts are under way to develop M. bovis BCG as a surrogate challenge for M. tuberculosis in humans86. Consequently, efficacy field trials remain essential once candidate vaccines have been selected — a process that is both long and expensive. A BCG prime and MVA–85A (an MVA vector containing the gene encoding the mycobacterial mycolyl transferase 85A antigen) boost regimen is now entering into a Phase IIb efficacy study in South Africa and has so far demonstrated high levels of T cell immunogenicity both in the UK and Africa, and an excellent safety record in over 500 people including adults, adolescents, infants, HIV-positive individuals and those who are latently infected with M. tuberculosis51,95,96,97. The TB vaccine field is also developing AdV-vector candidate vaccines and using similar approaches to circumvent human anti-HAdV-5 responses by using rare human serotypes such as HAdV-35. An HAdV-35 vaccine candidate expressing a fusion of three TB antigens, 85A, 85B and 10.4 (Ref. 98), can boost T cell responses that are primed by either BCG or a recombinant BCG in non-human primates99, establishing an approach that is being tested in humans100. A mixed CD8+ and CD4+ T cell response has been reported in Phase I studies in both the USA and South Africa137, but the contribution of CD8+ T cell responses to human immunity against TB remains unclear. For both leading viral-vector vaccine candidates, preclinical studies are now actively pursuing the potential for aerosolized rather than systemic delivery, possibly using nebulizers, to try to enhance the mucosal immunity that may be more protective against M. tuberculosis infection50 (Fig. 2). Further preclinical data from non-human primates are necessary if we are to address these outstanding immunological questions.
Flaviviruses. Yellow fever, a viral disease, is re-emerging in naive humans owing to a shift in its epidemiology. A highly effective attenuated vaccine (YFV-17D) has been used in humans for more than 70 years101. With the development of live attenuated flavivirus vectors based on YFV-17D, a programme of rational development of flavivirus vaccines has evolved102 in which the preM–Env genes, encoding the flavivirus pre-membrane and envelope proteins, have been replaced with those from West Nile virus, Japanese encephalitis virus or dengue virus to generate chimeric vectors. It has been far more difficult to stably express non-flavivirus antigens in these vectors, but insertion of non-flavivirus epitopes has been reported103, as has the stable insertion of GFP104. Following extensive and successful safety and immunogenicity testing in Phase I–III trials, the chimeric flavivirus for Japanese encephalitis may well be the first viral-vector vaccine to be licensed for use in humans105.
Influenza virus. The development of a cross-strain influenza vaccine would be a major and long-sought achievement, especially given the continued seasonal emergence of new influenza variants, the current H1N1 influenza A virus pandemic and the possibility of an outbreak of highly pathogenic avian influenza. The induction of protective T cell responses against conserved internal influenza antigens, rather than variant-specific antibody responses against surface haemagglutinin and neuraminidase antigens, is one approach that may stand a chance of success. Phase I trials of a new candidate cross-strain influenza vaccine are now under way, using an MVA vector that expresses the highly conserved internal nucleoprotein and matrix 1 (NP–M1) influenza A antigens. This vector can boost T cell responses in individuals who are presumably primed by natural exposure (S. Gilbert, personal communication). If these T cell responses are associated with protection, as some evidence suggests106, then a viral-vector vaccine approach could prove highly cost-effective in comparison with seasonal influenza immunizations aimed at inducing antibodies against the variant-specific surface coat antigens.
HIV. Perhaps a greater challenge for vaccine development than the seasonal variability and antigenic drift of influenza virus is the constantly evolving HIV epidemic. T cell-inducing vaccines for HIV-1 aim to reduce viral load and CD4+ T cell loss, prolong survival and reduce transmission107. Renewed efforts are focused on poxvirus regimes using attenuated canarypox virus (ALVAC), MVA or NYVAC. Each of these poxvirus vaccine vectors has been developed and evaluated for their ability to induce HIV-1-specific immune responses in humans. The most recent Phase III HIV-1 clinical trial used a combination of ALVAC and AIDSVAX (monomeric HIV-1 glycoprotein 120) in a large, community-based population in which the risk of transmission is based on heterosexual life style. Although the study found an encouraging trend towards prevention of HIV-1 infection (31.2% efficacy), there was no benefit for post-infection virus load or CD4+ T cell counts108. In preclinical efficacy studies in non-human primates, control of virus load and preservation of CD4+ T cell counts have been reported when certain pox-based vectors have been used in heterologous prime–boost combinations to boost DNA-primed responses41,48. They are able to induce antibody responses against Env and elicit T cell responses of varying levels. In humans, T cell responses are of a mixed CD4+ and CD8+ phenotype and are broadly multifunctional in terms of cytokine production37,109,110. Nonetheless, the consensus remains that these vaccine regimes need to be improved to increase the magnitude and breadth of the CD8+ and CD4+ T cell responses against antigens such as Gag and Pol, and to induce high-titre neutralizing antibodies or high-avidity antibodies against the native trimeric Env protein.
The development of HAdV-5 recombinants for HIV-1 vaccination produced the most immunogenic candidate vaccine to date, which was taken forward into a human efficacy trial, designated STEP. The HAdV-5 recombinants remained a leading approach for HIV-1 vaccine developers until the STEP trial was terminated on the grounds of futility52,61,111. Even though much stronger T cell responses have been elicited in malaria and TB prime–boost vaccine programmes in humans, the failure of the STEP trial was suggested by some as grounds for dismissal of the concept of vaccines aimed at inducing T cell-mediated immunity. Encouragingly, however, after some initial despondency, discussion has refocused on the further optimization of vaccine immunogens and technologies62. Indeed, now that far higher T cell induction is being reported for non-HIV infections, it is time to match these in the HIV vaccine field. Vaccine developers for HIV-1 have already begun to focus on other, less established, chimeric viral-vector technologies. Sendai virus, cytomegalovirus (CMV), reovirus, herpes simplex virus and NDV are currently under evaluation by a range of groups, including the International AIDS Vaccine Initiative (IAVI). The recent use of CMV as a vector has shown that this vector has substantial efficacy in the rigorous SIVMAC (simian immunodeficiency virus of the macaque) rhesus macaque challenge model, exceeding the suppression of virus load that was observed with another study using HAdV-5 (Refs 112, 113). Alternative strategies will probably combine the most immunogenic prime–boost regimes that are currently available, such as AdV–MVA or heterologous AdV–AdV regimes or even triple combinations, to facilitate the recruitment of both T cell and B cell responses41,114. A recombinant HAdV-26–HAdV-5 regime has recently been described that can induce a T cell response against SIVMAC Gag in rhesus macaques that is stronger, more polyfunctional and more protective than the response induced by a homologous HAdV-5 regime40. Indeed, a mean of >2,000 SFU per million PBMCs was achieved in these macaques40, in comparison with around 300 SFU per million PBMCs on average in volunteers in the HAdV-5 homologous prime–boost STEP trial52. Future studies will reveal whether these approaches can consistently improve on the immunological and viral control benchmarks that were set by the combination of DNA and HAdV-5 vaccine vectors in the SIVMAC rhesus macaque challenge model.
Cancer vaccines. The cancer vaccine field (for comprehensive reviews, see Refs 115, 116) has not only suffered the problem of inducing strong, durable, effective immune responses (an issue shared with many infectious-disease vaccines), but has also tried to do so in the face of the unique and complex challenge of breaking tolerance against self-antigens — which are, by definition, either weakly immunogenic or functionally non-immunogenic in an immunologically compromised environment. Intriguingly, there seems to be hope for certain cancer vaccines in specific settings. The use of therapeutic viral-vector vaccines targeting the carcinoembryonic antigen (CEA), in combination with local radiation, chemotherapy or cytotoxic T lymphocyte protein 4 (CTLA4)-specific monoclonal antibodies, has led to improved survival in some patients117. In this setting, an 'antigen cascade' has been observed, whereby T cell responses to the CEA were noted not only post-vaccination but also as de novo responses to other tumour-associated antigens (TAAs), which were possibly induced following more effective immune targeting of cancerous cells. MVA vectors expressing TAAs are also currently in advanced clinical trials as an addition to first-line therapies for renal, prostate and non-small-cell lung cancers118,119,120. Recent immunological analyses have indicated that there is a significant correlation between the level of activated natural killer cells at baseline and the subsequent efficacy in patients receiving chemotherapy plus MVA–MUC1–IL-2 for lung cancer121. The level of activated natural killer cells could potentially be used as a predictive biomarker for patients who might benefit from viral-vector immunization combination therapy. As with immune-compromising viral infections, the use of cancer vaccines in a therapeutic setting will require the re-establishment of a functional and robust immune response that can be sustained in a potentially tolerogenic setting — a setting induced as part of the immune evasion strategy of some cancers. In this light, combination therapies hold the greatest hope in the cancer vaccine field, although preclinical data from mouse122 and non-human primate123 studies indicate that AdV vectors expressing candidate TAAs can break tolerance. It remains to be seen whether candidate cancer vaccines based on AdV vectors can achieve even better immunogenicity in human trials than that observed with poxvirus-based immunotherapies.
Future directions
Most viruses have evolved mechanisms to evade host immune responses and to modulate the subsequent inflammatory environment in different ways. These evasive attributes are contrary to the immunostimulatory properties of ideal vaccine adjuvants. However, in the new era of genomics, specific viral genes encoding factors with immune-evasive properties are being targeted to enhance these immunostimulatory properties. Not only is this new science in vaccinology using well-established viral vectors, but as new viruses are discovered and characterized their utility as vaccine vectors is also being explored. For instance, investigations are under way using multiple strategies to find alternative vectors to circumvent the problem of pre-existing vector immunity. These vectors, combined with new vaccine prime–boost regimes that will induce stronger, broader and more durable effector responses, offer promising solutions to immunological problems. The emergence of many new vector systems must not distract from the great opportunities to improve current vectors. Approaches that knock out viral genes and result in improved antigen presentation, improved adjuvant effects and intentional skewing of immune responses are important new lines of investigation. New technologies include the easier manipulation of poxvirus genomes by 'recombineering' in bacterial artificial chromosomes28,124, and high-throughput transgene insertion into AdV vectors, including the use of bacterial tet operator elements in the transgene promoter to shut down the transcription of transgenes that may be toxic or deleterious to viral growth125. The use of such technologies will be adopted for a range of different vector platforms, therefore allowing the screening of novel viral vaccine vectors that remain safe but show improved immunogenicity for the recombinant vaccine antigens that they deliver.
The development of viral-vector vaccines for use in animals and humans faces substantial challenges, both in the scientific and regulatory arenas. However, these technologies are finally achieving the efficacy in animals and strong immune responses in humans that have been sought after for so long, in particular in the fields of malaria, TB and influenza. The translation of these new technologies will facilitate vaccine development against some of the toughest challenges in the field, such as cancer and HIV-1. The licensure of replication-deficient viral-vector vaccines is feasible, given that they lie between the two extremes of protein-in-adjuvant vaccines and live viral vaccines — both of which, after considerable effort, have been licensed before. With the first chimeric flavivirus vaccine against Japanese encephalitis virus pending regulatory approval, the road is paved for many other viral-vector vaccines in advanced clinical development. The next 25 years will prove a challenging but exciting time for this renaissance in vaccine science.
References
Smith, G. L., Mackett, M. & Moss, B. Infectious vaccinia virus recombinants that express hepatitis B virus surface antigen. Nature 302, 490–495 (1983).
Moss, B., Smith, G. L., Gerin, J. L. & Purcell, R. H. Live recombinant vaccinia virus protects chimpanzees against hepatitis B. Nature 311, 67–69 (1984). Along with reference 1, this is the first major study to use recombinant poxvirus as a potential vaccine for another infectious disease.
Peter, G. et al. Lessons learned from a review of the development of selected vaccines. National Vaccine Advisory Committee. Pediatrics 104, 942–950 (1999).
Mayr, A., Hochstein-Mintzel, V. & Stickl, H. Passage history, properties, and applicability of the attenuated vaccinia virus strain, MVA. Infection 3, 6–14 (1975).
Sutter, G. & Moss, B. Nonreplicating vaccinia vector efficiently expresses recombinant genes. Proc. Natl Acad. Sci. USA 89, 10847–10851 (1992).
Sutter, G., Wyatt, L. S., Foley, P. L., Bennink, J. R. & Moss, B. A recombinant vector derived from the host range-restricted and highly attenuated MVA strain of vaccinia virus stimulates protective immunity in mice to influenza virus. Vaccine 12, 1032–1040 (1994).
Tartaglia, J. et al. NYVAC: a highly attenuated strain of vaccinia virus. Virology 188, 217–232 (1992).
Ramirez, J. C., Gherardi, M. M. & Esteban, M. Biology of attenuated modified vaccinia virus Ankara recombinant vector in mice: virus fate and activation of B- and T-cell immune responses in comparison with the Western Reserve strain and advantages as a vaccine. J. Virol. 74, 923–933 (2000).
Hodge, J. W. et al. Modified vaccinia virus ankara recombinants are as potent as vaccinia recombinants in diversified prime and boost vaccine regimens to elicit therapeutic antitumor responses. Cancer Res. 63, 7942–7949 (2003).
Schneider, J. et al. Enhanced immunogenicity for CD8+ T cell induction and complete protective efficacy of malaria DNA vaccination by boosting with modified vaccinia virus Ankara. Nature Med. 4, 397–402 (1998).
Hirsch, V. M. et al. Patterns of viral replication correlate with outcome in simian immunodeficiency virus (SIV)-infected macaques: effect of prior immunization with a trivalent SIV vaccine in modified vaccinia virus Ankara. J. Virol. 70, 3741–3752 (1996).
Mann, B. A. et al. Vaccinia virus blocks Stat1-dependent and Stat1-independent gene expression induced by type I and type II interferons. J. Interferon Cytokine Res. 28, 367–380 (2008).
Stack, J. et al. Vaccinia virus protein A46R targets multiple Toll-like-interleukin-1 receptor adaptors and contributes to virulence. J. Exp. Med. 201, 1007–1018 (2005).
Bowie, A. G. & Unterholzner, L. Viral evasion and subversion of pattern-recognition receptor signalling. Nature Rev. Immunol. 8, 911–922 (2008). This article highlights the interactions between viruses and pattern-recognition receptors.
Alejo, A. et al. A chemokine-binding domain in the tumor necrosis factor receptor from variola (smallpox) virus. Proc. Natl Acad. Sci. USA 103, 5995–6000 (2006).
Alcami, A. New insights into the subversion of the chemokine system by poxviruses. Eur. J. Immunol. 37, 880–883 (2007).
Alcami, A. & Smith, G. L. A soluble receptor for interleukin-1β encoded by vaccinia virus: a novel mechanism of virus modulation of the host response to infection. Cell 71, 153–167 (1992).
Blair, G. E. & Blair-Zajdel, M. E. Evasion of the immune system by adenoviruses. Curr. Top. Microbiol. Immunol. 273, 3–28 (2004).
Dasgupta, A., Hammarlund, E., Slifka, M. K. & Fruh, K. Cowpox virus evades CTL recognition and inhibits the intracellular transport of MHC class I molecules. J. Immunol. 178, 1654–1661 (2007).
Antoniou, A. N. & Powis, S. J. Pathogen evasion strategies for the major histocompatibility complex class I assembly pathway. Immunology 124, 1–12 (2008).
Meyer, H., Sutter, G. & Mayr, A. Mapping of deletions in the genome of the highly attenuated vaccinia virus MVA and their influence on virulence. J. Gen. Virol. 72, 1031–1038 (1991).
Blanchard, T. J., Alcami, A., Andrea, P. & Smith, G. L. Modified vaccinia virus Ankara undergoes limited replication in human cells and lacks several immunomodulatory proteins: implications for use as a human vaccine. J. Gen. Virol. 79, 1159–1167 (1998).
Lehmann, M. H. et al. Modified vaccinia virus ankara triggers chemotaxis of monocytes and early respiratory immigration of leukocytes by induction of CCL2 expression. J. Virol. 83, 2540–2552 (2009).
Waibler, Z. et al. Vaccinia virus-mediated inhibition of type I interferon responses is a multifactorial process involving the soluble type I interferon receptor B18 and intracellular components. J. Virol. 83, 1563–1571 (2009).
Midgley, C. M., Putz, M. M., Weber, J. N. & Smith, G. L. Vaccinia virus strain NYVAC induces substantially lower and qualitatively different human antibody responses compared with strains Lister and Dryvax. J. Gen. Virol. 89, 2992–2997 (2008).
Delaloye, J. et al. Innate immune sensing of modified vaccinia virus Ankara (MVA) is mediated by TLR2-TLR6, MDA-5 and the NALP3 inflammasome. PLoS Pathog. 5, e1000480 (2009). This study identifies innate immune signalling pathways that are activated by MVA.
Mooij, P. et al. Differential CD4+ versus CD8+ T-cell responses elicited by different poxvirus-based human immunodeficiency virus type 1 vaccine candidates provide comparable efficacies in primates. J. Virol. 82, 2975–2988 (2008). This work uses different viral vectors to show that there is differential modulation of the vaccine-induced CD4+ and CD8+ T cell responses.
Cottingham, M. G. . et al. Recombination-mediated genetic engineering of a bacterial artificial chromosome clone of modified vaccinia virus Ankara (MVA). PLoS ONE 3, e1638 (2008).
Staib, C., Kisling, S., Erfle, V. & Sutter, G. Inactivation of the viral interleukin 1β receptor improves CD8+ T-cell memory responses elicited upon immunization with modified vaccinia virus Ankara. J. Gen. Virol. 86, 1997–2006 (2005).
Fischer, M. A., Tscharke, D. C., Donohue, K. B., Truckenmiller, M. E. & Norbury, C. C. Reduction of vector gene expression increases foreign antigen-specific CD8+ T-cell priming. J. Gen. Virol. 88, 2378–2386 (2007).
McConkey, S. J. et al. Enhanced T-cell immunogenicity of plasmid DNA vaccines boosted by recombinant modified vaccinia virus Ankara in humans. Nature Med. 9, 729–735 (2003).
Mwau, M. et al. A human immunodeficiency virus 1 (HIV-1) clade A vaccine in clinical trials: stimulation of HIV-specific T-cell responses by DNA and recombinant modified vaccinia virus Ankara (MVA) vaccines in humans. J. Gen. Virol. 85, 911–919 (2004).
Keating, S. M. et al. Durable human memory T cells quantifiable by cultured enzyme-linked immunospot assays are induced by heterologous prime boost immunization and correlate with protection against malaria. J. Immunol. 175, 5675–5680 (2005).
Lamoreaux, L., Roederer, M. & Koup, R. Intracellular cytokine optimization and standard operating procedure. Nature Protoc. 1, 1507–1516 (2006).
Vuola, J. M. et al. Differential immunogenicity of various heterologous prime–boost vaccine regimens using DNA and viral vectors in healthy volunteers. J. Immunol. 174, 449–455 (2005).
Mooij, P. et al. Comparison of human and rhesus macaque T-cell responses elicited by boosting with NYVAC encoding HIV-1 clade C immunogens. J. Virol. 83, 5881–5889 (2009).
Harari, A. et al. An HIV-1 clade C DNA prime, NYVAC boost vaccine regimen induces reliable, polyfunctional, and long-lasting T cell responses. J. Exp. Med. 205, 63–77 (2008).
Webster, D. P. et al. Enhanced T cell-mediated protection against malaria in human challenges by using the recombinant poxviruses FP9 and modified vaccinia virus Ankara. Proc. Natl Acad. Sci. USA 102, 4836–4841 (2005). This paper illustrates the effect of heterologous prime–boost protocols in humans using viral-vector vaccines that lead to enhanced immunogenicity and protection against malaria infection.
Draper, S. J. et al. Effective induction of high-titer antibodies by viral vector vaccines. Nature Med. 14, 819–821 (2008). This article highlights the ability of certain viral-vector vaccine combinations to elicite antibody as well as T cell responses.
Liu, J. et al. Immune control of an SIV challenge by a T-cell-based vaccine in rhesus monkeys. Nature 457, 87–91 (2009). This study describes the increased efficacy of heterologous adenoviral prime–boost regimes over homologous regimes for eliciting protection against a stringent SIV challenge in macaques, which suggests that such an approach should be adopted for HIV-1 vaccine development.
Shiver, J. W. et al. Replication-incompetent adenoviral vaccine vector elicits effective anti-immunodeficiency-virus immunity. Nature 415, 331–335 (2002).
Gherardi, M. M. et al. Prime–boost immunization schedules based on influenza virus and vaccinia virus vectors potentiate cellular immune responses against human immunodeficiency virus Env protein systemically and in the genitorectal draining lymph nodes. J. Virol. 77, 7048–7057 (2003).
Yu, S. et al. Potent specific immune responses induced by prime–boost–boost strategies based on DNA, adenovirus, and Sendai virus vectors expressing gag gene of Chinese HIV-1 subtype, B. Vaccine 26, 6124–6131 (2008).
Lemckert, A. A. et al. Immunogenicity of heterologous prime–boost regimens involving recombinant adenovirus serotype 11 (Ad11) and Ad35 vaccine vectors in the presence of anti-Ad5 immunity. J. Virol. 79, 9694–9701 (2005).
Folgori, A. et al. A T-cell HCV vaccine eliciting effective immunity against heterologous virus challenge in chimpanzees. Nature Med. 12, 190–197 (2006).
Fattori, E. et al. Efficient immunization of rhesus macaques with an HCV candidate vaccine by heterologous priming–boosting with novel adenoviral vectors based on different serotypes. Gene Ther. 13, 1088–1096 (2006).
Heeney, J. L. et al. A vaccine strategy utilizing a combination of three different chimeric vectors which share specific vaccine antigens. J. Med. Primatol. 29, 268–273 (2000).
Koopman, G. et al. Vaccine protection from CD4+ T-cell loss caused by simian immunodeficiency virus (SIV) mac251 is afforded by sequential immunization with three unrelated vaccine vectors encoding multiple SIV antigens. J. Gen. Virol. 85, 2915–2924 (2004). A report on the efficacy of a combination of three different vectors to elicit vaccine protection against SIV in macaques.
Darrah, P. A. et al. Multifunctional TH1 cells define a correlate of vaccine-mediated protection against Leishmania major. Nature Med. 13, 843–850 (2007). This work highlights the importance of measuring antigen-specific T cell quality and not just quantity when analysing immune responses for correlates of protection.
Forbes, E. K. et al. Multifunctional, high-level cytokine-producing Th1 cells in the lung, but not spleen, correlate with protection against Mycobacterium tuberculosis aerosol challenge in mice. J. Immunol. 181, 4955–4964 (2008).
Beveridge, N. E. et al. Immunisation with BCG and recombinant MVA85A induces long-lasting, polyfunctional Mycobacterium tuberculosis-specific CD4+ memory T lymphocyte populations. Eur. J. Immunol. 37, 3089–3100 (2007).
McElrath, M.J. et al. HIV-1 vaccine-induced immunity in the test-of-concept Step Study: a case-cohort analysis. Lancet 372, 1894–1905 (2008).
Liu, J . et al. Magnitude and phenotype of cellular immune responses elicited by recombinant adenovirus vectors and heterologous prime–boost regimens in rhesus monkeys. J. Virol. 82, 4844–4852 (2008).
Geiben-Lynn, R., Greenland, J. R., Frimpong-Boateng, K. & Letvin, N. L. Kinetics of recombinant adenovirus type 5, vaccinia virus, modified vaccinia ankara virus, and DNA antigen expression in vivo and the induction of memory T-lymphocyte responses. Clin. Vaccine Immunol. 15, 691–696 (2008).
Jego, G. et al. Plasmacytoid dendritic cells induce plasma cell differentiation through type I interferon and interleukin 6. Immunity 19, 225–234 (2003).
Xiang, Z. et al. Novel, chimpanzee serotype 68-based adenoviral vaccine carrier for induction of antibodies to a transgene product. J. Virol. 76, 2667–2675 (2002).
Hensley, S. E., Giles-Davis, W., McCoy, K. C., Weninger, W. & Ertl, H. C. Dendritic cell maturation, but not CD8+ T cell induction, is dependent on type I IFN signaling during vaccination with adenovirus vectors. J. Immunol. 175, 6032–6041 (2005).
Hensley, S. E. et al. Type I interferon inhibits antibody responses induced by a chimpanzee adenovirus vector. Mol. Ther. 15, 393–403 (2007).
Hornemann, S. et al. Replication of modified vaccinia virus Ankara in primary chicken embryo fibroblasts requires expression of the interferon resistance gene E3L. J. Virol. 77, 8394–8407 (2003).
Ludwig, H. et al. Role of viral factor E3L in modified vaccinia virus Ankara infection of human HeLa cells: regulation of the virus life cycle and identification of differentially expressed host genes. J. Virol. 79, 2584–2596 (2005).
Buchbinder, S. P. et al. Efficacy assessment of a cell-mediated immunity HIV-1 vaccine (the Step Study): a double-blind, randomised, placebo-controlled, test-of-concept trial. Lancet 372, 1881–1893 (2008). Along with reference 52, this paper reports on the first Phase III clinical trial to use a T cell-based adenoviral vaccine vector in humans, and warns of the concerns of anti-HAdV-5 vector immunity.
Bradac, J. & Dieffenbach, C. W. HIV vaccine development: Lessons from the past, informing the future. IDrugs 12, 435–439 (2009).
Roberts, D. M. et al. Hexon-chimaeric adenovirus serotype 5 vectors circumvent pre-existing anti-vector immunity. Nature 441, 239–243 (2006). This study demonstrates that modification of viral surface proteins can increase the efficacy of HAdV-5-based vectors.
Tatsis, N. & Ertl, H. C. Adenoviruses as vaccine vectors. Mol. Ther. 10, 616–629 (2004).
Harro, C. et al. Safety and immunogenicity of the MRKAd5 and MRKAd6 HIV-1 trigene vaccines alone and in combination in healthy adults. Clin. Vaccine Immunol. 16, 1285–1292 (2009).
Tatsis, N. et al. Chimpanzee-origin adenovirus vectors as vaccine carriers. Gene Ther. 13, 421–429 (2006).
Sridhar, S. et al. Single-dose protection against Plasmodium berghei by a simian adenovirus vector using a human cytomegalovirus promoter containing intron, A. J. Virol. 82, 3822–3833 (2008).
Farina, S. F. et al. Replication-defective vector based on a chimpanzee adenovirus. J. Virol. 75, 11603–11613 (2001).
Reyes-Sandoval, A. et al. Single-dose immunogenicity and protective efficacy of simian adenoviral vectors against Plasmodium berghei. Eur. J. Immunol. 38, 732–741 (2008).
Dudareva, M. et al. Prevalence of serum neutralizing antibodies against chimpanzee adenovirus 63 and human adenovirus 5 in Kenyan children, in the context of vaccine vector efficacy. Vaccine 27, 3501–3504 (2009).
Shen, X., Wong, S. B., Buck, C. B., Zhang, J. & Siliciano, R. F. Direct priming and cross-priming contribute differentially to the induction of CD8+ CTL following exposure to vaccinia virus via different routes. J. Immunol. 169, 4222–4229 (2002).
Bronte, V. et al. Antigen expression by dendritic cells correlates with the therapeutic effectiveness of a model recombinant poxvirus tumor vaccine. Proc. Natl Acad. Sci. USA 94, 3183–3188 (1997).
Tewalt, E. F. et al. Viral sequestration of antigen subverts cross presentation to CD8+ T cells. PLoS Pathog. 5, e1000457 (2009).
Moorthy, V.S. et al. Phase 1 evaluation of 3 highly immunogenic prime–boost regimens, including a 12-month reboosting vaccination, for malaria vaccination in Gambian men. J. Infect. Dis. 189, 2213–2219 (2004).
Yang, L., Bailey, L., Baltimore, D. & Wang, P. Targeting lentiviral vectors to specific cell types in vivo. Proc. Natl Acad. Sci. USA 103, 11479–11484 (2006).
Luo, X. M. et al. Engineering human hematopoietic stem/progenitor cells to produce a broadly neutralizing anti-HIV antibody after in vitro maturation to human B lymphocytes. Blood 113, 1422–1431 (2008). This article describes the basis of engineering immunity with lentiviruses. These mediate gene transfer into human hematopoietic stem cells, which can then be differentiated in vitro into B cells that secrete neutralizing HIV-1 antibodies.
Garcia Casado, J. et al. Lentivector immunization induces tumor antigen-specific B and T cell responses in vivo. Eur. J. Immunol. 38, 1867–1876 (2008).
Yang, L. et al. Engineered lentivector targeting of dendritic cells for in vivo immunization. Nature Biotech. 26, 326–334 (2008).
Meeusen, E.N., Walker, J., Peters, A., Pastoret, P. P. & Jungersen, G. Current status of veterinary vaccines. Clin. Microbiol. Rev. 20, 489–510 (2007). A summary of the current status of the viral vaccines that are in veterinary use.
Poulet, H. et al. Development and registration of recombinant veterinary vaccines. The example of the canarypox vector platform. Vaccine 25, 5606–5612 (2007).
Swayne, D. E. & Kapczynski, D. Strategies and challenges for eliciting immunity against avian influenza virus in birds. Immunol. Rev. 225, 314–331 (2008).
Steel, J. et al. A combination in-ovo vaccine for avian influenza virus and Newcastle disease virus. Vaccine 26, 522–531 (2008).
Kitching, P. et al. Global FMD control — is it an option? Vaccine 25, 5660–5664 (2007).
Pacheco, J. M., Brum, M. C., Moraes, M. P., Golde, W. T. & Grubman, M.J. Rapid protection of cattle from direct challenge with foot-and-mouth disease virus (FMDV) by a single inoculation with an adenovirus-vectored FMDV subunit vaccine. Virology 337, 205–209 (2005).
Pena, L. et al. Delivery of a foot-and-mouth disease virus empty capsid subunit antigen with nonstructural protein 2B improves protection of swine. Vaccine 26, 5689–5699 (2008).
Minassian, A. M. & McShane, H. Tuberculosis vaccines: present and future. Expert Rev. Respir. Med. 2, 721–738 (2008).
Vordermeier, H. M., Huygen, K., Singh, M., Hewinson, R. G. & Xing, Z. Immune responses induced in cattle by vaccination with a recombinant adenovirus expressing mycobacterial antigen 85A and Mycobacterium bovis BCG. Infect. Immun. 74, 1416–1418 (2006).
Vordermeier, H. M. et al. Cellular immune responses induced in cattle by heterologous prime–boost vaccination using recombinant viruses and bacille Calmette-Guerin. Immunology 112, 461–470 (2004).
Buddle, B. M. et al. Cattle as a model for development of vaccines against human tuberculosis. (Tuberculosis Edinb.) 85, 19–24 (2005).
Limbach, K. J. & Richie, T. L. Viral vectors in malaria vaccine development. Parasite Immunol. 31, 501–519 (2009).
Draper, S. J. et al. Recombinant viral vaccines expressing merozoite surface protein-1 induce antibody- and T cell-mediated multistage protection against malaria. Cell Host Microbe 5, 95–105 (2009).
Hill, A. V. Pre-erythrocytic malaria vaccines: towards greater efficacy. Nature Rev. Immunol. 6, 21–32 (2006).
Nwanegbo, E. et al. Prevalence of neutralizing antibodies to adenoviral serotypes 5 and 35 in the adult populations of The Gambia, South Africa, and the United States. Clin. Diagn. Lab. Immunol. 11, 351–357 (2004).
Bejon, P. et al. A Phase 2b randomised trial of the candidate malaria vaccines FP9 ME-TRAP and MVA ME-TRAP among children in Kenya. PLoS Clin. Trials 1, e29 (2006).
McShane, H. et al. Recombinant modified vaccinia virus Ankara expressing antigen 85A boosts BCG-primed and naturally acquired antimycobacterial immunity in humans. Nature Med. 10, 1240–1244 (2004). The first clinical study to demonstrate that immune responses primed by BCG can be boosted using a viral-vector system encoding an antigen from mycobacteria.
Hawkridge, T. et al. Safety and immunogenicity of a new tuberculosis vaccine, MVA85A, in healthy adults in South Africa. J. Infect. Dis. 198, 544–552 (2008).
Sander, C. R. et al. Safety and immunogenicity of a new TB vaccine, MVA85A, in M. tuberculosis infected individuals. Am. J. Respir. Crit. Care Med. 179, 724–733 (2009).
Radosevic, K. et al. Protective immune responses to a recombinant adenovirus type 35 tuberculosis vaccine in two mouse strains: CD4 and CD8 T-cell epitope mapping and role of gamma interferon. Infect. Immun. 75, 4105–4115 (2007).
Magalhaes, I. et al. rBCG induces strong antigen-specific T cell responses in rhesus macaques in a prime-boost setting with an adenovirus 35 tuberculosis vaccine vector. PLoS ONE 3, e3790 (2008).
Hoft, D. F. et al. A new recombinant bacille Calmette-Guerin vaccine safely induces significantly enhanced tuberculosis-specific immunity in human volunteers. J. Infect. Dis. 198, 1491–1501 (2008).
Roukens, A. H. & Visser, L. G. Yellow fever vaccine: past, present and future. Expert Opin. Biol. Ther. 8, 1787–1795 (2008).
Pugachev, K. V., Guirakhoo, F., Trent, D. W. & Monath, T. P. Traditional and novel approaches to flavivirus vaccines. Int. J. Parasitol. 33, 567–582 (2003).
McAllister, A., Arbetman, A. E., Mandl, S., Pena-Rossi, C. & Andino, R. Recombinant yellow fever viruses are effective therapeutic vaccines for treatment of murine experimental solid tumors and pulmonary metastases. J. Virol. 74, 9197–9205 (2000).
Bonaldo, M. C. et al. Construction and characterization of recombinant flaviviruses bearing insertions between E and NS1 genes. Virol. J. 4, 115 (2007).
Monath, T. P. et al. Clinical proof of principle for ChimeriVax: recombinant live, attenuated vaccines against flavivirus infections. Vaccine 20, 1004–1018 (2002).
McMichael, A. J., Gotch, F. M., Noble, G. R. & Beare, P. A. Cytotoxic T-cell immunity to influenza. N. Engl. J. Med. 309, 13–17 (1983).
Heeney, J. L. & Plotkin, S. A. Immunological correlates of protection from HIV infection and disease. Nature Immunol. 7, 1281–1284 (2006).
Rerks-Ngarm, S. et al. Vaccination with ALVAC and AIDSVAX to prevent HIV-1 infection in Thailand. N. Engl. J. Med. 19 November 2009 (doi: 10.1056/NEJMoa0908492). The first HIV-1 vaccine Phase III clinical trial reporting a modest trend of protection from infection.
Thongcharoen, P. et al. A phase 1/2 comparative vaccine trial of the safety and immunogenicity of a CRF01_AE (subtype E) candidate vaccine: ALVAC-HIV (vCP1521) prime with oligomeric gp160 (92TH023/LAI-DID) or bivalent gp120 (CM235/SF2) boost. J. Acquir. Immune Defic. Syndr. 46, 48–55 (2007).
Sandstrom, E. et al. Broad immunogenicity of a multigene, multiclade HIV-1 DNA vaccine boosted with heterologous HIV-1 recombinant modified vaccinia virus Ankara. J. Infect. Dis. 198, 1482–1490 (2008).
Priddy, F. H. et al. Safety and immunogenicity of a replication-incompetent adenovirus type 5 HIV-1 clade B gag/pol/nef vaccine in healthy adults. Clin. Infect. Dis. 46, 1769–1781 (2008).
Hansen, S. G. et al. Effector memory T cell responses are associated with protection of rhesus monkeys from mucosal simian immunodeficiency virus challenge. Nature Med. 15, 293–299 (2009). This paper describes the use of CMV as an HIV-1 vaccine vector in macaques and shows that this vector can induce T cells with an effector memory phenotype that are associated with protection from a mucosal challenge with SIV.
Wilson, N. A. et al. Vaccine-induced cellular immune responses reduce plasma viral concentrations after repeated low-dose challenge with pathogenic simian immunodeficiency virus SIVmac239. J. Virol. 80, 5875–5885 (2006).
Casimiro, D. R. et al. Comparative immunogenicity in rhesus monkeys of DNA plasmid, recombinant vaccinia virus, and replication-defective adenovirus vectors expressing a human immunodeficiency virus type 1 gag gene. J. Virol. 77, 6305–6313 (2003).
Ward, S., Copier, J. & Dalgleish, A. Technical challenges facing therapeutic cancer vaccines. Curr. Opin. Drug Discov. Devel. 11, 168–177 (2008).
Emens, L. A. Cancer vaccines: on the threshold of success. Expert Opin. Emerg. Drugs 13, 295–308 (2008).
Hodge, J. W., Guha, C., Neefjes, J. & Gulley, J. L. Synergizing radiation therapy and immunotherapy for curing incurable cancers. Opportunities and challenges. Oncology 22, 1064–1070; discussion 1075, 1080–1081, 1084 (2008).
Kaufman, H. L. et al. Phase II trial of modified vaccinia Ankara (MVA) virus expressing 5T4 and high dose interleukin-2 (IL-2) in patients with metastatic renal cell carcinoma. J. Transl. Med. 7, 2 (2009).
Ramlau, R. et al. A phase II study of Tg4010 (Mva-Muc1-Il2) in association with chemotherapy in patients with stage III/IV non-small cell lung cancer. J. Thorac. Oncol. 3, 735–744 (2008).
Dreicer, R. et al. MVA-MUC1-IL2 vaccine immunotherapy (TG4010) improves PSA doubling time in patients with prostate cancer with biochemical failure. Invest. New Drugs 27, 379–386 (2009).
Quoix, E. et al. Randomized, controlled phase IIb trial evaluating the therapeutic vaccine TG4010 (MVA-MUC1-IL-2) as an adjunct to chemotherapy in patients with advanced non-small cell lung cancer (NSCLC). Ann. Oncol. 19, viii92 (2008).
Salucci, V. et al. CD8+ T-cell tolerance can be broken by an adenoviral vaccine while CD4+ T-cell tolerance is broken by additional co-administration of a Toll-like receptor ligand. Scand. J. Immunol. 63, 35–41 (2006).
Aurisicchio, L. et al. Immunogenicity and safety of a DNA prime/adenovirus boost vaccine against rhesus CEA in nonhuman primates. Int. J. Cancer 120, 2290–2300 (2007).
Domi, A. & Moss, B. Engineering of a vaccinia virus bacterial artificial chromosome in Escherichia coli by bacteriophage λ-based recombination. Nature Methods 2, 95–97 (2005).
Stanton, R. J., McSharry, B. P., Armstrong, M., Tomasec, P. & Wilkinson, G. W. Re-engineering adenovirus vector systems to enable high-throughput analyses of gene function. Biotechniques 45, 659–662, 664–668 (2008). This article shows how toxic transgenes can be switched off during the production of adenoviral-vector vaccines in cell culture, thereby enabling the growth and generation of recombinant AdV vaccines encoding such toxic proteins.
Paton, D. J. et al. Application of non-structural protein antibody tests in substantiating freedom from foot-and-mouth disease virus infection after emergency vaccination of cattle. Vaccine 24, 6503–6512 (2006).
Ko, S. Y. et al. Enhanced induction of intestinal cellular immunity by oral priming with enteric adenovirus 41 vectors. J. Virol. 83, 748–756 (2009).
Gallego-Gomez, J. C. et al. Differences in virus-induced cell morphology and in virus maturation between MVA and other strains (WR, Ankara, and NYCBH) of vaccinia virus in infected human cells. J. Virol. 77, 10606–10622 (2003).
Smith, G. L., Vanderplasschen, A. & Law, M. The formation and function of extracellular enveloped vaccinia virus. J. Gen. Virol. 83, 2915–2931 (2002).
Antoine, G., Scheiflinger, F., Dorner, F. & Falkner, F. G. The complete genomic sequence of the modified vaccinia Ankara strain: comparison with other orthopoxviruses. Virology 244, 365–396 (1998).
Gaggar, A., Shayakhmetov, D. M. & Lieber, A. CD46 is a cellular receptor for group B adenoviruses. Nature Med. 9, 1408–1412 (2003).
Cheng, C. et al. Mechanism of Ad5 vaccine immunity and toxicity: fiber shaft targeting of dendritic cells. PLoS Pathog. 3, e25 (2007).
Ghosh-Choudhury, G., Haj-Ahmad, Y., Brinkley, P., Rudy, J. & Graham, F. L. Human adenovirus cloning vectors based on infectious bacterial plasmids. Gene 50, 161–171 (1986).
Bett, A. J., Haddara, W., Prevec, L. & Graham, F. L. An efficient and flexible system for construction of adenovirus vectors with insertions or deletions in early regions 1 and 3. Proc. Natl Acad. Sci. USA 91, 8802–8806 (1994).
Smith, G. L. & Moss, B. Infectious poxvirus vectors have capacity for at least 25 000 base pairs of foreign DNA. Gene 25, 21–28 (1983).
Liu, X., Kremer, M. & Broyles, S. S. A natural vaccinia virus promoter with exceptional capacity to direct protein synthesis. J. Virol. Methods 122, 141–145 (2004).
Crucell & AERAS. Aeras and Crucell announce TB vaccine clinical trial in Kenya; promising vaccine candidate advances to Phase II safety study in South Africa. Crucell [online], (2008).
Acknowledgements
We thank A. Hill, M. Friede and G. Nabel for organizing the recent meeting 'Viral Vector Vaccines 2008' in Hinxton, which stimulated the writing of this Review. We thank A. Hill for comments on a draft of the manuscript and G.J. Nabel for providing the drawing on which figure 1 is based. J.L.H. is currently sponsored by grants from the Bill and Melinda Gates Foundation, USA, and the National Institutes of Health, USA.
Author information
Authors and Affiliations
Corresponding author
Related links
Glossary
- Reactogenicity
-
The capacity of a vaccine formulation to induce an adverse reaction following immunization.
- Ex vivo interferon-γ ELISPOT
-
An ELISPOT (enzyme-linked immunosorbent spot) assay that is used to measure antigen-specific effector T cell responses. Typically, splenocytes or peripheral blood mononuclear cells are incubated with the antigen (peptide or protein) for 18 hours, and the cytokines (typically interferon-γ) that are released during that time by antigen-specific cells are captured by a monoclonal antibody, thereby allowing enumeration by a modified enzyme-linked immunosorbent assay (ELISA) technique.
- Cultured interferon-γ ELISPOT
-
A modified ex vivo interferon-γ (IFN-γ) ELISPOT (enzyme-linked immunosorbent spot) assay that involves a prolonged period of in vitro culture of lymphocytes before the IFN-γ ELISPOT assay. Typically, cells are cultured for 10 days in the presence of antigen and supplemented with interleukin-2. Evidence indicates that a different population of T cells, most likely central memory T cells that differentiate into effector T cells during the culture period, are measured by this assay, as compared with the measurement of circulating effector T cells that are quantified by the immediate ex vivo ELISPOT.
- Heterologous prime–boost
-
Repeated immunization using different vaccines; used to stimulate a better immune response when a single application is not sufficient to induce a protective response.
- Pre-erythrocytic-stage malaria
-
Plasmodium sporozoites invade hepatocytes and develop over a period of 6–7 days. This time is known as the liver or pre-erythrocytic stage and precedes subsequent blood-stage infection. The liver stage is clinically silent and infected persons do not show any signs or symptoms of infection.
- Blood-stage malaria
-
During the liver stage of malaria infection, parasites develop into merozoites in infected liver cells. These rupture out into the blood after a period of 6–7 days and undergo a continuous cycle of asexual growth in erythrocytes, lasting approximately 48 hours.
- Spot-forming unit
-
(SFU). The ELISPOT assay is enumerated by a modified enzyme-linked immunosorbent assay (ELISA) technique. During the development stage of the assay, so-called 'spots' are formed on the membrane of the plates that are used. After counting the spots, results are routinely expressed as SFU per million cells, given that it is possible to calculate this from the number of cells known to have been used in the assay
- Carcinoembryonic antigen
-
(CEA). An oncofetal membrane glycoprotein and member of the immunoglobulin gene superfamily. CEA is expressed in fetal gastrointestinal tissue, but is also overexpressed in many human carcinomas of the colon, rectum, breast, lung and pancreas. CEA can be used as a serological marker of malignancy and is a tumour-associated antigen that is a widely used target for cancer vaccines.
Rights and permissions
About this article
Cite this article
Draper, S., Heeney, J. Viruses as vaccine vectors for infectious diseases and cancer. Nat Rev Microbiol 8, 62–73 (2010). https://doi.org/10.1038/nrmicro2240
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrmicro2240
This article is cited by
-
Phylogenetic analysis of prospective M. bovis antigens with the aim of developing candidate vaccines for bovine tuberculosis
Journal of Genetic Engineering and Biotechnology (2023)
-
Facile hermetic TEM grid preparation for molecular imaging of hydrated biological samples at room temperature
Nature Communications (2023)
-
Simple chitin-based cell culture platform for production of biopharmaceuticals
Biotechnology Letters (2023)
-
The use of viral vectors in vaccine development
npj Vaccines (2022)
-
Safety and immune response kinetics of GRAd-COV2 vaccine: phase 1 clinical trial results
npj Vaccines (2022)