Key Points
-
Anaerobic methane formation and anaerobic methane oxidation are important microbial processes in the global carbon cycle. Both processes are mediated by syntrophic communities of bacteria and archaea. In methane formation, bacteria degrade organic compounds to form products that are substrates for the methanogenic archaea. In anaerobic methane oxidation, methanotrophic archaea degrade methane and form currently unknown compounds that are used as electron donors by sulphate-reducing bacteria.
-
Hydrogen and formate are key components in interspecies electron transfer in facultative and obligate syntrophic methanogenic communities. In facultative syntrophy, anaerobic bacteria have an energetic advantage over the methanogens, but they are not essential for growth. In obligate syntrophic communities, bacteria and archaea degrade and grow on a substrate that each organism alone could not metabolize. This results in physical aggregation of bacteria and archaea, which may have led to the evolution of the first eukaryotic cell.
-
Bacteria that grow in obligate syntrophic association with methanogens live at the limits of what is thermodynamically possible. They encounter an energetic barrier in the recycling of redox mediators. NADH oxidation can be coupled to proton reduction only at low hydrogen concentrations, which are created by the methanogen. FADH2 oxidation coupled to proton reduction requires not only a low hydrogen concentration but also supplementary energy input from reverse electron transfer. Possible mechanisms of reverse electron transfer can be deduced from available genome sequences.
-
Simple substrates that are known typical substrates for methanogenic archaea, such as methanol, acetate and formate, can also be degraded by syntrophic communities of bacteria and archaea. Also, substrates that are considered easily fermentable might require syntrophic communities. This suggests that the anaerobic food chain in methanogenic environments is even more complex and more versatile than previously thought.
-
The occurrence of the anaerobic oxidation of methane coupled to sulphate reduction has been demonstrated in many studies. It is clear that syntrophic communities of methanotrophic archaea, which perform reverse methanogenesis, and sulphate-reducing bacteria are involved. The mechanism of this syntrophic interaction is unclear. Analogous to methanogenic communities, we propose that multiple compounds are involved.
Abstract
Interspecies electron transfer is a key process in methanogenic and sulphate-reducing environments. Bacteria and archaea that live in syntrophic communities take advantage of the metabolic abilities of their syntrophic partner to overcome energy barriers and break down compounds that they cannot digest by themselves. Here, we review the transfer of hydrogen and formate between bacteria and archaea that helps to sustain growth in syntrophic methanogenic communities. We also describe the process of reverse electron transfer, which is a key requirement in obligately syntrophic interactions. Anaerobic methane oxidation coupled to sulphate reduction is also carried out by syntrophic communities of bacteria and archaea but, as we discuss, the exact mechanism of this syntrophic interaction is not yet understood.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
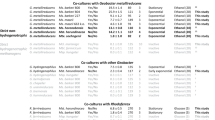
Breas, O., Guillou, C., Reniero, F. & Wada, E. The global methane cycle: isotopes and mixing ratios, sources and sinks. Isotop. Environ. Health Stud. 37, 257–379 (2002).
Falkowski, P. G., Fenchel, T. & deLong, E. F. The microbial engines that drive Earth's biogeochemical cycles. Science 280, 1034–1039 (2008).
Tilche, A. & Galatola, M. The potential of bio-methane as bio-fuel/bio-energy for reducing greenhouse gas emissions: a qualitative assessment for Europe in a life cycle perspective. Water Sci. Technol. 57, 1683–1692 (2008).
McCarty, P. L. The development of anaerobic treatment and its future. Water Sci. Technol. 44, 149–156 (2001).
Whitman, W., Bowen, T. & Boone D. in The Prokaryotes: an Evolving Electronic Resource for the Microbiological Community 3rd edn Vol. 3 (eds Dworkin, M., Falkow, S., Rosenberg, E., Schleifer, K. H. & Stackebrandt, E.) 165–207 (Springer, New York, 2006).
Stams, A. J. M. et al. Exocellular electron transfer in anaerobic microbial communities. Environ. Microbiol. 8, 371–382 (2006).
Schink, B. & Stams, A. J. M. in The Prokaryotes: an Evolving Electronic Resource for the Microbiological Community 3rd edn Vol. 2 (eds Dworkin, M., Falkow, S., Rosenberg, E., Schleifer, K. H. & Stackebrandt, E.) 309–335 (Springer, New York, 2006).
Reguera, G. et al. Extracellular electron transfer via microbial nanowires. Nature 435, 1098–1101 (2005). This article provided the first description of possible electron transfer through conductive pili.
Gorby, Y. A. et al. Electrically conductive bacterial nanowires produced by Shewanella oneidensis strain MR-1 and other microorganisms. Proc. Natl Acad. Sci. USA 103, 11358–11363 (2006).
Stams, A. J. M. Metabolic interactions between anaerobic bacteria in methanogenic environments. Antonie van Leeuwenhoek 66, 271–294 (1994).
Nealson, K. H., Inagaki, F. & Takai, K. Hydrogen-driven subsurface lithoautotrophic microbial ecosystems (SLiMEs): do they exist and why should we care? Trends Microbiol. 13, 405–410 (2005). Gives a description of the development of an entire food chain fueled by the biotransformation of H 2 and CO 2 in the absence of light.
Thiele, J. H. & Zeikus, J. G. Control of interspecies electron flow during anaerobic digestion: significance of formate transfer versus hydrogen transfer during syntrophic methanogenesis in flocs. Appl. Environ. Microbiol. 54, 20–29 (1988).
Conrad, R., Phelps, T. J. & Zeikus, J. G. Gas metabolism evidence in support of the juxtaposition of hydrogen-producing and methanogenic bacteria in sewage sludge and lake sediments. Appl. Environ. Microbiol. 50, 595–601 (1985).
Schink, B. & Thauer, R. K. in Granular Anaerobic Sludge: Microbiology and Technology (eds Lettinga, G., Zehnder, A. J. B., Grotenhuis, J. T. C. & Hulshoff, L. W.) 5–17 (Pudoc, Wageningen, The Netherlands, 1988).
Ishii, S., Kosaka, T., Hori, K., Hotta, Y. & Watanabe, K. Coaggregation facilitates interspecies hydrogen transfer between Pelotomaculum thermopropionicum and Methanothermobacter thermautotrophicus. Appl. Environ. Microbiol. 71, 7838–7845 (2005).
Jackson, B. E. & McInerney, M. J. Anaerobic microbial metabolism can proceed close to thermodynamic limits. Nature 415, 454–456 (2002).
McInerney, M. J. et al. Physiology, ecology, phylogeny, and genomics of microorganisms capable of syntrophic metabolism. Ann. NY Acad. Sci. 1125, 58–72 (2008).
Martin, W. & Muller, M. The hydrogen hypothesis for the first eukaryote. Nature 392, 37–41 (1998).
Boone, D. R., Johnson, R. L. & Liu, Y. Diffusion of the interspecies electron carriers H2 and formate in methanogenic ecosystems, and implications in the measurement of KM for H2 or formate uptake. Appl. Environ. Microbiol. 55, 1735–1741 (1989).
Conrad, R., Schink, B. & Phelps, T. J. Thermodynamics of H2-consuming and H2-producing metabolic reactions in diverse methanogenic environments under in situ conditions. FEMS Microbiol. Ecol. 38, 353–360 (1986).
Vignais, P. M. & Billoud, B. Occurrence, classification, and biological function of hydrogenases: an overview. Chem. Rev. 107, 4206–4272 (2007).
Hedderich, R. & Forzi, L. Energy-converting [NiFe] hydrogenases: more than just H2 activation. J. Mol. Microbiol. Biotechnol. 10, 92–104 (2005). Shows that proton translocation by membrane-bound hydrogenases is a method of energy conservation that is important in anaerobic microbial communities.
Casalot, L. & Rousset, M. Maturation of the [NiFe] hydrogenases. Trends Microbiol. 9, 228–237 (2001).
Thauer, R. K., Jungermann, K. & Decker, K. Energy conservation in chemotrophic anaerobic bacteria. Bacteriol. Rev. 41, 100–180 (1977).
Thauer, R. K., Kaster, A. K., Seedorf, H., Buckel, W. & Hedderich, R. Methanogenic archaea: ecologically relevant differences in energy conservation. Nature Rev. Microbiol. 6, 579–591 (2008).
Carepo, M. et al. Hydrogen metabolism in Desulfovibrio desulfuricans strain New Jersey (NCIMB 8313) — comparative study with D. vulgaris and D. gigas species. Anaerobe 8, 325–332 (2002).
Bagramyan, K. & Trchounian, A. Structural and functional features of formate hydrogen lyase, an enzyme of mixed-acid fermentation from Escherichia coli. Biochem. (Mosc.) 68. 1159–1170 (2003).
Bott, M. Anaerobic citrate metabolism and its regulation in enterobacteria. Arch. Microbiol. 167, 78–88 (1997).
Meshulam-Simon, G., Behrens, S., Choo, A. D. & Spormann, A. M. Hydrogen metabolism in Shewanella oneidensis MR-1. Appl. Environ. Microbiol. 73, 1153–1165 (2007).
Sawers, R. G. Formate and its role in hydrogen production in Escherichia coli. Biochem. Soc. Trans. 33, 42–46 (2005).
Hulshoff Pol, L. W., deCastro Lopes, S, I., Lettinga, G. & Lens, P. N. L. Anaerobic sludge granulation. Water Res. 38, 1376–1389 (2004).
Lettinga, G. et al. Use of the upflow sludge blanket reactor concept for biological waste water treatment, especially for anaerobic treatment. Biotechnol. Bioeng. 22, 699–734 (1980). Shows that the spontaneous self-aggregation of mixed methanogenic communities in upward-flow bioreactors to compact and dense aggregates enables efficient anaerobic wastewater treatment.
Liu, Y., Xu, H. L., Yang, S. F. & Tay, J. H. Mechanisms and models for anaerobic granulation in upflow anaerobic sludge blanket reactor. Water Res. 37, 661–673 (2003).
Grotenhuis, J. T. et al. Bacteriological composition and structure of granular sludge adapted to different substrates. Appl. Environ. Microbiol. 57, 1942–1949 (1991).
Schmidt, J. E. & Ahring, B. K. Effects of hydrogen and formate on the degradation of propionate and butyrate in thermophilic granules from an upflow anaerobic sludge blanket reactor. Appl. Environ. Microbiol. 59, 2546–2551 (1993).
Stams, A. J. M., Grolle, K. C., Frijters, C. T. & Van Lier, J. B. Enrichment of thermophilic propionate-oxidizing bacteria in syntrophy with Methanobacterium thermoautotrophicum or Methanobacterium thermoformicicum. Appl. Environ. Microbiol. 58, 346–352 (1992).
Ozturk, S. S., Palsson, B. O. & Thiele, J. H. Control of interspecies electron transfer flow during anaerobic digestion: dynamic diffusion reaction models for hydrogen gas transfer in microbial flocs. Biotechnol. Bioeng. 33, 745–757 (1989).
Schmidt, J. E. & Ahring, B. K. Interspecies electron transfer during propionate and butyrate degradation in mesophilic, granular sludge. Appl. Environ. Microbiol. 61, 2765–2767 (1995).
Krumholz, L. R. & Bryant, M. P. Syntrophococcus sucromutans sp. nov. gen. nov. uses carbohydrates as electron donors and formate, methoxymonobenzenoids or Methanobrevibacter as electron acceptor systems. Arch. Microbiol. 143, 313–318 (1986). Shows that substrates that are considered easily fermentable may be degraded by obligately syntrophic communities of bacteria and methanogens.
Müller, N., Griffin, B. M., Stingl, U. & Schink, B. Dominant sugar utilizers in sediment of Lake Constance depend on syntrophic cooperation with methanogenic partner organisms. Environ. Microbiol. 10, 1501–1511 (2008).
Jackson, B. E., Bhupathiraju, V. K., Tanner, R. S., Woese, C. R. & McInerney, M. J. Syntrophus aciditrophicus sp. nov., a new anaerobic bacterium that degrades fatty acids and benzoate in syntrophic association with hydrogen-using microorganisms. Arch. Microbiol. 171, 107–114 (1999).
McInerney, M. J. et al. The genome of Syntrophus aciditrophicus: life at the thermodynamic limit of microbial growth. Proc. Natl Acad. Sci. USA 104, 7600–7605 (2007).
Imachi, H. et al. Pelotomaculum thermopropionicum gen. nov., sp. nov., an anaerobic, thermophilic, syntrophic propionate-oxidizing bacterium. Int. J. Syst. Evol. Microbiol. 52, 1729–1735 (2002).
Kosaka, T. et al. Reconstruction and regulation of the central catabolic pathway in the thermophilic propionate-oxidizing syntroph Pelotomaculum thermopropionicum. J. Bacteriol. 188, 202–210 (2006).
Kosaka, T. et al. The genome of Pelotomaculum thermopropionicum reveals niche-associated evolution in anaerobic microbiota. Genome Res. 18, 442–448 (2008).
Harmsen, H. J. M. et al. Syntrophobacter fumaroxidans sp. nov., a syntrophic propionate-degrading sulfate-reducing bacterium. Int. J. Syst. Bacteriol. 48, 1383–1387 (1998).
Nakanishi-Matsui, M. & Futai, M. Stochastic rotational catalysis of proton pumping F-ATPase. Philos. Trans. R. Soc. Lond. B Biol. Sci. 363, 2135–2142 (2008). Shows that the stoichiometry of proton translocation and ATP hydrolysis or ATP synthesis can vary.
Stams, A. J. M., Van Dijk, J. B., Dijkema, C. & Plugge, C. M. Growth of syntrophic propionate-oxidizing bacteria with fumarate in the absence of methanogenic bacteria. Appl. Environ. Microbiol. 59, 1114–1119 (1993).
Kröger, A. et al. Fumarate respiration of Wolinella succinogenes: enzymology, energetics and coupling mechanism. Biochim. Biophys. Acta 1553, 23–38 (2002).
Schirawski, J. & Unden, G. Menaquinone-dependent succinate dehydrogenase of bacteria catalyzes reversed electron transport driven by the proton potential. Eur. J. Biochem. 257, 210–215 (1998).
Van Kuijk, B. L. M., Schlösser, E. & Stams, A. J. M. Investigation of the fumarate metabolism of the syntrophic propionate-oxidizing bacterium strain MPOB. Arch. Microbiol. 169, 346–352 (1998).
Wallrabenstein, C. & Schink, B. Evidence of reversed electron transport in syntrophic butyrate or benzoate oxidation by Syntrophomonas wolfei and Syntrophus buswellii. Arch. Microbiol. 162, 136–142 (1994).
Schink, B. & Friedrich, M. Energetics of syntrophic fatty acid oxidation. FEMS Microbiol. Rev. 15, 85–94 (1994).
Herrmann, G., Jayamani, E., Mai, G. & Buckel, W. Energy conservation via electron-transferring flavoprotein in anaerobic bacteria. J. Bacteriol. 190, 784–791 (2008). Provides a description of a biochemical mechanism of energy conservation involving high and low potential redox mediators.
Li, F. et al. Coupled ferredoxin and crotonyl coenzyme A (CoA) reduction with NADH catalyzed by the butyryl-CoA dehydrogenase/Etf complex from Clostridium kluyveri. J. Bacteriol. 190, 843–850 (2008).
Plugge, C. M., Dijkema, C. & Stams, A. J. M. Acetyl-CoA cleavage pathway in a syntrophic propionate oxidizing bacterium growing on fumarate in the absence of methanogens. FEMS Microbiol. Lett. 110, 71–76 (1993).
Wofford, N. Q., Beaty, P. S. & McInerney, M. J. Preparation of cell-free extracts and the enzymes involved in fatty acid metabolism in Syntrophomonas wolfei. J. Bacteriol. 167, 179–185 (1986).
Dong, X. & Stams, A. J. M. Evidence for H2 and formate formation during syntrophic butyrate and propionate degradation. Anaerobe 1, 35–39 (1995).
Dong, X., Plugge, C. M. & Stams, A. J. M. Anaerobic degradation of propionate by a mesophilic acetogenic bacterium in coculture and triculture with different methanogens. Appl. Environ. Microbiol. 60, 2834–2838 (1994).
De Bok, F. A. M., Roze, E. H. & Stams, A. J. M. Hydrogenases and formate dehydrogenases of Syntrophobacter fumaroxidans. Antonie van Leeuwenhoek 81, 283–291 (2002).
De Bok, F. A. M. et al. Two W-containing formate dehydrogenases (CO2-reductases) involved in syntrophic propionate oxidation by Syntrophobacter fumaroxidans. Eur. J. Biochem. 270, 2476–2485 (2003).
Andreesen, J. R. & Makdessi, K. Tungsten, the surprisingly positively acting heavy metal element for prokaryotes. Ann. NY Acad. Sci. 1125, 215–229 (2008).
Plugge, C. M., Balk, M. & Stams, A. J. M. Desulfotomaculum thermobenzoicum subsp. thermosyntrophicum subsp. nov., a thermophilic, syntrophic, propionate-oxidizing, spore-forming bacterium. Int. J. Syst. Evol. Microbiol. 52, 391–399 (2002).
Sieber, J. et al. in Abstr. Gen. Meet. Am. Soc. Microbiol. I-002,071 (2008).
Dong, X. & Stams, A. J. M. Localization of the enzymes involved in H2 and formate metabolism in Syntrophospora bryantii. Antonie van Leeuwenhoek 67, 345–350 (1995).
Bryant, M. P., Campbell, L. L., Reddy, C. A. & Crabill, M. R. Growth of Desulfovibrio in lactate or ethanol media low in sulfate in association with H2-utilizing methanogenic bacteria. Appl. Environ. Microbiol. 33, 1162–1169 (1977).
Scholten, J. C. et al. Evolution of the syntrophic interaction between Desulfovibrio vulgaris and Methanosarcina barkeri: involvement of an ancient horizontal gene transfer. Biochem. Biophys. Res. Commun. 352, 48–54 (2007).
Winter, J. & Wolfe, R. S. Methane formation from fructose by syntrophic associations of Acetobacterium woodii and different strains of methanogens. Arch. Microbiol. 124, 73–79 (1980).
Cord-Ruwisch, R. & Ollivier, B. Interspecific hydrogen transfer during methanol degradation by Sporomusa acidovorans and hydrogenophilic anaerobes. Arch. Microbiol. 144, 163–165 (1986).
Phelps, T. J., Conrad, R. & Zeikus, J. G. Sulfate-dependent interspecies H2 transfer between Methanosarcina barkeri and Desulfovibrio vulgaris during coculture metabolism of acetate or methanol. Appl. Environ. Microbiol. 50, 589–594 (1985).
Valentine, D. L., Blanton, D. C. & Reeburgh, W. S. Hydrogen production by methanogens under low-hydrogen conditions. Arch. Microbiol. 174, 415–421 (2000).
Calteau, A., Gouy, M. & Perrière, G. Horizontal transfer of two operons coding for hydrogenases between bacteria and archaea. J. Mol. Evol. 60, 557–565 (2005).
Stolyar, S. et al. Metabolic modeling of a mutualistic microbial community. Mol. Syst. Biol. 3,92 (2007).
Reeburgh, W. S. Methane consumption in Cariaco Trench waters and sediments. Earth Planet. Sci. Lett. 28, 337–344 (1976). This article proposed for the first time that methane is oxidized anaerobically.
Boetius, A. et al. A marine microbial consortium apparently mediating anaerobic oxidation of methane. Nature 407, 623–626 (2000). Describes a syntrophic community of archaea and bacteria involved in sulphate-dependent anaerobic methane oxidation.
Raghoebarsing, A. A. et al. A microbial consortium couples anaerobic methane oxidation to denitrification. Nature 440, 918–921 (2006). Describes AOM by a denitrifying microbial community.
Ettwig, K. F. et al. Denitrifying bacteria anaerobically oxidize methane in the absence of Archaea. Environ. Microbiol. 10, 3164–3173 (2008).
Hallam, S. J. et. al. Reverse methanogenesis: testing the hypothesis with environmental genomics. Science 305, 1457–1462 (2004).
Krüger, M. et al. A conspicuous nickel protein in microbial mats that oxidize methane anaerobically. Nature 426, 878–881 (2003). Shows the purification and characterization of a key enzyme of anaerobic methane oxidation from sediments in which anaerobic methane oxidation is occurring.
Mayr, S. et al. Structure of an F430 variant from archaea associated with anaerobic oxidation of methane. J. Am. Chem. Soc. 130, 10758–10767 (2008).
Friedrich, M. W. Methyl-coenzyme M reductase genes: unique functional markers for methanogenic and anaerobic methane-oxidizing Archaea. Methods Enzymol. 397, 428–442 (2005).
Nauhaus, K., Treude, T., Boetius, A. & Krüger, M. Environmental regulation of the anaerobic oxidation of methane: a comparison of ANME-I and ANME-II communities. Environ. Microbiol. 1, 98–106 (2005).
Treude, T. et al. Consumption of methane and CO2 by methanotrophic microbial mats from gas seeps of the anoxic Black Sea. Appl. Environ. Microbiol. 73, 2271–2283 (2007).
Sørensen, K. B., Finster, K. & Ramsing N. B. Thermodynamic and kinetic requirements in anaerobic methane oxidizing consortia exclude hydrogen, acetate, and methanol as possible electron shuttles. Microb. Ecol. 42, 1–10 (2001).
Moran, J. J. et al. Methyl sulfides as intermediates in the anaerobic oxidation of methane. Environ. Microbiol. 10, 162–173 (2007).
Thauer, R. K. & Shima, S. Methane as fuel for anaerobic organisms. Ann. NY Acad. Sci. 1125, 158–170 (2008).
Orcutt, B., Samarkin, V., Boetius, A. & Joye, S. On the relationship between methane production and oxidation by anaerobic methanotrophic communities from cold seeps of the Gulf of Mexico. Environ. Microbiol. 10, 1108–1117 (2008).
Niemann, H. et al. Novel microbial communities of the Haakon Mosby mud volcano and their role as a methane sink. Nature 443, 854–858 (2006).
Lösekann, T. et al. Diversity and abundance of aerobic and anaerobic methane oxidizers at the Haakon Mosby mud volcano, Barents Sea. Appl. Environ. Microbiol. 73, 3348–3362 (2007).
Nauhaus, K., Albrecht, M., Elvert, M., Boetius, A. & Widdel, F. In vitro cell growth of marine archaeal-bacterial consortia during anaerobic oxidation of methane with sulfate. Environ. Microbiol. 9, 187–196 (2007).
Orphan, V. J., House, C. H., Hinrichs, K. U., McKeegan, K. D. & DeLong, E. F. Multiple archaeal groups mediate methane oxidation in anoxic cold seep sediments. Proc. Natl Acad. Sci. USA 99, 7663–7668 (2002).
Pernthaler, A. et al. Diverse syntrophic partnerships from deep-sea methane vents revealed by direct cell capture and metagenomics. Proc. Natl Acad. Sci. USA 105, 7052–7057 (2008).
Michaelis, W. et al. Microbial reefs in the Black Sea fueled by anaerobic oxidation of methane. Science 297, 1013–1015 (2002).
Orphan, V. J. et al. Comparative analysis of methane-oxidizing archaea and sulfate-reducing bacteria in anoxic marine sediments. Appl. Environ. Microbiol. 67, 1922–1934 (2001).
Finke, N., Hoehler, T. M. & Jørgensen, B. B. Hydrogen 'leakage' during methanogenesis from methanol and methylamine: implications for anaerobic carbon degradation pathways in aquatic sediments. Environ. Microbiol. 9, 1060–1071 (2007).
Keltjens, J. T. & van der Drift, C. Electron transfer reactions in methanogens. FEMS Microbiol. Rev. 39, 259–303 (1986).
Rother, M., Oelgeschläger, E. & Metcalf, W. M. Genetic and proteomic analyses of CO utilization by Methanosarcina acetivorans. Arch. Microbiol. 188, 463–472 (2007).
Henstra, A. M., Dijkema, C. & Stams, A. J. M. Archaeoglobus fulgidus couples CO oxidation to sulfate reduction and acetogenesis with transient formate accumulation. Environ. Microbiol. 9, 1836–1841 (2007).
Guss, A. M., Mukhopadhyay, B., Zhang, J. K. & Metcalf, W. W. Genetic analysis of mch mutants in two Methanosarcina species demonstrates multiple roles for the methanopterin-dependent C-1 oxidation/reduction pathway and differences in H2 metabolism between closely related species. Mol. Microbiol. 55, 1671–1680 (2005). Shows the role of hydrogen metabolism during growth on different substrates through the analysis of Methanosarcina mutants.
Rabus, R., Hansen, T. A. & Widdel, F. in The Prokaryotes: an Evolving Electronic Resource for the Microbiological Community 3rd edn Vol. 2 (eds Dworkin, M., Falkow, S., Rosenberg, E., Schleifer, K. H. & Stackebrandt, E.) 659–768 (Springer, New York, 2006).
Wegener, G., Niemann, H., Elvert, M., Hinrichs, K. U. & Boetius, A. Assimilation of methane and inorganic carbon by microbial communities mediating the anaerobic oxidation of methane. Environ. Microbiol. 10, 2287–2298 (2008).
Lupa, B., Hendrickson, E. L., Leigh, J. A. & Whitman, W. B. Formate-dependent H2 production by the mesophilic methanogen Methanococcus maripaludis. Appl. Environ. Microbiol. 74, 6584–6590 (2008).
Sprenger, W. W., Hackstein, J. H. & Keltjens, J. T. The energy metabolism of Methanomicrococcus blatticola: physiological and biochemical aspects. Antonie van Leeuwenhoek 87, 289–299 (2005).
López-García, P. & Moreira, D. Tracking microbial biodiversity through molecular and genomic ecology. Res. Microbiol. 159, 67–73 (2008).
Pisani, D., Cotton, J. A. & McInerney, J. O. Supertrees disentangle the chimerical origin of eukaryotic genomes. Mol. Biol. Evol. 24, 1752–1760 (2007).
Searcy, D. G. Metabolic integration during the evolutionary origin of mitochondria. Cell Res. 13, 229–238 (2003).
Barker, H. A. Studies upon the methane fermentation. IV: the isolation and culture of Methanobacterium omelianskii. Antonie van Leeuwenhoek 6, 201–220 (1940).
Brill, W. J. & Wolfe, R. S. Acetaldehyde oxidation by Methanobacillus — a new ferredoxin-dependent reaction. Nature 212, 253–255 (1966).
Bryant, M. P., Wolin, E. A., Wolin, M. J. & Wolfe, R. S. Methanobacillus omelianskii, a symbiotic association of two species of bacteria. Arch. Mikrobiol. 59, 20–31 (1967).
de Bruyn, J. C., Boogerd, F. C., Bos, P. & Kuenen, J. G. Floating filters, a novel technique for isolation and enumeration of fastidious, acidophilic, iron-oxidizing, autotrophic bacteria. Appl. Environ. Microbiol. 56, 2891–2894 (1990).
Ianotti, E. L., Kafkewitz, D., Wolin, M. J. & Bryant, M. P. Glucose fermentation products by Ruminococcus albus grown in continuous culture with Vibrio succinogenes: changes caused by interspecies transfer of H2 . J. Bacteriol. 114, 1231–1240 (1973).
Chen, M. & Wolin, M. J. Influence of CH4 production by Methanobacterium ruminantium on the fermentation of glucose and lactate by Selenomonas ruminantium. Appl. Environ. Microbiol. 34, 756–759 (1977).
Latham, M. J. & Wolin, M. J. Fermentation of cellulose by Ruminococcus flavefaciens in the presence and absence of Methanobacterium ruminantium. Appl. Environ. Microbiol. 34, 97–301 (1977).
Acknowledgements
Our research was supported by grants of the divisions of Chemical Sciences, Earth and Life Sciences and the Technology Foundation of the Netherlands Science Foundation and the Darwin Center for Biogeology.
Author information
Authors and Affiliations
Corresponding author
Related links
Glossary
- Methanogenic environment
-
An anoxic environment in which organic matter is degraded and protons and CO2 act as the main electron acceptors. The limited range of substrates that are used by methanogenic archaea results in a syntrophic cooperation with microorganisms that degrade more complex organic compounds and form substrates for methanogens.
- Syntrophic
-
A nutritional situation in which two or more organisms combine their metabolic capabilities to catabolize a substrate that cannot be catabolized by either one of them alone.
- Reducing equivalent
-
Any kind of reduced redox mediator that is formed by the oxidation of organic or inorganic compounds.
- Midpoint redox potential
-
The quantitative expression of the electrochemical property of redox-active compounds, relative to the redox couple H+/H2. A solution of 1 M H+ saturated with H2 at atmospheric pressure has a redox potential (E°) of 0 V. The E°′ at pH 7 is −0.414 V.
- Standard Gibbs free energy change
-
(ΔG°′). The amount of energy that is released or needed in a chemical conversion. The standard conditions refer to 1 M for solutes, 105 Pa (1 atm) for gases, 298 K and a pH of 7. A reaction in which energy is released is an exergonic reaction and a reaction that requires energy is an endergonic reaction. ΔG°′ values are expressed as kJ mol−1.
- Substrate level phosphorylation
-
The synthesis of high-energy phosphate bonds through the reaction of inorganic phosphate with an activated organic substrate. For fermentative bacteria, it is often the sole biochemical mechanism of energy conservation.
- High-rate methanogenic bioreactor
-
A reactor that is used for the anaerobic treatment of industrial wastewaters with a high concentration of organic compounds. High loading rates can be applied because the biomass is present as dense aggregates (granular sludge) that allow uncoupling of the liquid retention time from the biomass retention time.
- Reverse electron transport
-
The biochemical mechanism by which microorganisms can perform a chemical transformation that is endergonic under the prevailing conditions. It resembles the electron transport-driven mechanism of energy conservation from an exergonic reaction but operates in reverse.
- Menaquinone
-
Abbreviation for methylnaphthoquinone, an electron carrier in the cytoplasmic membrane of many bacteria and archaea. Reduction by one electron yields the menasemiquinone anion, which on further reduction by a second electron takes two protons and thus forms menahydroquinone (also called menaquinol).
- Electron bifurcation
-
Separation of the two electrons from ubiquinol at the quinol oxidation site of the bc1 complex (complex III) of the respiratory chain, which leads to a bifurcation of the two electrons to a high and a low potential pathway.
- Homoacetogen
-
An anaerobic bacterium that can grow on H2 and CO2, forming acetate as a product. As homoacetogenesis is conditional, homoacetogens are also known as acetogens.
Rights and permissions
About this article
Cite this article
Stams, A., Plugge, C. Electron transfer in syntrophic communities of anaerobic bacteria and archaea. Nat Rev Microbiol 7, 568–577 (2009). https://doi.org/10.1038/nrmicro2166
Issue Date:
DOI: https://doi.org/10.1038/nrmicro2166
This article is cited by
-
Experimental study on the inhibition of glucose and olive mill wastewater degradation by volatile fatty acids in anaerobic digestion
Euro-Mediterranean Journal for Environmental Integration (2024)
-
H2 generated by fermentation in the human gut microbiome influences metabolism and competitive fitness of gut butyrate producers
Microbiome (2023)
-
Steering the product spectrum in high-pressure anaerobic processes: CO2 partial pressure as a novel tool in biorefinery concepts
Biotechnology for Biofuels and Bioproducts (2023)
-
Inter-kingdom interactions and stability of methanogens revealed by machine-learning guided multi-omics analysis of industrial-scale biogas plants
The ISME Journal (2023)
-
Methylotrophic methanogens and bacteria synergistically demethylate dimethylarsenate in paddy soil and alleviate rice straighthead disease
The ISME Journal (2023)